Abstract
Purpose
The purpose of the current investigation was to characterize biomechanical differences between the supra- and infra-meniscal sections of the anterolateral ligament (ALL). We hypothesized that the supra-meniscal fibers of the ALL would be stronger and stiffer than the infra-meniscal fiber.
Methods
Nine cadaveric knee specimens [mean (SD) age = 79 (14.6) years] were dissected to identify the borders of the ALL while maintaining the anatomy of the lateral meniscus. The specimens were randomly assigned to either a supra-meniscal (the ALL below the meniscus was sectioned leaving only the supra-meniscal ALL intact) or an infra-meniscal (the ALL above the meniscus was sectioned leaving only the infra-meniscal attachment intact) group. The specimens were potted into dental cement such that the ALL was pulling laterally on the meniscus when the specimens were secured within an Instron materials testing machine. The specimens were subjected to a tensile failure test at 1 mm/s. The load at failure and stiffness were calculated from the force–displacement curves, while peak stress was calculated by normalizing the peak force to the cross-sectional area of the ALL. Furthermore, one intact knee specimen was used to perform a histological analysis on the two ALL sections using Masson’s Trichome staining.
Results
The infra-meniscal ALL had a significantly (p = 0.03) higher load to failure (195.0 vs. 132.1 N) and was significantly (p = 0.03) stiffer than the supra-meniscal fibers (24.8 vs. 12.3 N/mm). The relatively similar cross-section areas also resulted in the infra-meniscal sections having a greater peak stress (p = 0.04) (11.1 vs. 5.4 MPa). Histological analysis showed relatively consistent fiber orientation with similar organization noted throughout the fibers.
Conclusions
The ALL-meniscal construct that includes the infra-meniscal fibers was significantly stronger and stiffer than the construct that includes the supra-meniscal fibers. The infra-meniscal ALL is another important component of the anterolateral complex of the knee, and should be considered when presented with an ACL and/or meniscal injury.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recent investigations have suggested that injury to the anterolateral ligament (ALL), an extra-articular structure within the anterolateral capsule of the knee [1–6], is associated with injury to the anterior cruciate ligament (ACL) and the lateral meniscus [7, 8]. Investigators have proposed that this association is secondary to the ALL’s anatomic connection to the lateral meniscus [9–11]. Posterior root tears of the lateral meniscus are commonly observed with ACL rupture [12] and this may be related to the strong attachment of the ALL to the lateral tibial plateau [9]. Furthermore, Vincent et al. [11] have suggested that the additional tibial translation that occurs following an avulsion of the anterolateral capsule and iliotibial band (Segond fracture) from the proximal lateral tibia, could increase the tension within the lateral meniscus due to the pull of the ALL’s intact femoral origin.
Heltio et al. [9] also observed that the ALL can be divided into two distinct sections: supra-meniscal fibers that run from the femoral origin to the meniscus, and infra-meniscal fibers that extend from the meniscus to the tibial insertion. Given the ALL’s proposed role in assisting in the control of rotational laxity during internal rotation of the knee [6, 12–17], it is possible that the two ALL sections have different biomechanical functions [9]. Although past research has quantified the mechanical properties of the ALL as a whole [4, 18], no investigation has determined whether the different ALL sections with respect to their attachment to the lateral meniscus have different biomechanical functions that would ultimately affect the contribution the ALL makes to anterolateral stability of the knee.
Furthermore, previous histological analysis has shown that the ALL is composed of dense regular connective tissue which is characteristic of ligamentous structures [1, 19] and primarily consists of Type I collagen [20]. However, to our knowledge a comparison has not been performed between the two ALL sections (infra-meniscal and supra-meniscal). Therefore, the primary purpose of this study was to determine if there are biomechanical and histological differences between the supra-and infra-meniscal fibers of the ALL. It was hypothesized that the supra-meniscal attachment would be stronger than the infra-meniscal section, given the higher rate of injury seen in the infra-meniscal section compared to the supra-meniscal fibers. However, it was also hypothesized that there would be no differences in the morphological appearance between the two sections as shown histologically.
Materials and methods
The approval for the use of cadaveric material was granted by the Committee for Cadaveric Use in Research, Division of Clinical Anatomy, Western University in accordance with the Anatomy Act of Ontario, Canada (Approval No. 10032014). Fourteen fresh-frozen cadaveric knee specimens [seven males; mean (SD) age of 79 (14) years] were thawed at room temperature for 24 h prior to dissection and testing. Once thawed, the skin was removed from the knee and the IT band was identified and reflected inferiorly to its insertion, making sure that the anterolateral capsule was preserved throughout. With the knee placed in 45° of flexion, a varus and internal rotational moment was applied placing the ALL fibers under tension, thus allowing better visualization and identification of the ALL. The ALL was defined as any tissue running from the lateral femoral epicondyle (or just posterior and proximal to it), to the lateral aspect of the tibia, just below the tibial plateau and midway between Gerdy’s tubercle and the fibular head [21]. The borders of the ALL were defined and the lateral collateral ligament (LCL) was isolated by blunt dissection due to its shared origin at the lateral femoral epicondyle. All of the soft tissues, except the ALL and lateral meniscus were removed from the knee using both an extra-articular and intra-articular dissection approach to fully isolate the ALL from the tissue and joint capsule on the lateral aspect of the knee. The popliteal tendon was dissected off the inner aspect of the ALL and resected along with the meniscofemoral ligaments. Finally, the LCL was sectioned and removed along with any remaining tissue posterior to it. Using a digital caliper (EZcal iGaging; San Clemente CA), mid-substance width and thickness measurements of the ALL were taken superior and inferior to the meniscus to calculate cross-sectional area [1, 2, 9]. Both the thickness and width measurements were taken three times and an assessment of the repeatability was performed.
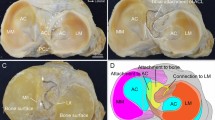
The specimens were randomly assigned to either a supra-meniscal or an infra-meniscal group. The supra-meniscal specimens (n = 7) were prepared by sectioning through the middle of the femur, leaving the tibial plateau and the lateral femoral condyle intact (Fig. 1a). Any ALL fibers running from the meniscus to the tibia were sectioned, leaving only the supra-meniscal fibers intact and attached to the lateral meniscus. The infra-meniscal specimens (n = 7) were prepared by removing the femur and subsequently all ALL fibers attached to it. The lateral corner of the tibia was sectioned leaving the tibial plateau intact (Fig. 1b) and ensuring that no damage occurred to any part of the meniscus and the infra-meniscal ALL (Fig. 1b).
The bones (tibial plateau and femur for the supra-meniscal specimens and the tibia and tibial plateau for the infra-meniscal specimens) were potted into sections of PVC via dental cement (Denstone dental cement; Hereaus Holdings GmbH, Hanau, Germany) such that the remaining portion of ALL was pulling laterally on the meniscus (Fig. 2). Sections of wire were threaded through the bone and held in place with bone cement prior to potting to increase adherence within the PVC sections.
The specimens were then rigidly secured within an Instron® materials testing machine (Instron® 8874; Norwood MA) (Fig. 2) where they were statically preloaded at 20 N for 2 min. Cyclical pre-loading was then performed between 10 and 30 N for 10 cycles at 1 Hz, immediately followed by a tensile test at 1 mm/s. The peak force (i.e., the force just prior to ultimate failure) and stiffness were extracted from the force–time curves. Stiffness was calculated as the slope of the linear portion of the force–deformation curve between 30 and 70% of the peak force [22, 23]. Peak stress was also calculated by normalizing the peak force to the cross-sectional area of each ALL specimen.
One intact cadaveric knee specimen (81 years) was used for histological analysis. Following anatomical dissection, the ALL was resected from its bony attachments and was trimmed such that a small section of lateral meniscus remained attached. The specimen was fixed in 10% formaldehyde, and embedded longitudinally in paraffin wax. Using a microtome (microm HM-325; GMI Inc, Ramsey, MN), the specimen was sectioned at a thickness of 5 μm, placed in a warm water bath, transferred onto glass slides and placed in a 40 °F oven to fix the slides. The slides were then stained with Masson’s Trichrome using a standard protocol to allow for better visualization of collagen arrangement. Qualitative analysis of the tissue was carried out using a Zeiss AxioScope.A1 microscope (Zeiss, Toronto, ON) and the images were captured using AxioVision Microscopy Software (LE Rel 4.8; Zeiss, Toronto, ON). The lateral inferior geniculate artery (LIGA) was used as a reference point to landmark where the supra- and infra-meniscal fibers were located, as it travels between the lateral meniscus and the ALL.
Statistical analysis
The repeatability of the thickness and width measurements were assessed using an intra-class correlation analysis (ICC) with the following criteria: ICC < 0.4 = poor; 0.4 < ICC < 0.59 = fair; 0.60 < ICC < 0.74 = good; ICC > 0.74 = excellent [24]. The structural properties of each ALL-meniscus construct were categorized with the load to failure and stiffness. An independent sample, one-tailed t test was used to determine if there were statistically significant differences in peak load and stiffness between the supra- and infra-meniscal fibers. One of the specimens in the infra-meniscal group had porous bone that prevented secure potting for testing, and therefore, was not included in analysis, resulting in a sample size of six for the infra-meniscal group. A post-hoc calculation of effect size (Cohens d) was performed with G*power statistical software (v3.1.9.2; Heinrich-Heine-Universitat, Dusseldorf, Germany) [25], and were interpreted as small (0.2), medium (0.5), or large (0.8), as described by Cohen’s d criteria [26]. All statistical analyses were performed with SPSS statistical software (v21; IBM Corp, Armonk, NY) and alpha was set at 0.05.
Results
Overall, the width and thickness measurements were found to have good repeatability with ICCs of 0.67 and 0.60, respectively. There was a significant difference in the peak force between the supra- and infra-meniscal attachments such that the infra-meniscal fibers resulted in greater forces compared to the supra-meniscal fibers (p = 0.01; effect size = 1.38) (Fig. 3). The infra-meniscal fibers were also significantly stiffer than the supra-meniscal fiber (p = 0.01; effect size = 1.36) (Fig. 3). Furthermore, the mean (SD) cross-sectional area of both the supra- (22.23 [8.94] mm2) and infra-meniscal (20.45 [9.95] mm2) fibers were statistically the same (ns; effect size = 0.18) (Table 1), thus the peak stress in the infra-meniscal fibers was 45% greater than the supra-meniscal fibers, a difference that was statistically different (p = 0.02; effect size = 1.18) (Fig. 4).
For the seven supra-meniscal specimens, failure occurred near the femoral insertion, mid substance, or the meniscal insertion (Fig. 5). For the seven infra-meniscal specimens, failure of the ALL-meniscal construct occurred either mid-substance or at the tibial attachment which presented with a similar pattern seen following a Segond fracture (Fig. 6).
The LIGA was identified in the histological section and appears bright pink when stained with Masson’s Trichrome (Fig. 7). Throughout the ALL, the collagen was arranged in a dense regular organization that is consistent with the arrangement of ligamentous tissue. In the longitudinal section, the fibers of the ALL revealed collagen running parallel to each other and they exhibited a crimped pattern that is characteristic of dense regular connective tissue (Fig. 8). The pattern of collagenous arrangement was relatively consistent between the supra-meniscal (Fig. 8a) and the infra-meniscal fibers (Fig. 8b) of the ALL.
Discussion
The primary finding of this study was that the ALL-meniscal construct that included the infra-meniscal fibers was significantly stronger and stiffer when compared to the supra-meniscal fibers. This occurred despite limited differences in the histological findings indicating that the two ALL sections have the same morphological characteristics.
In a study by Zens et al. [18] that used a standard load-to-failure protocol, the ultimate load that the ALL could withstand was 49 N with a failure point at approximately 1/3 the length of the ALL (closer to the femoral insertion). Conversely, Kennedy et al. reported a peak load of 175 N and an average stiffness of 20 N/mm (range of 9–39 N/mm) [4]. Although Kennedy et al. did not analyze the separate sections of the ALL, the results in the current investigation agree well with their reported values. The large differences observed when compared to Zens et al. [18] may have occurred as a result of identifying tissue that was considered ALL, as the cross-sectional areas recorded in the current investigation are an order of magnitude larger that those presented by Zens et al. [4, 18].
The histological analysis of the ALL was performed to determine if the biomechanical differences observed between the supra- and infra-meniscal fibers of the ALL could be attributed to a difference in collagen organization. However, the results of the analysis indicated similarities in the collagen organization between the supra- and infra- meniscal fibers, therefore the microscopic composition of the ALL likely does not account for the biomechanical differences observed here. Furthermore, both the supra- and infra-meniscal fibers presented with the same cross-sectional area, and therefore the stress in the infra-meniscal fibers was also greater than that experienced by the supra- meniscal fibers.
It is possible that the differences in peak force between the two ALL sections occurred because the ALL-meniscal constructs were tested, as opposed to isolated sections of ALL. There may be differences between the ALL-meniscal interface and the connection that exists between the two sections of fibers, thus affecting their mechanical behavior. It was shown that a relatively strong connection exists between the ALL and meniscus, as evidenced by only a single specimen failing at the meniscal insertion. This demonstrates a strong mechanical relationship between the anterolateral tibial plateau and the lateral meniscus, and could in part explain the association of lateral meniscus tears with ACL injury. Injury to the lateral meniscus is found in approximately 44% of ACL reconstructions [27, 28], with tears to the posterior root accounting for upwards of 23% of these tears [27]. It also not uncommon (14% incidence) to find a radial tear of the middle third of the lateral meniscus in the ACL deficient knee during arthroscopy [29]. The strong attachment of the ALL at the meniscal body may apply significant shear forces through the meniscus during anterior subluxation of the knee, with the posterior root being held posteriorly by both the root insertion and the buttress effect of the lateral femoral condyle. Van Dyck et al. observed that 71% of ALL lesions were found in the infra-meniscal portion [7] and they noted that of those patients with an ALL injury, 61% had lateral meniscus tears compared to 31% with no ALL abnormality. This suggests that injury to the infra-meniscal ALL may result in a more mobile lateral meniscus that could place greater tension on the posterior root during anterolateral subluxation episodes, causing an increased risk of lateral meniscus tear.
The differences in the infra-meniscal and supra-meniscal tensile properties may be due to the supra-meniscal fibers not being subjected to the same load magnitude as the infra-meniscal sections during normal loading scenarios. To our knowledge, sectioning protocols utilized in the majority of biomechanical studies investigating the ALL have incorporated infra-meniscal sectioning of the ALL. We therefore hypothesize that it is the meniscotibial ALL structure that exerts the majority of anterolateral control, not the supra-meniscal attachment or the ALL structure as a whole. These observations are further supported by the idea that during a rupturing event of the ACL, the knee collapses into a combination of valgus and flexion, causing the lateral meniscus to move posteriorly off the tibial plateau as it subluxes anterolaterally, causing the infra-meniscal fibers to be placed under tension while the supra-meniscal fibers remain relatively unloaded.
More recent studies have identified the iliotibial band, in particular the deep layer of the ITB and its Kaplan fiber attachment, to provide the greatest control of anterolateral rotation [30]. An additional study by Rahnemai-Azar et al., quantified mean peak forces of the anterolateral capsule (319 N) and the ITB (487 N) demonstrating a clear difference in the peak forces in these structures compared to the ALL found in this present study [31]. The results of the study presented here, and previous research, suggest that the combination of the ITB with its Kaplan fiber attachment, the meniscotibial (i.e., infra-meniscal) portion of the ALL and the lateral meniscus, can be thought of in terms of an anterolateral capsulomeniscal complex—the anterolateral corner [30]. As such, reconstruction or repair of one or a combination of these structures alongside ACL reconstruction may help restore anterolateral rotational laxity and warrants further investigation.
As is common with biomechanical studies of this nature, the cadaveric specimens used in the current investigation were from older individuals, and thus the limitations associated with them would be present. Furthermore, although this study used a relatively small sample size the large effect sizes suggest that the findings are still meaningful.
Conclusion
The results of this study indicate that the ALL-meniscal construct that includes the infra-meniscal fibers is significantly stronger and stiffer than the construct that includes the supra-meniscal fibers. Therefore, the infra-meniscal ALL is another important component of the anterolateral complex of the knee and should be considered when patients present with an ACL and/or lateral meniscal injury as it may have clinical implications with respect to anterolateral rotational laxity.
References
Caterine S, Litchfield R, Johnson M, Chronik B, Getgood A (2014) A cadaveric study of the anterolateral ligament: re-introducing the lateral capsular ligament. Knee Surg Sport Traumatol Arthrosc 23:3186–3195
Claes S, Vereecke E, Maes M, Victor J, Verdonk P, Bellemans J (2013) Anatomy of the anterolateral ligament of the knee. J Anat 223:321–328
Helito CP, Bonadio MB, Soares TQ, Pecora JR, Natalino R, Camanho GL, Demange MK (2016) The meniscal insertion of the knee anterolateral ligament. Surg Radiol Anat 38:223–228
Kennedy MI, Claes S, Fuso FAF, Williams BT, Goldsmith MT, Turnbull TL, Wijdicks CA, LaPrade RF (2015) The anterolateral ligament: an anatomic, radiographic, and biomechanical analysis. Am J Sports Med 43:1–10
Kosy JD, Soni A, Venkatesh R, Mandalia VI (2016) The anterolateral ligament of the knee: unwrapping the enigma. Anatomical study and comparison to previous reports. J Orthop Traumatol 17:303–308
Pomajzl R, Maerz T, Shams C, Guettler J, Bicos J (2015) A review of the anterolateral ligament of the knee: current knowledge regarding its incidence, anatomy, biomechanics, and surgical dissection. Arthroscopy 31:583–591
Van Dyck P, Clockaerts S, Vanhoenacker FM, Lambrecht V, Wouters K, de Smet E, Gielen JL, Parizel PM (2016) Anterolateral ligament abnormalities in patients with acute anterior cruciate ligament rupture are associated with lateral meniscal and osseeous injuries. Eur Radiol 26:1–9
Hughston JC, Andrews JR, Cross MJ, Moschi A (1976) Classification of knee ligament instabilities part II: the lateral compartment. J Bone Jt Surg 58:173–179
Helito CPC, Demange MMK, Bonadio MB, Tírico LEP, Gobbi RG, Pécora JR, Camanho GL (2013) Anatomy and histology of the knee anterolateral ligament. Orthop J Sport Med. doi:10.1177/2325967113513546
Shybut TB, Vega CE, Haddad J, Alexander JW, Gold JE, Noble PC, Lowe WR (2015) Effect of lateral meniscal root tear on the stability of the anterior cruciate ligament-deficient knee. Am J Sports Med 43:905–911
Vincent J-P, Magnussen RA, Gezmez F, Uguen A, Jacobi M, Weppe F, Al-Saati MF, Lustig S, Demey G, Servien E, Neyret P (2012) The anterolateral ligament of the human knee: an anatomic and histologic study. Knee Surg Sport Traumatol Arthrosc 20:147–152
Sonnery-Cottet B, Lutz C, Daggett M, Dalmay F, Freychet B, Niglis L, Imbert P (2016) The involvement of the anterolateral ligament in rotational control of the knee. Am J Sports Med 44:1209–1214
Monaco E, Ferretti A, Labianca L, Maestri B, Speranza A, Kelly MJ, D’Arrigo C (2012) Navigated knee kinematics after cutting of the ACL and its secondary restraint. Knee Surg Sport Traumatol Arthrosc 20:870–877
Parsons EM, Gee AO, Spiekerman C, Cavanagh PR, Parsons EM, Gee AO, Spiekerman C (2015) The biomechanical function of the anterolateral ligament of the knee. Am J Sports Med 43:669–674
Rasmussen MT, Nitri M, Williams BT, Moulton SG, Cruz RS, Dornan GJ, Goldsmith MT, LaPrade RF (2016) An in vitro robotic assessment of the anterolateral ligament, part 1: secondary role of the anterolateral ligament in the setting of an anterior cruciate ligament injury. Am J Sports Med 44:585–592
Spencer L, Burkhart TA, Tran MN, Rezansoff AJ, Deo S, Caterine S, Getgood AM (2015) Biomechanical analysis of simulated clinical testing and reconstruction of the anterolateral ligament of the knee. Am J Sports Med 43:2189–2197
Tavlo M, Eljaja S, Jensen JT, Siersma VD, Krogsgaard MR (2016) The role of the anterolateral ligament in ACL insufficient and reconstructed knees on rotatory stability: a biomechanical study on human cadavers. Scand J Med Sci Sports 26:960–966
Zens M, Feucht MJ, Ruhhammer J, Bernstein A, Mayr HO, Norbert PS, Woias P, Niemeyer P (2015) Mechanical tensile properties of the anterolateral ligament. J Exp Orthop 2:7
Guenther D, Griffith C, Lesniak B, Lopomo N, Grassi A, Zaffagnini S, Fu FH, Musahl V (2015) Anterolateral rotatory instability of the knee. Knee Surg Sport Traumatol Arthrosc 23:2909–2917
Macchi V, Porzionato A, Morra A, Stecco C, Tortorella C, Menegolo M, Grignon B, De Caro R (2016) The anterolateral ligament of the knee: a radiologic and histotopographic study. Surg Radiol Anat 38:341–348
Van der Watt L, Khan M, Rothrauff BB, Ayeni OR, Musahl V, Getgood A, Peterson D (2015) The structure and function of the anterolateral ligament of the knee: a systematic review. Arthroscopy 31:569–582
Burkhart TA, Andrews DM (2010) Activation level of extensor carpi ulnaris affects wrist and elbow acceleration responses following simulated forward falls. J Electromyogr Kinesiol 20:1203–1210
Duquette A, Andrews DM (2010) Comparing methods of quantifying tibial acceleration slope comparing methods of quantifying tibial acceleration slope. J Appl Biomech 26:229–233
Shrout PE, Fleiss JL (1979) Intraclass correlations: uses in assessing rater reliability. Psychol Bull 86:420–428
Faul F, Erdfelder E, Lang A-G, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191
Maher JM, Markey JC, Ebert-May D (2013) The other half of the story: effect size analysis in quantitative research. Cell Biol Educ 12:345–351
Feucht MJ, Bigdon S, Bode G, Salzmann GM, Dovi-Akue D, Südkamp NP, Niemeyer P (2015) Associated tears of the lateral meniscus in anterior cruciate ligament injuries: risk factors for different tear patterns. J Orthop Surg Res 10:1–8
Kilcoyne KG, Dickens JF, Haniuk E, Cameron KL, Owens BD (2012) Epidemiology of meniscal injury associated with ACL tears in young athletes. Orthopedics 35:208–212
Magee T, Shapiro M, Williams D (2002) MR accuracy and arthroscopic incidence of meniscal radial tears. Skeletal Radiol 31:686–689
Kittl C, El-daou H, Athwal KK, Gupte CM, Weiler A, Williams A, Amis AA (2016) The role of the anterolateral structures and the ACL in controlling laxity. Am J Sports Med 44:345–354
Rahnemai-Azar AA, Miller RM, Guenther D, Fu FH, Lesniak BP, Musahl V, Debski RE (2016) Structural properties of the anterolateral capsule and iliotibial band of the knee. Am J Sports Med 44:892–897
Acknowledgements
The authors wish to acknowledge the research funding support from an unrestricted internal research grant from Lawson Health Research and Western University Department of Surgery. We would also like to acknowledge funding from the Canadian Institute of Health Research. Funding was also provided by the Schulich School of Medicine and Dentistry. We would like to thank Smith and Nephew Inc for providing a research grant providing salary support for Dr Burkhart.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest in respect to the content of the work.
Rights and permissions
About this article
Cite this article
Corbo, G., Norris, M., Getgood, A. et al. The infra-meniscal fibers of the anterolateral ligament are stronger and stiffer than the supra-meniscal fibers despite similar histological characteristics. Knee Surg Sports Traumatol Arthrosc 25, 1078–1085 (2017). https://doi.org/10.1007/s00167-017-4424-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-017-4424-y