Abstract
Purpose
The study aimed to compare trochlear profiles of various total knee arthroplasty (TKA) models to anatomic profiles observed in healthy and pathologic knees and to evaluate trochlear designs against radiologic indicators for PF disorders and trochlear dysplasia.
Methods
The trochlear profiles of 14 different TKA models were digitized using a coordinate measurement machine at various flexion angles (0°, 15°, 30° and 45°) to deduce the following variables: sulcus angle, trochlear groove orientation, height of lateral facet, and mediolateral groove position. The effect of externally rotating the femoral component on those variables was simulated.
Results
The sulcus angle was greater than the indicators for trochlear dysplasia of 144° in 11 implants at 45° flexion, and in 13 implants at 30° flexion. The lateral facet height was less than average anatomic values of 5 mm in eight specimens through the entire range of early flexion (0°–30°). The trochlear groove was oriented laterally in 13 specimens (3.3°–11.7°) and was vertical in one specimen (0.3°). Applying an external rotation up to 6° resulted in noticeable lateral translation of the trochlear groove and facets, but negligible posterior translation.
Conclusions
The study presented a detailed description of previously overlooked TKA design parameters and revealed that some femoral components exhibit characteristics of trochlear dysplasia. The clinical relevance of this descriptive study is that surgeons should be aware of such design limitations to improve choice of implant for patients with history of PF disorders and to adapt surgical techniques as necessary to optimize PF tracking.
Level of evidence
Case–control study, Level III.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Patellofemoral (PF) complications are a common cause of patient dissatisfaction and a reason for revision after total knee arthroplasty (TKA) [1, 36], including anterior knee pain, patellar crepitus, and less frequently patellar subluxation, dislocation and fracture [20, 22, 36]. Such complications are usually caused by a combination of factors related to surgical technique (e.g. implant positioning and sizing, soft-tissue balancing, patellar resurfacing, etc.) and to implant design (e.g. trochlear depth, sagittal curvature, patellar component design) [22, 34, 44]. A number of biomechanical studies, using three-dimensional computer models of various TKA designs, suggest that even if the surgical technique is optimized, PF tracking is not always restored to physiological values, because the prosthetic trochlear groove may still be different from the normal trochlea, implying that related complications could arise due to implant design limitations [3, 34, 43, 44].
In a normal knee presenting PF disorders, the standard diagnostic approach is to inspect for the presence of trochlear dysplasia, patella alta, excessive patellar tilt or excessive tibial tuberosity to trochlear groove distance (TT-TG) [5, 13, 35, 38]. Trochlear dysplasia is the most frequent anatomic factor, observed in 96 % of patients suffering from patellar dislocation [15]. It can be assessed radiographically by measuring the sulcus angle in the ‘skyline view’: with the knees in 45° of flexion as defined by Merchant [29, 30] or with the knees in 30° of flexion as defined by Brattström [11]; and by looking for the ‘crossing sign’ of the trochlear groove in the sagittal view [15]. The average sulcus angle for healthy knees is 138° in the ‘merchant view’ [30, 32] and 142° in the ‘Brattström view’ [11]. The sulcus angle in knees with trochlear dysplasia is generally over 144° in the ‘Merchant view’ [41] or over 143° in the ‘Brattström view’ [12]. In severe cases of PF disorders, the anatomic anomalies can be addressed surgically by realignment of the extensor mechanism, medial PF ligament reconstruction, trochleoplasty or tibial tuberosity osteotomy; and in the presence of arthritis by PF arthroplasty or TKA [4, 10, 14, 21, 36].
In a TKA with PF complications the usual diagnostic approach is to inspect for the aforementioned conditions, with the exception of trochlear dysplasia, because it is generally assumed that implants are designed with adequate trochlear depth, width, sulcus angle, and groove orientation. In severe cases, revision surgery could be performed to resurface the patella if that was not already done or to change implant sizing or rotation [36]. It is perhaps because PF complications are multi-factorial that surgeons often overlook flaws in implant design. Implant manufacturers provide little or no useful information on the design of the trochlear compartment of their TKA implants, other than stating that the design is “anatomic”, or that the trochlear groove is inclined to reproduce the angulation between the mechanical and anatomic femoral axes [24]. Visual comparison of different TKA designs may reveal that trochlear compartments have various proximal lengths or mediolateral widths, but the trochlear depth and sulcus angle remain indiscernible, possibly due to the reflections off the shiny surfaces. There are little published data on trochlear designs of TKAs [37, 44], which are often difficult for the surgeon to perceive or quantify. In our experience, some prosthetic trochleae are designed with parameters close to those of natural healthy knees, whereas other prosthetic trochleae are excessively shallow and therefore exhibit pathologic or dysplastic properties.
In this study, the authors aimed to test the hypothesis that TKA implants are not always designed with anatomic trochlear parameters, such as sulcus angle, trochlear facet heights and groove orientation, and that some designs exhibit characteristics of trochlear dysplasia. The study aimed to (1) quantify the differences in TKA design parameters used in assessment of PF complications of trochlear dysplasia in the normal knee and (2) raise awareness about the different trochlear designs of common TKAs and the their potential drawbacks. Such knowledge could help surgeons to adapt their operative techniques to optimize the extensor mechanism kinematics for the implant used. Because PF complications are usually caused by a combination of factors related to surgical technique and implant design, the authors did not attempt to correlate the findings with clinical results of the studied implants.
Materials and methods
The authors formed a sample of 14 femoral components from commercially available TKAs. Each specimen was numbered and identified by its laser marking to determine its manufacturer, model, serial number, size and side. The anteroposterior (AP) and mediolateral (ML) dimensions of each specimen were measured using a Vernier caliper (Digimatic, Mitutoyo, Japan) with precision of 0.1 mm (Table 1). To facilitate visual reference to implants, each specimen was photographed in the frontal, sagittal and transverse views (Fig. 1).
An alignment jig was designed to enable consistent mounting of specimens within a bench angular divider, with rotation range of 360° and precision of 0.5° (Fig. 2). The alignment jig consisted of a threaded steel axle, adjusting the distance between two steel blocks, which supported the distal resection surface of the femoral component and fastened it by exertion of pressure mediolaterally. Each femoral component was in turn mounted onto the alignment jig and placed within a digital Coordinate Measurement Machine (CMM Legex Series, Mitutoyo, Japan) with the trochlea facing upwards. A spirit level was used to set the angular divider to zero when the distal resection surface was perfectly vertical.
The CMM was fitted with a one-millimetre measurement probe (MPP-310Q, Mitutoyo, Japan), giving a resolution of 0.01 mm and a scanning accuracy of 0.03 mm. The probe was manipulated using its manual remote control to record coordinates of the following landmarks: with the angular divider at 0° of rotation (femoral component horizontal) (1) the rotation axis of angular divider; (2) the most anterior point on the trochlea; (3) the most medial and most lateral points on the distal condyles; and with the angular divider at 90° of rotation (femoral component vertical) (4) the most posterior points on the posterior condyles; (5) four dispersed points on the posterior resection place and (6) four dispersed points on the anterior resection plane. The CMM was then programmed to automatically digitize the ML line that intersected the highest point on the trochlea: with the angular divider at the following angles: 0° of flexion; 15° of flexion; 30° of flexion and 45° of flexion.
All recorded coordinates were exported from the CMM and converted into spreadsheets using Microsoft® Excel (Microsoft Corp, Redmond, WA, USA). To speed data processing, the coordinates were imported into preformatted spreadsheets specifically for right- and left-sided femoral components, and macros were used to perform the required rotations, translations and measurements. The three-dimensional equations of the anterior and posterior resection planes were calculated, using the method of least squares, from the four points digitized on each of the planes. This enabled calculation of the inclination angles between the two planes for each specimen by simple trigonometry calculations.
To enable consistent geometric comparisons between all specimens, the coordinates of right-sided implants were mirrored to become superimposable with those of left-sided implants, and the scale of each axis was represented as a ratio to the respective ML and AP dimensions of each specimen. The two-dimensional ML profiles of each prosthetic trochlea could therefore be superposed and compared in a non-dimensional coordinate system, with its origin at the intersection of (1) the midpoint between the medial and lateral margin of each specimen and (2) the trochlear groove, or deepest point on the sulcus, of each profile. Measurements in the ML plane were expressed in a 0–100 % scale, where 0 % corresponded to the medial margin and 100 % corresponded to the lateral margin of the component.
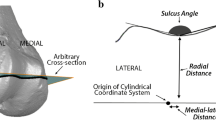
The ‘sulcus angle’ of each profile was calculated from the coordinates of the trochlear groove and those of the highest points of the medial and lateral facets (Fig. 3). The coordinates of the trochlear grooves were used to calculate a linear regression (using the method of least squares) in the frontal plane, and the trochlear groove orientation was calculated from the cosine of its gradient (Fig. 4). Furthermore, it was possible to apply virtual rotations to the trochlear profiles, to perceive the effect of externally rotating the femoral component about the intramedullary femoral axis, as applied in the surgical setting. The resulting lateral translation and minor posterior translation of the trochlear groove and facets was noted for external rotation up to 6° of about the geometric centre of the implant (Fig. 5).
Example of a two-dimensional trochlear profile at 30° of flexion (specimen #6), plotted in absolute coordinates (mm). The dashed vertical lines indicate the lateral and medial margins of the implant, and the letters indicate points of inflexion of the trochlear profile: A medial extremity B peak of medial facet C sulcus trough D peak of lateral facet E lateral extremity. The sulcus angle is ∠BCD, the height of the lateral facet is the z-coordinate difference between points C and D, and the trochlear groove is the x-coordinate of point C
The effect of applying 6° of external rotation about the geometric centre of the implant (C). The trochlear groove and lateral facet are translated proportionally to the rotation angle (θ) and radius (H). The following approximations apply for small angles (<10°) of external rotation. Lateral translation = H(sin θ); posterior translation = H(1 − cos θ)
Results
The trochlear profiles of all 14 implants at 30° of flexion are presented in a non-dimensional coordinate system to enable direct visual comparisons (Fig. 6).
The sulcus angles of all trochlear profiles are presented graphically (Fig. 7). Eleven specimens had a sulcus angle greater than 144° in the ‘Merchant view’ (45° of flexion), and 13 specimens had a sulcus angle greater than 143° in the ‘Brattström view’ (30° of flexion). We observed different sulcus angle progressions in the range of early flexion (0°–30°): a considerable decrease in seven specimens, a nearly constant value in five specimens, and a slight increase in two specimens. The sulcus angle progressions were irregular beyond early flexion (30°–45°), where the trochlea widens rapidly as it merges with the femoral condyles.
The heights of the lateral facets of all trochlear profiles are presented graphically (Fig. 8). Visual comparison reveals that the lateral trochlear facet height is inversely proportional to the sulcus angle. Eight specimens had a facet less than 5 mm high through the entire range of early flexion (0°–30°), and five specimens had a facet less than 5 mm high beyond early flexion (30°–45°).
The trochlear groove was oriented laterally in 13 specimens (3.3°–11.7°) and was vertical in one specimen (0.3°). The ML position of the groove in the components at 0° of flexion was 51–60 %, and at 45° of flexion was 46–54 %. Applying an external rotation up to 6° to specimens resulted in noticeable lateral translation of the trochlear groove and facets, but produced negligible posterior translation to these points (Fig. 5). The lateral and posterior translations were mathematically related to implant size and the rotation angle: Lateral translation = H(sin θ) and Posterior translation = H(1 – cos θ), where ‘H’ is the distance between the trochlea and the centre of rotation (related to implant AP size) and ‘θ’ is the rotation angle (for small angles cos θ = zero). Since implant AP size was between 52 and 70 mm, the lateral translation of the trochlear groove and facets could generally be approximated to 0.5 mm per degree of external rotation. The effect on the posterior translation of the lateral facet was negligible, less than 0.1 mm per degree of external rotation.
Discussion
The most important finding of this study is that TKA implants are not always designed with anatomic trochlear parameters, and that some designs exhibit characteristics similar to those observed in trochlear dysplasia. The design of the femoral component is of great importance to restore normal knee kinematics, and the geometry of the trochlear groove is a major factor of PF complications after TKA [25]. An optimal design should feature an anatomic, non-dysplastic and asymmetric trochlear groove that is wider, more proximally extended and deeper compared to earlier-generation designs [23]. The inability of current arthroplasty designs to reproduce normal anatomy and motion has been attributed to the lack of information available on the complex anatomy of the normal trochlea compared to that of the TKA component [8, 28, 44].
Of the 14 implants measured, 11 had a sulcus angle over 144° in the ‘Merchant view’, and 13 had a sulcus angle over 143° in the ‘Brattström view’. In many implants, the sulcus angle exceeded these radiographic indicators of trochlear dysplasia by more than 10°. The sulcus angle is inversely proportional to the depth of the trochlear groove, which is important to engage the patella in the trochlea, especially in early flexion (0°–30°) [8, 10, 28]. The average sulcus angle for healthy knees is 138° in the ‘merchant view’ [29, 30, 32] and 142° in the ‘Brattström view’ [11]. A high sulcus angle indicates a shallow or dysplastic trochleae, observed in the majority of patients suffering from PF disorders [15]. On the other hand, a normal sulcus angle does not exclude the presence of trochlear dysplasia, since sulcus angle may be high proximally and decrease distally to normal values [19, 27, 39].
Of the 14 implants measured, 8 had a lateral facet less than 5 mm high through the entire range of early flexion (0°–30°) and 5 had a lateral facet less than 5 mm high beyond early flexion (30°–45°). The lateral facet is essential to align the patella within the trochlea in early flexion and to prevent lateral subluxation and tilt [2, 10]. In a radiographic study of 200 normal knees, Brattström reported the range of lateral facet height to be 4.2–6.5 mm (at 30° of flexion) [11]. In a more recent cadaver study of 33 femora, Shih et al. [39] reported the mean height of the lateral facet to be 6.6 ± 1.8 mm (at 0° of flexion). Kulkarni et al. [26] emphasized the need for the component to fit normal trochlear anatomy, with a steep lateral facet, regardless whether the patella is resurfaced or not. A reduced facet would predispose to lateral patellar dislocation, while an elevated facet could exacerbate tension in the lateral patellar retinaculum and potentially lead to excessive PF contact pressures and impingement [2]. This variation of the lateral facet height observed in the present study is consistent with other authors who noticed that the trochlear depth or the lateral facet height could be different among TKA implants, and that they are also affected by rotation of the femoral component [8, 34, 40, 44].
The trochlear groove was oriented laterally in 13 implants, and for the majority, it was in the range of 5°–8°. The trochlear groove was vertical in one implant, and the small angular error of 0.3° is an indicator of the accuracy of our measurement technique. The ML position of the trochlear groove varied at different flexion angles, thus increasing the TT-TG distance, which is considered as an “instability factor” for patellar dislocation [15]. To compare our results with the literature, it is important to note that: (1) we measured the groove orientation angles with reference to the sagittal plane, which is close to the femoral mechanical axis and (2) we measured the orientation only for the proximal groove (0°–45°) and we excluded the distal groove (45°–90°). There is general consensus that the trochlear groove is bilinear, with different orientations in its proximal and distal positions [7, 23, 24, 42], but there are debates on whether its orientation is lateral [45], parallel [16, 17], or medial [6, 18] to the femoral anatomic axis. Barink et al. found from a study on 100 cadaver femora that the trochlea was oriented medially by 4.2° ± 3.2° [6]. Varadarajan et al. reported from Magnetic Resonance Images of 24 knees that the trochlear groove is oriented laterally by 10.0° ± 4.6° in females and 4.5° ± 6.2° in males [42]. Considering these variations, it is no surprise that there is scarce information on how the anatomy of the normal trochlea is reproduced by the femoral component [7, 23, 24].
The femoral component is typically positioned in a few degrees of external rotation [31, 33]. Insall proposed 3° of rotation to create balanced flexion and extension gaps and to favour patellar tracking [9, 33]. The present study proved that external rotation of the femoral component by up to 6° resulted in noticeable lateral translation of the trochlear groove and facet, as well as negligible posterior translation to these points. A relationship between the rotation angle and the amounts of lateral and posterior translations that applied to all implants was recorded: every degree of external rotation leads to 0.5 mm of lateral translation and to 0.025 mm of posterior translation. Thus an external rotation of 3°, commonly applied in the clinical setting, results in 1.5 mm of lateral translation and 0.075 mm (negligible) posterior translation. This is also supported by Varadarajan et al. who described that femoral component rotation affects AP sulcus depth, sulcus location and trochlear groove orientation [44]. Femoral component position in the coronal plane also affects patella tracking, as it affects the mediolateral position of the trochlear groove as well as the trochlear groove angle [24] and that of the mediolateral groove position at 30° of flexion, which ranged from 47.3–55.5 %. Shih et al. measured the mediolateral position of the trochlear groove in 33 cadavers and found it to be 49.0 % ± 3.7 % of the femur width [39]. Additionally, Varadajan et al. [44] and Meijerink et al. [28] reported prosthetic trochlear groove positions, which were respectively 0.8–2.5 mm more medial than in the normal knee. This difference changed substantially from distal to proximal on the trochlea, thus emphasizing the need for implants with an anatomically positioned and proximally extended trochlear flange [44].
The strengths of this study were the large number of implants used that included both new and old TKA designs. The methodology was consistent; we designed a fixation jig to position and orient all specimens in an accurate and reproducible manner. The ‘scale factor’ was eliminated and therefore results do not depend on specimen size; we relied on non-dimensional measurements such as sulcus angle (degrees rather than mm) and trochlear groove position (% rather than mm along distance from the medial margin to the lateral margin). The main weaknesses of the study were the consideration of the femoral component and not the patellar nor tibial component, and the focus on static design features rather than dynamic implant performance.
Conclusion
The present study presented a detailed description of previously overlooked TKA implant parameters and design limitations and revealed that some femoral components exhibit characteristics of trochlear dysplasia. The clinical relevance of this descriptive study is that surgeons should be aware of such design limitations in order to improve their choice of implants for specific patients and to improve diagnosis and treatment of post-operative PF complications. In the light of these results, it seems appropriate to advise surgeons to avoid implanting a femoral component with a shallow trochlea in a patient with history of PF disorders and to adapt their surgical techniques and implant rotation as necessary to optimize PF tracking. Further work is needed to correlate the postoperative PF complications with the trochlear designs, but this needs to be done with much scrutiny because of their multi-factorial nature.
References
Aglietti P, Buzzi R, Gaudenzi A (1988) Patellofemoral functional results and complications with the posterior stabilized total condylar knee prosthesis. J Arthroplast 1:17–25
Amis AA (2007) Current concepts on anatomy and biomechanics of patellar stability. Sports Med Arthrosc 2:48–56
Anouchi YS, Whiteside LA, Kaiser AD, Milliano MT (1993) The effects of axial rotational alignment of the femoral component on knee stability and patellar tracking in total knee arthroplasty demonstrated on autopsy specimens. Clin Orthop Relat Res 287:170–177
Arendt E (2005) Anatomy and malalignment of the patellofemoral joint: its relation to patellofemoral arthrosis. Clin Orthop Relat Res 436:71–75
Balcarek P, Ammon J, Frosch S, Walde TA, Schuttrumpf JP, Ferlemann KG, Lill H, Sturmer KM, Frosch KH (2010) Magnetic resonance imaging characteristics of the medial patellofemoral ligament lesion in acute lateral patellar dislocations considering trochlear dysplasia, patella alta, and tibial tuberosity-trochlear groove distance. Arthroscopy 7:926–935
Barink M, van de Groes S, Verdonschot N, de Waal Malefijt M (2003) The trochlea is bilinear and oriented medially. Clin Orthop Relat Res 411:288–295
Barink M, Van de Groes S, Verdonschot N, De Waal Malefijt M (2006) The difference in trochlear orientation between the natural knee and current prosthetic knee designs; towards a truly physiological prosthetic groove orientation. J Biomech 9:1708–1715
Barink M, Meijerink H, Verdonschot N, van Kampen A, de Waal Malefijt M (2007) Asymmetrical total knee arthroplasty does not improve patella tracking: a study without patella resurfacing. Knee Surg Sports Traumatol Arthrosc 2:184–191
Berger RA, Rubash HE, Seel MJ, Thompson WH, Crossett LS (1993) Determining the rotational alignment of the femoral component in total knee arthroplasty using the epicondylar axis. Clin Orthop Relat Res 286:40–47
Bicos J, Fulkerson JP, Amis A (2007) Current concepts review: the medial patellofemoral ligament. Am J Sports Med 3:484–492
Brattstroem H (1964) Shape of the intercondylar groove normally and in recurrent dislocation of patella. A clinical and X-ray-anatomical investigation. Acta Orthop Scand Suppl 68:61–148
Davies AP, Costa ML, Shepstone L, Glasgow MM, Donell S (2000) The sulcus angle and malalignment of the extensor mechanism of the knee. J Bone Joint Surg Br 8:1162–1166
Dejour D, Le Coultre B (2007) Osteotomies in patello-femoral instabilities. Sports Med Arthrosc 1:39–46
Dejour D, Saggin P (2010) The sulcus deepening trochleoplasty-the Lyon’s procedure. Int Orthop 2:311–316
Dejour H, Walch G, Nove-Josserand L, Guier C (1994) Factors of patellar instability: an anatomic radiographic study. Knee Surg Sports Traumatol Arthrosc 1:19–26
Eckhoff DG, Burke BJ, Dwyer TF, Pring ME, Spitzer VM, VanGerwen DP (1996) The ranawat award. sulcus morphology of the distal femur. Clin Orthop Relat Res 331:23–28
Eckhoff DG, Montgomery WK, Stamm ER, Kilcoyne RF (1996) Location of the femoral sulcus in the osteoarthritic knee. J Arthroplast 2:163–165
Feinstein WK, Noble PC, Kamaric E, Tullos HS (1996) Anatomic alignment of the patellar groove. Clin Orthop Relat Res 331:64–73
Fucentese SF, Schottle PB, Pfirrmann CW, Romero J (2007) CT changes after trochleoplasty for symptomatic trochlear dysplasia. Knee Surg Sports Traumatol Arthrosc 2:168–174
Grelsamer RP (1997) Patellofemoral complications following total knee arthroplasty. J Arthroplast 2:216
Grelsamer RP (2001) The sulcus angle and malalignment of the extensor mechanism of the knee. J Bone Joint Surg Br 5:772–773
Healy WL, Wasilewski SA, Takei R, Oberlander M (1995) Patellofemoral complications following total knee arthroplasty. Correlation with implant design and patient risk factors. J Arthroplast 2:197–201
Indelli PF, Marcucci M, Cariello D, Poli P, Innocenti M (2012) Contemporary femoral designs in total knee arthroplasty: effects on the patello-femoral congruence. Int Orthop 6:1167–1173
Iranpour F, Merican AM, Dandachli W, Amis AA, Cobb JP (2010) The geometry of the trochlear groove. Clin Orthop Relat Res 3:782–788
Kulkarni SK, Freeman MA, Poal-Manresa JC, Asencio JI, Rodriguez JJ (2000) The patellofemoral joint in total knee arthroplasty: is the design of the trochlea the critical factor? J Arthroplast 4:424–429
Kulkarni SK, Freeman MA, Poal-Manresa JC, Asencio JI, Rodriguez JJ (2001) The patello-femoral joint in total knee arthroplasty: is the design of the trochlea the critical factor? Knee Surg Sports Traumatol Arthrosc 9 Suppl 1:S8–S12
Martinez S, Korobkin M, Fondren FB, Hedlund LW, Goldner JL (1983) Computed tomography of the normal patellofemoral joint. Invest Radiol 3:249–253
Meijerink HJ, Barink M, van Loon CJ, Schwering PJ, Donk RD, Verdonschot N, de Waal Malefijt MC (2007) The trochlea is medialized by total knee arthroplasty: an intraoperative assessment in 61 patients. Acta Orthop 1:123–127
Merchant AC, Mercer RL, Jacobsen RH, Cool CR (1974) Roentgenographic analysis of patellofemoral congruence. J Bone Joint Surg Am 7:1391–1396
Merchant AC (1997) Femoral sulcus angle measurements. Am J Orthop (Belle Mead NJ) 12(820):822
Merican AM, Ghosh KM, Iranpour F, Deehan DJ, Amis AA (2011) The effect of femoral component rotation on the kinematics of the tibiofemoral and patellofemoral joints after total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc 9:1479–1487
Mulligan ME, Jones ED Jr (1997) Femoral sulcus angle measurements. Am J Orthop (Belle Mead NJ) 8:541–543
Olcott CW, Scott RD (2000) Determining proper femoral component rotational alignment during total knee arthroplasty. Am J Knee Surg 3:166–168
Ostermeier S, Buhrmester O, Hurschler C, Stukenborg-Colsman C (2005) Dynamic in vitro measurement of patellar movement after total knee arthroplasty: an in vitro study. BMC Musculoskelet Disord 6:30
Panni AS, Cerciello S, Maffulli N, Di Cesare M, Servien E, Neyret P (2011) Patellar shape can be a predisposing factor in patellar instability. Knee Surg Sports Traumatol Arthrosc 4:663–670
Patel J, Ries MD, Bozic KJ (2008) Extensor mechanism complications after total knee arthroplasty. Instr Course Lect 57:283–294
Petersilge WJ, Oishi CS, Kaufman KR, Irby SE, Colwell CW Jr (1994) The effect of trochlear design on patellofemoral shear and compressive forces in total knee arthroplasty. Clin Orthop Relat Res 309:124–130
Petri M, von Falck C, Broese M, Liodakis E, Balcarek P, Niemeyer P, Hofmeister M, Krettek C, Voigt C, Haasper C, Zeichen J, Frosch KH, Lill H, Jagodzinski M (2012) Influence of rupture patterns of the medial patellofemoral ligament (MPFL) on the outcome after operative treatment of traumatic patellar dislocation. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-012-2037-z
Shih YF, Bull AM, Amis AA (2004) The cartilaginous and osseous geometry of the femoral trochlear groove. Knee Surg Sports Traumatol Arthrosc 4:300–306
Tanzer M, McLean CA, Laxer E, Casey J, Ahmed AM (2001) Effect of femoral component designs on the contact and tracking characteristics of the unresurfaced patella in total knee arthroplasty. Can J Surg 2:127–133
Tecklenburg K, Dejour D, Hoser C, Fink C (2006) Bony and cartilaginous anatomy of the patellofemoral joint. Knee Surg Sports Traumatol Arthrosc 3:235–240
Varadarajan KM, Gill TJ, Freiberg AA, Rubash HE, Li G (2009) Gender differences in trochlear groove orientation and rotational kinematics of human knees. J Orthop Res 7:871–878
Varadarajan KM, Freiberg AA, Gill TJ, Rubash HE, Li G (2010) Relationship between three-dimensional geometry of the trochlear groove and in vivo patellar tracking during weight-bearing knee flexion. J Biomech Eng 6:061008
Varadarajan KM, Rubash HE, Li G (2011) Are current total knee arthroplasty implants designed to restore normal trochlear groove anatomy? J Arthroplast 2:274–281
Yoshioka Y, Siu D, Cooke TD (1987) The anatomy and functional axes of the femur. J Bone Joint Surg Am 6:873–880
Conflict of interest
Author D.D. receives royalties from Tornier SA. The authors P.G.N. and M.S. have not included a conflict of interest disclosure statement.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dejour, D., Ntagiopoulos, P.G. & Saffarini, M. Evidence of trochlear dysplasia in femoral component designs. Knee Surg Sports Traumatol Arthrosc 22, 2599–2607 (2014). https://doi.org/10.1007/s00167-012-2268-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-012-2268-z