Abstract
Purpose
The purpose of this study was to analyse the accuracy of component placement during unicompartmental knee arthroplasty (UKA) using a robotic-assisted system.
Methods
Two hundred and six patients (232 knees) who underwent medial robotic-assisted UKA were retrospectively studied. Femoral and tibial sagittal and coronal alignments were measured in the post-operative radiographs and were compared with the equivalent measurements collected during the intra-operative period by the robotic system. Mismatch between pre-planning and post-operative radiography was assessed against accuracy of the prosthesis insertion.
Results
Robotic-assisted surgery for medial UKA resulted in an average difference of 2.2° ± 1.7° to 3.6° ± 3.3° depending on the component and radiographic view between the intra-operatively planned and post-operative measurements. Mismatch between pre-planning and post-operative radiography (inaccuracy) was related to improper cementing technique of the prosthesis in all measurements (except for tibial sagittal axis) rather than wrong bony cuts performed by the robotic arm.
Conclusion
Robotic-assisted medial UKA results in accurate prosthesis position. Inaccuracy may be attributed to suboptimal cementing technique.
Level of evidence
Comparative retrospective study, Level III.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Unicompartmental knee arthroplasty (UKA) has recently found re-emergence for the treatment of knee arthritis, especially effecting the medial compartment [4, 24]. However, the minimal invasive approach to UKA is technically challenging with reduced visibility of bony landmarks used for sizing and alignment [3, 12, 13, 17, 19, 27]. Survival of UKA is mainly dependent on component position; malalignment of the prosthesis may result in poor post-operative function and progressive wear leading to early revision surgery [1, 4, 7, 9, 14, 15, 35].
Navigated and robotic-assisted UKA has been shown to improve component alignment and post-operative ligament balancing [2, 5, 10, 16, 18–23, 30–33]. Dunbar et al. measured the accuracy of component placement with the use of a robotic-assisted system on post-operative CT scans in 20 UKAs [11]. Components were placed within 1.6 mm and 3° of the pre-operative plan. However, component placement was compared to pre-operative CT measurements by the robotic systems, a comparison between the actual intra-operative plan and post-operative alignment has not been performed [5, 10, 17–21, 24].
The purpose of this study was to assess the accuracy of component placement using a robotic-assisted system for medial UKA by comparing intra-operative robotic alignment with post-operative alignment and to assess the quality of the component insertion. The findings of this study will help identify surgical factors that may lead to a deviation of planned component position with an otherwise accurate robotic-assisted system. The hypothesis of the study was that the robotic-assisted system produces accurate bone cuts, but post-operative component position is influenced by the quality of the cementing and component insertion technique.
Materials and methods
Two hundred and thirty two knees (206 patients) that had undergone medial unicompartmental knee replacement by the two senior authors, RHJ and GGP using robotic-assisted bone preparation with a tactile guidance system (MAKO Surgical Corp., Ft Lauderdale, FL, USA) were included in the study. This system consists of patient-specific pre-operative planning using a three-dimensional image which is acquired using pre-operative CT scan. The intra-operative procedure consists of soft dynamic tissue balancing which is achieved by recording the flexion and extension gaps through the full range of motion in 30° increments (in our series) with the knee in a corrected alignment [31]. This allows to reposition the prosthesis to minimize the tightness or looseness of the prosthesis rather than soft tissue release. Once proper alignment is achieved, haptic guided burring to remove the predefined chondral surface and underlying bone is performed. This is followed by trialling the chosen prosthesis and repeat assessment of soft tissue balance (Fig. 1) [22, 23, 30–32]. Accuracy of the haptically guided robotic system is within 1 mm and soft tissues can be balanced to <1 mm of imbalance throughout range of motion [31]. In all cases, a cemented, fixed-bearing tibial inlay component was inserted via a medial paramedian minimally invasive approach. Patients were mobilized on the first post-operative day and discharged within 24–48 h. Every patient underwent post-operative radiographs in the recovery room, followed by routine post-operative radiograph at 6 weeks.
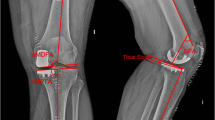
We performed a chart and radiographic review and accessed the robotic system to collect and compare the intra-operative planning alignment data saved on each patient and to compare it with the immediate post-operative radiological alignment data. The parameters which were collected and analysed were sagittal and coronal alignments of femoral and tibial prostheses as compared to the mechanical axis of the femur and tibia, respectively (Figs. 1, 2). In order to obtain a comparative measurement between intra-operative coronal angulation of the femoral prosthesis, the difference between the anatomical and the mechanical axis of the femur was identified. This measurement was taken using the pre-operative CT scan performed for pre-operative planning (Fig. 3). The contact point between femur and tibia was also measured by measuring the centre of the femoral and tibial prosthesis on a coronal plane to identify if these matched (Fig. 4). All pre-operative planning CT scans and post-operative radiographs were taken in a standardized format in the same radiology department measured and calibrated in Philips iSite PACS system (Philips Healthcare, Andover, MA, USA). All alignment measurements were taken by two researchers twice. The aim was to assess the intra- and inter-observer variability of the radiographic measurements. Average values of the measurements taken by the two researchers were used to derive the radiographic measurement of alignment.
Assessment of insertion technique (quality of cementation)
The quality of prosthesis insertion and cementation was assessed on post-operative radiographs by analysing the seating of the prosthesis. Incorrect seating was identified by a halo around the femoral prosthesis caused by the femoral prosthesis not seated in the burred femoral bed. A cement wedge identified on the tibial side signified that the tibial prosthesis had not been seated in its bed correctly, and the excess room was taken by cement (Fig. 5).
Malinsertion of tibial and femoral prosthesis. In case of a metallic femoral prosthesis, this is seen as a halo around the metallic prosthesis as the inlay prosthesis is not seated in the burred region (large arrows). The malinsertion of the tibial prosthesis is seen as cement wedge around an all polyethylene radiolucent prosthesis as the cement shifts the prosthesis from the correct insertion position (thin arrows)
Assessments
-
1.
Accuracy of robotic surgery: Parameters from the intra-operative planned cuts were compared to the post-operative radiological parameters. The difference between intra-operatively measured (planned) alignments and the post-operative measured alignments was calculated and presented in graphic format (Fig. 6). When assessing the accuracy of robotic surgery to avoid skewing results as a result of averaging positive and negative errors, the root mean square, or the quadratic mean, was used to quantify alignment errors.
-
2.
Quality of cementation: Total number of suboptimally cemented femoral and tibial prostheses was presented as the percentage of total unicompartmental knee replacement in this series (Fig. 5).
-
3.
Effect of poor cementation on the accuracy of robotic surgery: The quality of cementation of femoral and tibial prostheses was analysed against the accuracy of the robotic surgery.
Institutional review board approval was obtained from Wake Forest Baptist Health prior to commencement of this study.
Statistical analysis
Statistical data was collated in the Microsoft Excel 2007 (TM) spread sheet. The statistical assessment was performed using SPSS (TM) version 11.
The radiological measurements of the femoral and tibial prosthesis alignment were deemed to be exchangeable, and data could not be stratified; hence, kappa correlation coefficient was not possible. Because of this, inter-observer and intra-observer variation was analysed using Lin’s inter-class correlation coefficient.
Effect of poor cementation on the accuracy of robotic surgery was analysed using Mann–Whitney U test as the data was presumed to be non-parametric.
Results
The average age was 64 ± 11 years with a body mass index of 33.5 ± 10 kg/m2. Male to female ratio was 5.5:4.5. Average range of motion achieved at 6 months was 0.38° of extension lag or fixed flexion deformity (range 0°–15°) and 120° of active flexion (range 90°–140°) post-operatively. Only one patient was revised early for prosthesis malposition. This patient had range of motion of 15°–75° and no improvement in symptoms post UKA.
The average accuracy (the difference between intra-operatively measured [planned] alignment and the post-operatively measured alignment) of femoral prosthesis in the coronal plane was 2.8° ± 2.5° (range 0–26) and 3.6° ± 3.3° (range 0–17) in the sagittal plane. The average accuracy (the difference between intra-operatively measured (planned) alignment and the post-operatively measured alignment) of the tibial prosthesis in the sagittal plane was 2.4° ± 2° (range 0–8) and 2.2° ± 1.75° (range 0–11) in the coronal plane. Depending on the plane, between 62 and 75 % of femoral prosthesis had <3° of inaccuracy and between 83 and 91 % of the femoral prostheses had <5° of inaccuracy. Again, depending on the plane, 82–84 % of the tibial prostheses had <3 % inaccuracy and between 93 and 96 % of the tibial prostheses had <5 % inaccuracy. The accuracy for each plane and each component of the prosthesis is illustrated in Fig. 6.
Four femoral prostheses showed signs of malinsertion; however, tibial prosthesis malinsertion was much more common with 46 tibial prostheses malinserted. Correct tibiofemoral contact point was seen in 130 knees. However, one hundred and two cases had a mismatch in the contact point more than 1 mm. In 86 cases, the femoral prosthesis was medial to the tibial prosthesis, and in the other 16 cases, the tibial prosthesis was more medial to the femoral prosthesis (Fig. 5).
In the presence of femoral malinsertion, inaccuracy of femoral and tibial prosthesis increased significantly (Table 1). The average femoral sagittal inaccuracy increased from 2.4° ± 2.6° to 5° ± 1.4° (p < 0.05) in the malinserted prostheses when compared to the well-inserted prostheses. The average femoral coronal inaccuracy increased from 3° ± 2.7° to 19° ± 10° (p < 0.00005) in the malinserted prostheses when compared to the well-inserted prostheses. The average tibial coronal inaccuracy increased from 1.9° ± 1.5° to 3.6° ± 2.5° (p < 0.005) in the malinserted prostheses when compared to the well-inserted prostheses (Table 1). The difference between inaccuracy of malinserted prostheses and well-inserted prostheses in tibial sagittal plane was not significant with an average inaccuracy of 2.1° ± 2.1° as opposed to 1.7° ± 1.6° (N.S.) (Table 1).
Data variability
The data was consistent with minimal variability. The intra-observer variation was measured to be in average 0.8 ± 1 mm, and the average inter-observer variation was measured to be 1.56 ± 1.35 mm. The concordance correlation coefficient of the intra-observer variation all measurements was 0.9933. The concordance correlation coefficient of the inter-observer variation for all measurements was 0.9963.
Discussion
The most important finding of this study is the high degree of agreement between intra-operatively planned prosthesis alignment and post-operatively measured alignment. The findings of this study concur with previous assessments of the accuracy of prosthesis alignment using a robotic-assisted system for medial UKA [7, 8, 11, 23]. In this study, we defined accuracy as agreement between planned intra-operative alignment and post-operatively radiologically measured alignment as defined by Cobb et al. [8] and Lonner et al. [23]. We found accurate prosthesis insertion with over 70 % prosthesis inserted within 3° of planned alignment for femoral and tibial prosthesis in all coronal and sagittal planes (Fig. 6). While we have no ability to compare our data to a control group who were operated with conventional technique due to the retrospective nature of the study and lack of similarly sized historical control, a recent cadaveric study by Citak et al. [7] has shown significant improvements compared to conventional technique.
We identified a cohort of UKA in this study that had inaccuracy of more than 5° in sagittal or coronal planes (between 5 and 10 % depending on the component and plane). Further analysis revealed a correlation between the accuracy of component position and the quality of the cementing technique based on post-operative radiographs. Suboptimal cementing as defined by a halo around the femoral prosthesis due to malinsertion of femoral inlay prosthesis and cement wedge around the tibial all poly inlay prosthesis in knees in our series was associated with higher mismatch (Fig. 5). It remains unclear from our analysis if the cementing technique caused malalignment; however, as robotic-assisted surgery with low tolerance for error is used, the part of robotic-assisted UKA with highest risk of inaccuracy may be the insertion and cementation step. Malinsertion of tibial inlay prosthesis has not been reported in non-navigated and conventionally used inlay UKA but was independently described [25, 28, 34]. Sinha and Cutler revealed a relationship between malinsertion of inlay tibial component and component position in a similar study [34]. This fact is relevant to all surgeons who perform medial UKA, especially inlay prosthesis via minimally invasive approach. It highlights misalignment potentially caused by cementation and not by poor bone cuts and care must be taken to make sure each prosthesis is seated perfectly and pressurized during cementing.
Mismatch between surgically planned and radiologically measured alignment can also partly be explained by the inaccuracy in the radiological measurements [8, 23]. This is evident by the fact that in the parameters which the mismatch between measured the intra-operative (CT based) and post-operative (radiograph based) measurements is higher the inter- and intra-observer mismatch also increases.
Furthermore radiological measurement of the femoral insertion is hard to measure on plane radiographs and with imageless navigation [6, 10, 24, 26]. The difficulty and the resultant error can be attributed to the bow in the femur in the sagittal and coronal plane and has been measured to be on average 1.7° (range 0°–3.8°) [6, 10, 26]. Post-operative femoral prosthesis position and the degree of the femoral bow are accurately measured with CT scan; however, this was not available in our series [24]. This explains the higher mismatch in the femur in spite of more predictable insertion (Fig. 5). Assessment of soft tissue balance was not within the remit of this study; however, in a recent prospective study, we have measured soft tissue balance after robotic-assisted UKA to be within 1 mm in vast majority of cases throughout the range of motion [31].
The mismatch in the contact point in the post-operative radiograph could not be assessed as the intra-operative data was not available for the contact point. Hence, we do not know if the mismatch in the femorotibial contact point was pre-planned or caused by surgical inaccuracy. However, in this study, even when robotic-assisted surgery is performed, 40 % UKAs show signs of femorotibial mismatch of over 1 mm (Fig. 4). The femoral prosthesis is often translated medially. This post-operative translation may be associated with pre-operative tibiofemoral translation. Medial translation of the tibia in our series was much rarer and is caused by valgus insertion of the femoral prosthesis. The effect of mediolateral mismatch is diminished due to the flat on curved nature of this particular design of fixed-bearing prosthesis [4]. We feel more research is required to study this phenomenon.
This study is not without limitations. This study is a retrospective analysis of one robotic-assisted system at a single institution. Findings from this study in regards to proper cementing technique may need to be implemented in clinical practice, and the accuracy of component placement in robotic-assisted UKA re-evaluated prospectively. The aim of this study was to assess the accuracy of alignment of prosthesis not the actual long leg alignment of the leg or alignment of prosthesis. As such, the functional success or long-term survival cannot be extrapolated from accurate insertion [14, 15, 23, 29]. As mentioned previously, more accurate methods of assessment such as CT scanning exists and may need to be used to assess the prosthesis post-operative position of the prosthesis, especially the femoral component which as we mentioned earlier is harder to measure due to the femoral bow [8, 24, 26]. Finally, tibiofemoral mismatch has not been previously studied and is of questionable importance due to its small size. Increased stress of the load bearing surface may not be observed with the flat on curved prosthesis design even if significant tibiofemoral mismatch is observed. This phenomenon requires further study.
Conclusions
Minimal difference between intra-operatively measured (planned) alignments and the post-operatively measured alignments of the tibia and femur in the sagittal and the coronal planes was seen with robotic-assisted UKA in the current study. Mismatch in image-based alignment and post-operatively measured alignment may be attributed to suboptimal cementing technique as opposed to inaccuracy of robotic-assisted bone cuts. Proper cementation technique appears to be vital to achieve accurate post-operative alignment with a robotic-assisted UKA system.
References
Assor M, Aubaniac JM (2006) Influence of rotatory malposition of femoral implant in failure of unicompartimental medial knee prosthesis. Rev Chir Orthop Reparatrice Appar Mot 92(5):473–484
Banks SA (2009) Haptic robotics enable a systems approach to design of a minimally invasive modular knee arthroplasty. Am J Orthop (Belle Mead NJ) 38(2 Suppl):23–27
Berend KR, Lombardi AV Jr, Mallory TH, Adams JB, Groseth KL (2005) Early failure of minimally invasive unicompartmental knee arthroplasty is associated with obesity. Clin Orthop Relat Res 440:60–66
Borus T, Thornhill T (2008) Unicompartmental knee arthroplasty. J Am Acad Orthop Surg 16(1):9–18
Buckup K, Lars-Christoph Linke LC, Hahne V (2007) Minimally invasive implantation and computer navigation for a unicondylar knee system. Orthopedics 30(8 Suppl):66–69
Chung BJ, Kang YG, Chang CB, Kim SJ (2009) Differences between sagittal and femoral mechanical and distal reference axes should be considered in navigated TKA. Clin Orthop Relat Res 467:2403–2413
Citak M, Suero EM, Citak M, Dunbar NJ, Branch SH, Conditt MA, Banks SA, Pearle AD (2012) Unicompartmental knee arthroplasty: is robotic technology more accurate than conventional technique? Knee 20(4):268–271
Cobb J, Henckel J, Gomes P, Harris S, Jakopec M, Rodriguez F, Barrett A, Davies B (2006) Hands-on robotic unicompartmental knee replacement: a prospective, randomised controlled study of the acrobot system. J Bone Joint Surg Br 88(2):188–197
Dalury DF, Dennis DA (2005) Mini-incision total knee arthroplasty can increase risk of component malalignment. Clin Orthop Relat Res 440:77–81
Davies BL, Rodriguez y Baena FM, Barrett AR, Gomes MP, Harris SJ, Jakopec M, Cobb JP (2007) Robotic control in knee joint replacement surgery. Proc Inst Mech Eng H 221(1):71–80
Dunbar NJ, Roche MW, Park BH, Branch SH, Conditt MA, Banks SA (2012) Accuracy of dynamic tactile-guided unicompartmental knee arthroplasty. J Arthroplasty 27(5):803–808
Fisher DA, Watts M, Davis KE (2003) Implant position in knee surgery: a comparison of minimally invasive, open unicompartmental, and total knee arthroplasty. J Arthroplasty 18(Suppl):2–8
Hamilton WG, Collier MB, Tarabee E, McAuley JP, Engh CA Jr, Engh GA (2006) Incidence and reasons for reoperation after minimally invasive unicompartmental knee arthroplasty. J Arthroplasty 21(Suppl):98–107
Hernigou P, Deschamps G (2004) Alignment influences wear in the knee after medial unicompartmental arthroplasty. Clin Orthop Relat Res 423:161–165
Hernigou P, Deschamps G (2004) Posterior slope of the tibial implant and the outcome of unicompartmental knee arthroplasty. J Bone Joint Surg Am 86:506–511
Jakopec M, Harris SJ, Rodriguez y Baena F, Gomes P, Cobb J, Davies BL (2001) The first clinical application of a “hands-on” robotic knee surgery system. Comput Aided Surg 6(6):329–339
Jenny JY, Müller PE, Weyer R, John M, Weber P, Ciobanu E, Schmitz A, Bacher T, Neumann W, Jansson V (2006) Navigated minimally invasive unicompartmental knee arthroplasty. Orthopedics 29(10 Suppl):S117–S121
Jenny JY, Ciobanu E, Boeri C (2007) The rationale for navigated minimally invasive unicompartmental knee replacement. Clin Orthop Relat Res 463:58–62
Jenny JY (2008) Unicompartmental knee replacement: a comparison of four techniques combining less invasive approach and navigation. Orthopedics 31(10 Suppl 1):2
Jenny JY (2008) Navigated unicompartmental knee replacement. Sports Med Arthrosc 16(2):103–107
Keene G, Simpson D, Kalairajah Y (2006) Limb alignment in computer-assisted minimally-invasive unicompartmental knee replacement. J Bone Joint Surg Br 88:44–48
Lang JE, Mannava S, Floyd AJ, Goddard MS, Smith BP, Mofidi A, Seyler TM, Jinnah RH (2011) Robotic systems in orthopaedic surgery. J Bone Joint Surg Br 93:1296–1299
Lonner JH (2009) Indications for unicompartmental knee arthroplasty and rationale for robotic arm-assisted technology. Am J Orthop (Belle Mead NJ) 38(2 Suppl):3–6
Lonner JH, John TK, Conditt MA (2010) Robotic arm-assisted UKA improves tibial component alignment a pilot study. Clin Orthop Relat Res 468:141–146
Lustig S, Paillot JL, Servien E, Henry J, Ait Si Selmi T, Neyret P (2009) Cemented all polyethylene tibial insert unicompartmental knee arthroplasty: a long term follow-up study. Orthop Traumatol Surg Res 95(1):12–21
Minoda Y, Kobayashi A, Iwaki H, Mitsuhiko I, Kadoya Y, Ohashi H, Takaoka K, Nakamura H (2010) The risk of notching the anterior femoral cortex with the use of navigation systems in total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc 18(6):718–722
Müller PE, Pellengahr C, Witt M, Kircher J, Refior HJ, Jansson V (2004) Influence of minimally invasive surgery on implant positioning and the functional outcome for medial unicompartmental knee arthroplasty. J Arthroplasty 19(3):296–301
O’Donnell T, Neil MJ (2010) The Repicci II® unicondylar knee arthroplasty: 9-year survivorship and function. Clin Orthop Relat Res 468(11):3094–3102
Pandit H, Jenkins C, Gill HS, Barker K, Dodd CA, Murray DW (2011) Minimally invasive Oxford phase 3 unicompartmental knee replacement: results of 1000 cases. J Bone Joint Surg Br 93(2):198–204
Pearle AD, O’Loughlin PF, Kendoff DO (2010) Robot-assisted unicompartmental knee arthroplasty. J Arthroplasty 25(2):230–237
Plate JF, Mofidi A, Mannava S, Smith BP, Lang JE, Poehling GG, Conditt MA, Jinnah RH (2013) Achieving accurate ligament balancing using robotic-assisted unicompartmental knee arthroplasty. Adv Orthop: 837167
Roche M, O’Loughlin PF, Kendoff D, Musahl V, Pearle AD (2009) Robotic arm-assisted unicompartmental knee arthroplasty: preoperative planning and surgical technique. Am J Orthop (Belle Mead NJ) 38(2 Suppl):10–15
Sinha RK (2009) Outcomes of robotic arm-assisted unicompartmental knee arthroplasty. Am J Orthop (Belle Mead NJ) 38(2 Suppl):20–22
Sinha RK, Cutler M (2012) Effect of cement technique on component position during robotic assisted unicompartmental arthroplasty. J Bone Joint Surg Br 94:219
Swienckowski J, Page BJ 2nd (1989) Medial unicompartmental arthroplasty of the knee. Use of the L-cut and comparison with the tibial inset method. Clin Orthop Relat Res 239:161–167
Acknowledgments
We thank Mr. Jeremy Tolbert from Mako Surgical Corp. for his assistance in digitally stored data collection. We also thank Denise Buckley for her help with manuscript preparation.
Conflict of interest
The authors A. Mofidi, J. F. Plate, B. Lu, and J. E. Lang report no conflict of interest. The authors R. H. Jinnah, G. G. Poehling, and M. A. Conditt have received financial support from MAKO Surgical Corp., Fort Lauderdale, FL, USA. R. H. Jinnah and G. G. Poehling have received payment as consultants. M. A. Conditt receives compensation as Senior Director of Clinical Research. All authors certify that this investigation was performed in conformity with ethical principles of research. Institutional ReviewBoard approval was obtained prior to the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mofidi, A., Plate, J.F., Lu, B. et al. Assessment of accuracy of robotically assisted unicompartmental arthroplasty. Knee Surg Sports Traumatol Arthrosc 22, 1918–1925 (2014). https://doi.org/10.1007/s00167-014-2969-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-014-2969-6