Abstract
Purpose
The authors hypothesise that the trochlear dysplastic distal femur is not only characterised by morphological changes to the trochlea. The purpose of this study is to describe the morphological characteristics of the trochlear dysplastic femur in and outside the trochlear region with a landmark-based 3D analysis.
Methods
Arthro-CT scans of 20 trochlear dysplastic and 20 normal knees were used to generate 3D models including the cartilage. To rule out size differences, a set of landmarks were defined on the distal femur to isotropically scale the 3D models to a standard size. A predefined series of landmark-based reference planes were applied on the distal femur. With these landmarks and reference planes, a series of previously described characteristics associated with trochlear dysplasia as well as a series of morphometric characteristics were measured.
Results
For the previously described characteristics, the analysis replicated highly significant differences between trochlear dysplastic and normal knees. Furthermore, the analysis showed that, when knee size is taken into account, the cut-off values of the trochlear bump and depth would be 1 mm larger in the largest knees compared to the smallest knees. For the morphometric characteristics, the analysis revealed that the trochlear dysplastic femur is also characterised by a 10 % smaller intercondylar notch, 6–8 % larger posterior condyles (lateral–medial) in the anteroposterior direction and a 6 % larger medial condyle in the proximodistal direction compared to a normal femur.
Conclusions
This study shows that knee size is important in the application of absolute metric cut-off values and that the posterior femur also shows a significantly different morphology.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Trochlear dysplasia is the most important morphological abnormality associated with patellar dislocation. The lateral border of the trochlea normally acts as a restraint against the lateral pull of the quadriceps muscle. In a trochleodysplastic knee, the trochlea is shallow, flat or convex [12]. As a result, the patella is unable to engage properly in the trochlear groove and tends to dislocate laterally in early flexion of the knee joint [9].
Many variations in trochlear dysplasia exist, and several authors have tried to capture these variations in a classification system [5, 12, 14, 28]. Henri Dejour [14] was the first to describe three types of trochlear dysplasia based on conventional lateral radiographs, on which he evaluated the crossing sign, the trochlear bump and the trochlear depth. Later, David Dejour implemented the use of CT scans in the Dejour classification and proposed a four-grade classification which is now widely used to grade the severity of trochlear dysplasia and to help in the selection of the appropriate surgical treatment [11–13]. This four-grade Dejour classification is still considered insufficient to quantify the dysplasia and to predict the severity of the patellofemoral instability [29, 30]. Currently, there is an important tendency to reduce the four-grade classification to a two-grade classification [24, 27]; low-grade trochlear dysplasia (type A) can successfully be distinguished from high-grade trochlear dysplasia (types B, C and D), while distinguishing types B, C and D were found difficult due to the high variability of the trochlear geometry [24].
Lately, 3D computer models have also been used to analyse the morphology of trochlear dysplastic knees [4, 41]. Indeed, 3D models provide an accurate and more realistic representation of the complex shape of the trochlea, whereas single cuts have been reported to be unable to fully describe the true morphology of the trochlea [27].
Despite the extensive and profound research on the characteristics of trochlear dysplasia, certain interesting questions are still not fully explored.
From a biomechanical point of view, the actual stability of the patellofemoral joint is not only defined by the subchondral bone, but more importantly also by the shape of the articular cartilage. For many years now, authors have indicated that subchondral osseous anatomy of the patella and the femoral trochlea can differ markedly from the corresponding surface geometry of the articular cartilage, particularly in patients with trochlear dysplasia [33, 35, 37, 38]. Consequently, the cartilage should be included in the 3D models, e.g., using arthro-CT scans. To date, absolute metric cut-off values are frequently used to define the trochlear dysplastic knee, although it has been reported that knee size can influence these measurements [9]. Recent population scale studies have shown that size is the principal mode of variation between subjects, making up for 50–60 % of variation between individuals [7, 16]. Standardisation of size is therefore crucial to allow for objective quantification of morphological differences between subjects. Such can easily be performed by means of generalised Procrustes analysis [18].
The aim of this study is to apply a new method that takes cartilage–bone mismatch and size differences into account to investigate the overall morphology of the distal femur in addition to previously described characteristics associated with trochlear dysplasia. The authors hypothesise that the trochlear dysplastic distal femur is not only characterised by morphological changes to the trochlea.
Materials and methods
A semi-automated landmark-based 3D analysis was performed on the distal femur to quantify the knee joint geometry, including the cartilage. Based on a set of manually identified landmarks, the knees are isotropically scaled to match the standard size and captured in a set of reference planes. This method allows quantification of the widely accepted characteristics associated with trochlear dysplasia, and at the same time, it may reveal morphometric differences between normal and trochleodysplastic knees which have to our knowledge not been reported before.
The trochlear dysplastic group (TD group) included retrospectively 20 patients (12 females and 8 males; mean age 29 ± 13 years) with a history of recurrent patellar dislocation. All patients were diagnosed with trochlear dysplasia and were classified as types A, B, C or D by a senior orthopaedic surgeon (PV) according to the Dejour classification (5 patients in each class) [11–13]. None of these patients underwent a surgical treatment for patellar dislocation prior to imaging.
As a control group, 20 patients with a normal anatomy of the patellofemoral joint were selected (11 females and 9 males; mean age 30 ± 8 years). The control group consulted the orthopaedic surgeon for a complaint unrelated to the patellofemoral area.
Generation and isotropic scaling of 3D computer models
Arthro-CT scans were performed in a supine position with 15° of knee flexion and the toes pointing straight up. The images were loaded in a 3D image processing software system (Mimics 14.12, Materialise, Haasrode, Belgium) to create 3D models of the knee bones including the cartilage.
Because differences in knee size can significantly affect the metric measurements in the trochlear dysplastic knee [9], the 3D models were isotropically sized to match the standard size prior to the analyses. A generalised Procrustes transformation of the surface models was applied in a custom developed code in MATLAB (Matlab 7.8.0, R2009a, Mathworks, Natick, MA) to minimise the total size variance between the knees, while preserving the shape of the knees [19]. The standard size was determined by non-rigid point set registration of the surface meshes of the 20 normal femurs [26]. Twelve anatomical landmarks, covering the extremes of the distal femur in the anteroposterior, proximodistal and mediolateral direction, were defined to calculate a rescaling factor for each knee. This factor was considered as a measure of the femoral size.
Definition of the landmarks
All landmarks were defined in Mimics. For precise location of the landmarks on the femur, tibia and patella, both the 3D models and the arthro-CT images were used. Intra- and inter-observer precision of landmarks defined on 3D models of the femur and tibia has been reported in a range of 0.4–1.4 mm and 0.3–3.5 mm, respectively [39]. Since the landmarks in the current study were also verified on the CT images, it is assumed that the landmark precision in the current study achieves an equivalent or higher level. In the current study, two observers defined the landmarks on 20 knees to assess the intra-observer precision.
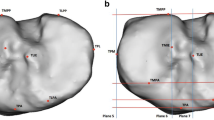
Twelve anatomical landmarks were defined on the femur to rescale the bone models (Fig. 1, “Appendix”). Additionally, 11 other landmarks on the femur, five landmarks on the patella and two landmarks on the tibia (“Appendix”) were defined to create the reference planes and to measure the characteristics associated with trochlear dysplasia.
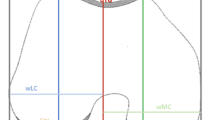
3D model of a distal femur composed of bone (light grey) and cartilage (dark grey). a Location of the 12 landmarks used for Procrustes rescaling: lateral, distal and medial view on the femur. b Reference planes to evaluate the morphometric parameters in the anteroposterior (left and middle figure), mediolateral (middle figure) and proximodistal (left figure) direction and an auxiliary plane (right figure) to evaluate trochlear depth
Definition of the reference planes
The reference planes are predefined by the authors in Mimics in the anteroposterior (AP), proximodistal (PD) and mediolateral (ML) direction (Fig. 1, “Appendix”). By identifying the landmarks, the reference planes are automatically fitted on the geometry of the distal femur.
To evaluate the femur size in the AP direction, three planes were defined: a plane tangent to the anterior cortex of the femur and parallel with the surgical transepicondylar axis (sTEA) (plane 1), a plane tangent to the posterior cortex of the femur and parallel with sTEA (plane 2) and a plane tangent to the posterior condylar line and parallel with the longitudinal axis of the femur (plane 3). To evaluate the femur size in the PD direction, two other planes were defined: a distal plane tangent to the distal condyles (plane 4) and a proximal plane on top of the medial and lateral condyles (plane 5). Finally, seven planes were required to evaluate the proportions in the ML direction: a medial, central and lateral plane, respectively, through the medial epicondyle, the centre of the notch and the lateral epicondyle (planes 6–8), a plane tangent to the internal and external side of the medial and lateral condyle (planes 9–12).
In addition to these reference planes, an auxiliary plane subtended 15° from the perpendicular plane to the tangent plane of the posterior femoral cortex was defined to measure the trochlear depth as described by Dejour et al. [14] (plane 13).
Measurements
Based on this set of landmarks and reference planes, the 3D models were evaluated for a series of well-known characteristics of trochlear dysplasia (hereafter referred to as conventional parameters). These characteristics address the trochlear region as well as the relation of the patella to the trochlea. Furthermore, novel characteristics describing the geometry of the distal femur in its ML, AP and PD aspect (hereafter referred to as morphometric parameters) were also introduced.
Eleven conventional parameters were measured (trochlear depth [14], sulcus angle [25], medial and lateral inclination angle of the trochlea [31, 36], trochlear bump [14], height of the trochlear cartilage [41], Caton-Deschamps index [8], lateral patellar displacement [23], patellar tilt [20], asymmetry of the trochlear facets [28] and asymmetry of the patellar facets [15]). Because most of these parameters were intended to be measured on 2D images without cartilage, the measurement procedure was slightly different (the details of the measurement method are listed in the “Appendix”). In contrast to the original 2D measurements previously published, the current 3D measurements are not flawed by differences in cartilage thickness, differences in knee size and alignment errors, which can considerably affect the measurements in 2D.
Additionally, a series of morphometric parameters of the distal femur were measured in three directions; the ML direction (width of the femur, width of the medial and lateral condyle and the notch), the PD direction (the PD size of the medial and lateral posterior condyle) and AP direction (AP size of the medial and lateral condyle, the medial, lateral and central trochlea and the medial and lateral posterior condyle). And in the axial plane, the angle between the posterior condylar line and the surgical transepicondylar axis was measured (PCL-sTEA angle).
Statistical analysis
All data were analysed using IBM SPSS Statistics 20. To analyse the inter-observer reliability, two observers (AVH and KDR) defined the landmarks on a set of 20 knees (four knees of the control group and 16 knees of the TD group, equally divided over the four Dejour types). The inter-observer reliability of the landmarks was then determined by calculating the intra-class correlation coefficient of the x, y and z coordinates. The intra-class correlation coefficient and the mean error ± SD are reported. The rescaling factor of the control group and the TD group are reported as mean ± SD. A Mann–Whitney U test was performed on the rescaling factor to evaluate if the actual knee size was different in the TD group compared to the control group. The measurements of the conventional and morphometric parameters are reported as mean ± SD. A Mann–Whitney U Test was performed to compare the data in the TD group with the data in the control group. To facilitate visualisation of the parameters, the results of the TD group are expressed as a percentage with respect to the results of the control group. In the TD group, a Spearman’s correlation test was also performed to investigate which parameters correlate with each other and with the order of the applied classification (Dejour types A, B, C and D).
For all statistical tests, a p value less than 0.05 was considered statistically significant.
Results
The knees in the control group (N = 20) were rescaled with a rescaling factor of 1.00 ± 0.08 and the knees in the TD group (N = 20) with a rescaling factor of 1.03 ± 0.07. The rescaling factor did not show a difference between both groups (n.s.). The total study population (N = 40) showed a maximum difference of 35 % in the rescaling factor.
Two observers defined all landmarks on 20 knees (4 of the control group and 16 of the TD group) with an intra-class correlation coefficient of 0.99 (mean error 1.0 ± 1.5 mm).
The results of all the conventional parameters show significant differences between the TD group and the control group (p < 0.05) (Table 1; Fig. 2).
Results of 11 conventional parameters in the TD group, expressed as their ratio to the control group (%). The mean value of the control group is presented as 100 %. For each parameter, the four thin bars represent the results of trochlear dysplasia types A, B, C and D (in this order), the gridded bars represents the mean results of the TD group. A significant difference between the TD group and the control group is indicated by asterisk. A significant correlation (p < 0.05) with the Dejour classification is indicated by dagger
Within the TD group, the lateral inclination angle correlated with the trochlear depth (r = 0.583, p = 0.007) and the medial inclination angle with the trochlear sulcus angle (r = −0.674, p = 0.001). The assigned four-grade Dejour classification showed a correlation with an increased trochlear sulcus angle (r = 0.636, p = 0.003) and patellar tilt (r = 0.465, p = 0.039).
For the morphometric measurements, there were significant differences between the TD group and the control group in the three directions (Table 2; Figs. 3, 4). In the AP direction, the distal femur was larger at three locations in the TD group; the medial posterior condyle was 6 % larger (p = 0.048), the lateral posterior condyle was 8 % larger (p = 0.013) and the total medial condyle was 3 % larger (p = 0.009) compared to the control group. In the ML direction, the distal femur was smaller at two measured levels in the TD group; the total width was 1 % smaller (p = 0.045), and the notch was 10 % smaller (p = 0.037). In the PD direction, the medial posterior condyle was 4 % larger in the TD group (p = 0.021).
Results of 12 morphometric parameters in the AP, PD and ML direction in the TD group, expressed as their ratio to the control group (%). The mean value of the control group is presented as 100 %. For each parameter, the four thin bars represent the results of trochlear dysplasia types A, B, C and D (in this order), the gridded bars represents the mean results of the TD group. A significant difference between the TD group and the control group is indicated by asterisk. A significant correlation (p < 0.05) with the Dejour classification is indicated by dagger
Within the TD group, the size of the lateral trochlea correlated with the trochlear depth (r = 0.684, p = 0.001) and with the lateral inclination angle (r = 0.492, p = 0.028), while the size of the medial trochlea correlated with the trochlear bump (r = 0.839, p < 0.001).
The assigned four-grade Dejour classification correlated negatively with the AP size of the lateral condyle (r = −0.504, p = 0.023) and the lateral trochlea (r = −0.465, p = 0.039).
The PCL-sTEA angle did not differ between the control group (2.6 ± 1.6°) and the TD group (2.6 ± 2.3°) (n.s.).
Discussion
The most important finding in this study is the evidence that dysplasia of the trochlea is accompanied by abnormalities in the posterior condylar morphology, which was demonstrated by applying a novel semi-automated landmark-based analysis on a set of normal and trochlear dysplastic rescaled knee bones. As such, it can be stated that isolated trochlear dysplasia does not exist; the affected knees are also characterised by a smaller intercondylar notch, larger posterior condyles in the AP direction and a larger medial condyle in the PD direction.
Rescaling is particularly important in the interpretation of metric parameters used for diagnostic purposes. The current population of 40 subjects showed up to 35 % difference in knee size. This difference in size is based on the rescaling factor, which takes the size in the AP, PD and ML direction into account (e.g. the ML size prior to rescaling showed a range of 66.8–94.5 mm).
In this study, the rescaled distal femur models were investigated by applying a predefined set of reference planes to measure a series of conventional parameters together with a set of overall morphometric characteristics.
Regarding the conventional parameters, this study replicates the highly significant differences between trochlear dysplastic knees and normal knees, which confirm that the proposed method is suitable for measuring these parameters. An important difference, however, between the current study and most previous studies is that the cartilage was included in the current analyses. Because the cartilage tends to be thicker at the deepest point of the trochlea [37], the trochlear depth was smaller, the sulcus angle was larger and the medial and lateral inclination angles were smaller in the current study compared to measurements in previous studies [14, 36]. Trochlear bump, which is a measure of trochlear floor elevation, is traditionally measured between the anterior cortex and the deepest point of the trochlear floor [14]. Obviously, if the cartilage on the trochlear floor is taken into account, a larger trochlear bump will be measured. Arthro-CT is proven to be an appropriate imaging method to generate 3D bone–cartilage models [1].
For the morphometric parameters, to date, limited research results were published focussing on the AP and ML size of the distal femur affected by trochlear dysplasia. In 2000, Pfirrmann et al. [28] found no significant differences in terms of the AP size of the medial and lateral femoral condyles and their ratio, the absolute and relative ML width of the distal femur and the trochlea, and the distance between the deepest point of the trochlear floor and the posterior condylar line. These findings do not correspond with the current study and with the findings of Biedert and Bachmann [5]. This might be due to the smaller population size in the first study, to the inclusion criteria or to the location of the axial slice where the measurement took place. In 2009, Biedert and Bachmann investigated the AP size of the distal femur in relation to the ML size to detect if a decreased trochlear depth is caused by a heightened trochlear floor or by a flattened lateral and/or medial trochlea. In line with the current results, they found that the ratio was not altered at the level of the lateral trochlea and that it was increased at the level of the centre and medial trochlea. With the use of the reference planes, it was possible to look into these measurements in more detail. In the TD group, the posterior part of the lateral condyle was 8 % longer, while the lateral trochlea tended to be 7 % shorter (not significant) compared to the control group. So even in knees in which the total lateral condyle is unaltered, it is possible that a difference in the posterior part compensates for a difference in the anterior part. At the level of the medial trochlea, the current data suggest that the enlarged medial condyle in the TD group is at least partially due to an increase in the posterior condyle. The mean medial trochlea was also increased by 24 %, but this increase was not significant. At the level of the centre of the trochlea, the anterior offset (trochlear bump) was substantially increased in the TD group, which is in agreement with earlier findings of numerous other authors [4, 6, 14].
In the ML direction, the overall width of the trochleodysplastic knees was slightly smaller compared to the normal group, while the size of the medial and lateral condyles shows no significant differences. This small difference in the overall width of the distal femur can be explained by the narrow intercondylar notch in the TD group. Intercondylar notch size and shape are known to be related to ACL damage and osteoarthritis [2, 32, 34], but has not yet been reported in association with trochlear dysplasia.
Limitations of this study include the relatively small number of subjects and the use of a four-grade classification system which has recently been shown to be insufficient to distinguish types B, C and D from one another [24]. In the current study, only one senior surgeon evaluated the arthro-CT scans to classify the patients and the intra-observer agreement was not tested. So it could be argued that the reliability of the patient group classification is insufficient. Testing the repeatability of the Dejour classification or assigning new characteristics to the classification, however, was beyond the scope of this study. The Dejour classification was only used to evaluate if the measured parameters correlated with the assigned classification. The focus of this study was on the morphological differences between normal and trochlear dysplastic knees.
A Procrustes rescaling procedure was performed to rule out absolute knee size differences from the analysis. It must be noted that gender, femoral size and to a lesser extent somatotype (endo-, meso- and ectomorph type) are also known to influence the geometry of the distal femur in general and more specifically the ML/AP ratio of the distal femur [3, 5]. In the current study, the control group and TD group were controlled for sex as well as size differences, while somatotype was not analysed.
In clinical settings, generating and rescaling 3D models set rather high technical requirements and are considerably more time-consuming than conventional measurements on axial or lateral slices. Technological evolution, however, might improve the cost, time-efficiency and complexity of these processes. In anticipation of such developments, this study shows that size differences irrespective of the type of measurement can considerably affect the interpretation of measurements. For example, knees are found pathological if the trochlear bump is equal to or greater than 3 mm or if the trochlear depth is less than or equal to 4 mm [14]. If these cut-off values would be adjusted for the size differences observed in the current data set, these values would be 1 mm larger in the largest knees compared to the smallest knees. This difference is of clinical importance, since clinicians might rely on these absolute cut-off values to diagnose or grade TD. Since the Dejour classification is not only applied to grade the severity but also to assist in the treatment decision, over- or underestimation of morphological characteristics might indirectly lead to inappropriate treatment. Moreover, the presence of a trochlear bump (Dejour types B and D) is a key factor to decide if trochleoplasty is designated [10, 13, 17]. A recent study by Dejour et al. [10] brought the problem of undiagnosed or underestimated trochlear dysplasia to the attention. Dejour et al. indicated that missed high-grade dysplasia (defined as types B and D) may lead to inadequate treatment of patellar dislocation. Sizing the knees, as applied in the current study, provides a more accurate measure of the morphological characteristics, such as the trochlear bump, which can improve identification of high-grade dysplasia and consequently improve treatment decision. To date, 3D modelling and sizing sets high technological and time-consuming requirements, but with the vast evolution of technology, these processes might become more accessible in clinical settings.
Conclusions
With this semi-automated landmark-based 3D analysis of rescaled bones evidence was found that not only the trochlear region is affected in the trochleodysplastic femur, but that the posterior femur also has a significantly different morphology. The proposed method successfully excluded differences in cartilage thickness, differences in knee size and alignment errors from the analysis.
Notes
Because the CT scan only captured the knee joint, the longitudinal axis of the femur was estimated by drawing 2 circles on a sagittal view through the shaft of the femur as described by Biedert et al. [6].
Abbreviations
- TD group:
-
Trochlear dysplastic group
- AP:
-
Anteroposterior
- PD:
-
Proximodistal
- ML:
-
Mediolateral
- sTEA:
-
Surgical transepicondylar axis
- PCL:
-
Posterior condylar line
- ACL:
-
Anterior cruciate ligament
- FME:
-
Femoral medial epicondyle
- FMS:
-
Femoral medial sulcus
- FLE:
-
Femoral lateral epicondyle
- FMCP:
-
Femoral medial condyle posterior
- FLCP:
-
Femoral lateral condyle posterior
- FMCD:
-
Femoral medial condyle distal
- FLCD:
-
Femoral lateral condyle distal
- FMTA:
-
Femoral medial trochlea anterior
- FLTA:
-
Femoral lateral trochlea anterior
- FMCPP:
-
Femoral medial condyle posterior proximal
- FLCPP:
-
Femoral lateral condyle posterior proximal
- PE:
-
Proximal edge of the trochlea cartilage
- FMCIP:
-
Femoral medial condyle internal point
- FMCEP:
-
Femoral medial condyle external point
- FLCIP:
-
Femoral lateral condyle internal point
- FLCEP:
-
Femoral lateral condyle external point
- PL:
-
Patella lateral point
- PM:
-
Patella medial point
- PS:
-
Patella superior point
- PI:
-
Patella inferior point
References
Allen BC, Peters CL, Brown NAT, Anderson AE (2010) Acetabular cartilage thickness: accuracy of three-dimensional reconstructions from multidetector CT Arthrograms in a cadaver study. Radiology 255(2):544–552
Anderson AF, Lipscomb AB, Liudahl KJ, Addlestone RB (1987) Analysis of the intercondylar notch by computed-tomography. Am J Sport Med 15(6):547–552
Bellemans J, Carpentier K, Vandenneucker H, Vanlauwe J, Victor J (2010) The John Insall Award: both morphotype and gender influence the shape of the knee in patients undergoing TKA. Clin Orthop Relat Res 468:29–36
Biedert R, Sigg A, Gal I, Gerber H (2011) 3D representation of the surface topography of normal and dysplastic trochlea using MRI. Knee 18(5):340–346
Biedert RM, Bachmann M (2009) Anterior-posterior trochlear measurements of normal and dysplastic trochlea by axial magnetic resonance imaging. Knee Surg Sports Traumatol Arthrosc 17(10):1225–1230
Biedert RM, Netzer P, Gal I, Sigg A, Tscholl PM (2011) The lateral condyle index: a new index for assessing the length of the lateral articular trochlea as predisposing factor for patellar instability. Int Orthop 35(9):1327–1331
Bredbenner TL, Eliason TD, Potter RS, Mason RL, Havill LM, Nicolella DP (2010) Statistical shape modeling describes variation in tibia and femur surface geometry between control and incidence groups from the osteoarthritis initiative database. J Biomech 43(9):1780–1786
Caton J, Deschamps G, Chambat P, Lerat JL, Dejour H (1982) The low patellas—report of 128 cases—patella-inferae. Rev Chir Orthop 68(5):317–325
Chhabra A, Subhawong TK, Carrino JA (2011) A systematised MRI approach to evaluating the patellofemoral joint. Skeletal Radiol 40(4):375–387
Dejour D, Byn P, Ntagiopoulos PG (2013) The Lyon’s sulcus-deepening trochleoplasty in previous unsuccessful patellofemoral surgery. Int Orthop 37(3):433–439
Dejour D, Le Coultre B (2007) Osteotomies in patello-femoral instabilities. Sports Med Arthrosc 15(1):39–46
Dejour D, Reynaud P, Lecoultre B (1998) Douleurs et instabilité rotulienne. Essai de classification. Méd Hyg 56:1466–1471
DeJour D, Saggin P (2010) The sulcus deepening trochleoplasty—the Lyon’s procedure. Int Orthop 34(2):311–316
Dejour H, Walch G, Nove-Josserand L, Guier C (1994) Factors of patellar instability: an anatomic radiographic study. Knee Surg Sports Traumatol Arthrosc 2(1):19–26
Endo Y, Schweitzer ME, Bordalo-Rodrigues M, Rokito AS, Babb JS (2007) MRI quantitative morphologic analysis of patellofemoral region: lack of correlation with chondromalacia patellae at surgery. Am J Roentgenol 189(5):1165–1168
Fitzpatrick CK, Baldwin MA, Laz PJ, FitzPatrick DP, Lerner AL, Rullkoetter PJ (2011) Development of a statistical shape model of the patellofemoral joint for investigating relationships between shape and function. J Biomech 44(13):2446–2452
Fucentese SF, Zingg PO, Schmitt J, Pfirrmann CW, Meyer DC, Koch PP (2011) Classification of trochlear dysplasia as predictor of clinical outcome after trochleoplasty. Knee Surg Sports Traumatol Arthrosc 19(10):1655–1661
Goodall C (1991) Procrustes methods in the statistical-analysis of shape. J R Stat Soc B 53(2):285–339
Gower JC (1975) Generalized procrustes analysis. Psychometrika 40(1):33–51
Grelsamer RP, Bazos AN, Proctor CS (1993) Radiographic analysis of patellar tilt. J Bone Jt Surg Br 75(5):822–824
LaPrade RE, Engebretsen AH, Ly TV, Johansen S, Wentorf FA, Engebretsen L (2007) The anatomy of the medial part of the knee. J Bone Jt Surg Am 89A(9):2000–2010
LaPrade RF, Ly TV, Wentorf FA, Engebretsen L (2003) The posterolateral attachments of the knee—a qualitative and quantitative morphologic analysis of the fibular collateral ligament, popliteus tendon, popliteofibular ligament, and lateral gastrocnemius tendon. Am J Sport Med 31(6):854–860
Laurin CA, Levesque HP, Dussault R, Labelle H, Peides JP (1978) Abnormal lateral patello-femoral angle—diagnostic roentgenographic sign of recurrent patellar subluxation. J Bone Jt Surg Am 60(1):55–60
Lippacher S, Dejour D, Elsharkawi M, Dornacher D, Ring C, Dreyhaupt J, Reichel H, Nelitz M (2012) Observer agreement on the Dejour trochlear dysplasia classification. A comparison of true lateral radiographs and axial magnetic resonance images. Am J Sport Med 40(4):837–843
Merchant AC, Mercer RL, Jacobsen RH, Cool CR (1974) Roentgenographic analysis of patellofemoral congruence. J Bone Jt Surg Am A 56(7):1391–1396
Myronenko A, Song XB (2010) Point set registration: coherent point drift. IEEE T Pattern Anal 32(12):2262–2275
Nelitz M, Lippacher S, Reichel H, Dornacher D (2013) Evaluation of trochlear dysplasia using MRI: correlation between the classification system of Dejour and objective parameters of trochlear dysplasia. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-012-2321-y
Pfirrmann CWA, Zanetti M, Romero J, Hodler J (2000) Femoral trochlear dysplasia: MR findings. Radiology 216(3):858–864
Remy F, Chantelot C, Fontaine C, Demondion X, Migaud H, Gougeon F (1998) Inter- and intraobserver reproducibility in radiographic diagnosis and classification of femoral trochlear dysplasia. Surg Radiol Anat 20(4):285–289
Remy F, Gougeon F, Eddine TA, Migaud H, Fontaine C, Duquennoy A (2002) Reproducibility of the new classification of femoral trochlea dysplasia proposed by Dejour: predictive value for severity of femoropatellar instability in 47 knees. J Bone Jt Surg Br 84:1
Sasaki T, Yagi T (1986) Subluxation of the patella—investigation by computerized-tomography. Int Orthop 10(2):115–120
Shepstone L, Rogers J, Kirwan JR, Silverman BW (2001) Shape of the intercondylar notch of the human femur: a comparison of osteoarthritic and non-osteoarthritic bones from a skeletal sample. Ann Rheum Dis 60(10):968–973
Shih YF, Bull AM, Amis AA (2004) The cartilaginous and osseous geometry of the femoral trochlear groove. Knee Surg Sports Traumatol Arthrosc 12(4):300–306
Sonnery-Cottet B, Archbold P, Cucurulo T, Fayard JM, Bortolletto J, Thaunat M, Prost T, Chambat P (2011) The influence of the tibial slope and the size of the intercondylar notch on rupture of the anterior cruciate ligament. J Bone Jt Surg Br 93B(11):1475–1478
Staubli HU, Durrenmatt U, Porcellini B, Rauschning W (1999) Anatomy and surface geometry of the patellofemoral joint in the axial plane. J Bone Jt Surg Br 81B(3):452–458
Stefanik JJ, Roemer FW, Zumwalt AC, Zhu YY, Gross KD, Lynch JA, Frey-Law LA, Lewis CE, Guermazi A, Powers CM, Felson DT (2012) Association between measures of trochlear morphology and structural features of patellofemoral joint osteoarthritis on MRI: the MOST study. J Orthopaed Res 30(1):1–8
Toms AP, Cahir J, Swift L, Donell ST (2009) Imaging the femoral sulcus with ultrasound, CT, and MRI: reliability and generalizability in patients with patellar instability. Skeletal Radiol 38(4):329–338
van Huyssteen AL, Hendrix MRG, Barnett AJ, Wakeley CJ, Eldridge JDJ (2006) Cartilage-bone mismatch in the dysplastic trochlea—an MRI study. J Bone Jt Surg Br 88B(5):688–691
Victor J, Van Doninck D, Labey L, Innocenti B, Parizel PM, Bellemans J (2009) How precise can bony landmarks be determined on a CT scan of the knee? Knee 16(5):358–365
Victor J, Wong P, Witvrouw E, Sloten JV, Bellemans J (2009) How isometric are the medial patellofemoral, superficial medial collateral, and lateral collateral ligaments of the knee? Am J Sports Med 37(10):2028–2036
Yamada Y, Toritsuka Y, Yoshikawa H, Sugamoto K, Horibe S, Shino K (2007) Morphological analysis of the femoral trochlea in patients with recurrent dislocation of the patella using three-dimensional computer models. J Bone Jt Surg Br 89B(6):746–751
Acknowledgments
This work was funded by a Research fund of University College Ghent.
Ethical standard
The authors confirm that the study has been approved by the ethics committee and has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. All persons gave their informed consent prior to their inclusion in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Landmarks femur for rescaling
-
Femoral medial epicondyle (FME): the most anterior and distal osseous prominence over the medial aspect of the medial femoral condyle [21, 39, 40]
-
Femoral medial sulcus (FMS): depression on the bony surface slightly proximal and posterior to FME [21, 39]
-
Femoral lateral epicondyle (FLE): the most anterior and distal osseous prominence over the lateral aspect of the lateral femoral condyle [22, 39]
-
Femoral medial condyle posterior (FMCP): the most posterior point of the medial condyle on the 3D model of the femur, aligned along the longitudinal axis of the femur
-
Femoral lateral condyle posterior (FLCP): the most posterior point of the lateral condyle on the 3D model of the femur, aligned along the longitudinal axis of the femurFootnote 1
-
Femoral medial condyle distal (FMCD): the most distal point of the medial condyle on the 3D model of the femur, aligned along the longitudinal axis of the femur
-
Femoral lateral condyle distal (FLCD): the most distal point of the lateral condyle on the 3D model of the femur, aligned along the longitudinal axis of the femur
-
Femoral medial trochlea anterior (FMTA): the most anterior point of the medial trochlea on the 3D model of the femur, aligned along the longitudinal axis of the femur
-
Femoral lateral trochlea anterior (FLTA): the most anterior point of the lateral trochlea on the 3D model of the femur, aligned along the longitudinal axis of the femur
-
Femoral medial condyle posterior proximal (FMCPP): the most proximal point of the cartilage on the posterior medial condyle, aligned along the longitudinal axis of the femur
-
Femoral lateral condyle posterior proximal (FLCPP): the most proximal point of the cartilage on the posterior lateral condyle, aligned along the longitudinal axis of the femur
-
Notch: the most anterior point in the middle of the femoral notch on a caudal to cranial view of the femur, aligned along the longitudinal axis of the femur.
Additional landmarks
Femur
-
Deepest point on the trochlea along a plane subtended 15° from the perpendicular to the tangent of the posterior femoral cortex [14]
-
Highest point on the trochlea along a plane subtended 15° from the perpendicular to the tangent of the posterior femoral cortex [14]
-
Proximal edge of the trochlea cartilage (PE) [41]
-
Femoral medial condyle internal point (FMCIP): most lateral point of the cartilage of the medial condyle in the axial view of the notch
-
Femoral medial condyle external point (FMCEP): most medial point of the cartilage of the medial condyle in the axial view of the notch
-
Femoral lateral condyle internal point (FLCIP): most medial point of the cartilage of the lateral condyle in the axial view of the notch
-
Femoral lateral condyle external point (FLCEP): most lateral point of the cartilage of the lateral condyle in the axial view of the notch
-
Femoral lateral trochlea: the most anterior point of the lateral trochlea in the axial slice of the femoral lateral condyle posterior
-
Femoral medial trochlea: the most anterior point of the medial trochlea in the axial slice of the femoral lateral condyle posterior
-
Trochlear deepest point: the deepest point of the trochlea in the axial slice of the femoral lateral condyle posterior
-
Trochlear most prominent point: most prominent point of the trochlear groove.
Patella
-
Patella lateral point (PL): most lateral point of the articular surface of the patella
-
Patella medial point (PM): most medial point of the articular surface of the patella
-
Patella superior point (PS): most superior point of the articular surface of the patella
-
Patella inferior point (PI): most inferior point of the articular surface of the patella
-
Patella ridge: point on the border of the medial and lateral facet of the patella.
Tibia
-
Tuberositas tibiae
-
Most anterior point of the tibial articulating surface of the tibia.
Reference planes
-
Plane 1: anterior sTEA plane: plane tangent to the anterior cortex, parallel to the surgical transepicondylar axis (OF: plane parallel to the surgical transepicondylar axis, parallel to FAAx and through the anterior cortex)
-
Plane 2: posterior sTEA plane: plane tangent to the posterior cortex, parallel to the surgical transepicondylar axis (OF: plane parallel to the surgical transepicondylar axis, parallel to FAAx and through the posterior cortex)
-
Plane 3: PCL plane: plane through FMCP and FLCP and parallel to FAAx
-
Plane 4: distal condylar plane: plane through FMCD and FLCD and perpendicular to FAAx
-
Plane 5: proximal condylar plane: plane through FMCPP and FLCPP and perpendicular to FAAx
-
Plane 6: medial plane: plane through FME, parallel to FAAx and perpendicular to PCL
-
Plane 7: notch plane: plane through the notch, parallel to FAAx and perpendicular to PCL
-
Plane 8: lateral plane: plane through FLE, parallel to FAAx and perpendicular to PCL
-
Plane 9: internal medial condyle plane: plane through FMCIP, parallel to FAAx and perpendicular to PCL
-
Plane 10: external medial condyle plane: plane through FMCEP, parallel to FAAx and perpendicular to PCL
-
Plane 11: internal lateral condyle plane: plane through FLCIP, parallel to FAAx and perpendicular to PCL
-
Plane 12: external lateral condyle plane: plane through FLCEP, parallel to FAAx and perpendicular to PCL
-
Plane 13: trochlear depth plane: plane subtended 15° from the perpendicular to the tangent of the posterior femoral cortex to measure the trochlear depth as defined by Dejour et al. [14].
Measurements
Characteristics of trochlear dysplasia
-
Trochlear depth (mm): distance between the highest and deepest point of the trochlea measured perpendicular to plane 3
-
Sulcus angle (°): the angle formed by the intersection of the medial and lateral trochlear facets measured in the axial slice of FLCP
-
Lateral inclination angle of the trochlea (°): the angle between the posterior condylar line and a line along the lateral facet measured in the axial slice of FLCP
-
Medial inclination angle of the trochlea (°): the angle between the posterior condylar line and a line along the medial facet measured in the axial slice of FLCP
-
Trochlear bump (mm): the distance between plane 1 and the most prominent point of the trochlear groove
-
Trochlear cartilage height (mm): the distance between plane 4 and PE
-
Caton-Deschamps index (−): the ratio of the distance between PI and most anterior point of the tibial articulating surface to the length of the articular surface of the patella
-
Lateral patellar displacement (mm): the distance from PM to the perpendicular to plane 3, passing through the most anterior point of the medial condyle
-
Patellar tilt (°): the angle between the patellar width line (line between PL and PM) and PCL
-
Asymmetry trochlear facets (%): percentage of the medial to the lateral trochlear facet length (medial facet/lateral facet × 100)
-
Asymmetry patellar facets (%): percentage of the medial to the lateral patellar facet length (medial facet/lateral facet length × 100).
Morphometric characteristics
-
Medial femur: distance between the FMTA and plane 3
-
Medial trochlea: distance between FMTA and plane 1
-
Medial posterior condyle: distance between the FMCP and plane 2
-
Lateral femur: distance between FLTA and plane 3
-
Lateral trochlea: distance between FLTA and plane 1
-
Lateral posterior condyle: distance between FLCP condyle and plane 2
-
Medial condyle: distance between FMCPP and plane 4
-
Lateral condyle: distance between FLCPP and plane 4
-
Distal femur: distance between FLE and plane 6
-
Medial condyle: distance between FMCIP and plane 10
-
Lateral condyle: distance between FLCIP and plane 12
-
Notch width: distance between FMCIP and plane 11.
Rights and permissions
About this article
Cite this article
Van Haver, A., De Roo, K., De Beule, M. et al. Semi-automated landmark-based 3D analysis reveals new morphometric characteristics in the trochlear dysplastic femur. Knee Surg Sports Traumatol Arthrosc 22, 2698–2708 (2014). https://doi.org/10.1007/s00167-013-2573-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-013-2573-1