Abstract
Purpose
Regaining adequate strength of the quadriceps and hamstrings after anterior cruciate ligament (ACL) reconstruction is important for maximizing functional performance. However, the outcome of muscle strength after either BPTB or hamstrings autograft is unclear given the plethora of published studies that report post-operative muscle strength. The purpose of this study was to systematically compare the muscle strength of patients who have undergone ACL reconstruction using either Bone Patellar Tendon Bone (BPTB) or Hamstrings (HST) autograft.
Methods
The databases of MEDLINE, Cinahal and EMBASE were systematically searched for articles that report muscle strength outcome following ACL reconstruction. The quality of the studies was evaluated and a meta-analysis of the muscle strength outcomes was conducted on reported data.
Results
Fourteen studies were included in this systematic review: eight Randomized Control Studies (RCT) and six non-Randomized Control Studies (non-RCT). A meta-analysis was performed involving eight of the included studies (4 RCTs & 3 non-RCTs). At 60°/s and 180°/s, patients with BPTB graft showed a greater deficit in extensor muscle strength and lower deficit in flexor muscle strength compared with patients with HST.
Conclusion
This systematic review of Level III evidence showed that isokinetic muscle strength deficits following ACL reconstruction are associated with the location of the donor site. These deficits appear to be unresolved up to 2 years after ACL reconstruction.
Level of evidence
III.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rupture of the Anterior Cruciate Ligament (ACL) is one of the most common athletic injuries of the knee [14, 17, 61]. Lyman et al. recently estimated that the frequency of ACL reconstruction is increasing in the United States and that younger patients are at a higher risk for re-rupture of the ACL graft [41]. The consequences of an ACL rupture to the function of the involved limb are multifaceted and possibly include a decrease in joint stability, muscle weakness, meniscal damage, pain and, in the long term, development of osteoarthritis [29, 30, 46, 47, 50, 51, 54, 58, 62, 67, 73]. In an attempt to prevent these deficits in joint function, reconstruction of ACL has become one of the most common orthopaedic interventions. Although many different surgical techniques and an increasing number of graft types have been described in the literature, autograft reconstruction using Bone Patellar Tendon Bone (BPTB) or Hamstrings Tendon (HST) appears to be the most popular graft choices [4, 12, 23, 24, 26, 45, 53].
Despite a plethora of recently published comparative studies, the relative effectiveness of the different grafts used for the reconstruction of ACL remains unclear [4, 6, 7, 11, 12, 15, 23, 24, 27, 33]. Maximizing knee stability after ACL reconstruction is one of the most important criteria for the choice of graft. Post-operative stability allows the performance of rehabilitation protocols that aimed to restore normal function and thus safe and fast return to pre-injury activity level [48]. The superior post-operative stability afforded by BPTB autograft is likely to be related to enhanced healing from bone-to-bone attachments [3, 59, 72]. However, increased donor-site morbidity has been reported after harvesting BPTB autograft. Specifically, anterior knee pain, quadriceps weakness and worse results in functional tests along with an increased rate of patellar fractures have been observed [27, 40, 43, 44, 49]. Harvesting of the HST autograft may avoid some of these post-operative problems, but it is associated with hamstring muscle weakness and slower healing of the graft attachment site that may predispose patients to higher risk of re-rupture [2, 66, 70]. Thus, both of these graft choices are limited in the ability to restore knee function for people with ACL rupture, and there is consequently an ongoing debate concerning the superiority of one graft over the other. An important aspect of this debate is the outcome of lower limb muscle strength following either of these graft types.
Evaluation of muscle strength can be accomplished using functional tools (incorporating hop or twisting) or single-joint evaluation tools [2, 9, 52]. One of the most commonly used tools that is reliable in assessing single-joint muscle strength is isokinetic dynamometry [19, 56]. In comparison to other measures of strength, isokinetic dynamometry allows quantification of muscle strength deficit through the assessment of specific parameters such as work per unit, torque at specific joint angles and the widely used peak torque value [19, 22, 56, 67]. In the majority of studies that investigate muscle strength following ACL reconstruction, the strength of the operated limb is recorded as a deficit or gain in comparison to the contralateral healthy limb. Restoration of similar muscle strength between reconstructed and healthy knee is considered to be a critical factor for a safely return back to dynamic activities [48]. Thus, the restoration of muscle strength ratio between the operated and contralateral limbs for both the quadriceps and the hamstrings is crucial after an ACL reconstruction for a fast and uneventful return to pre-injury activities [48]. There is evidence that muscular recovery is closely related to pre-operative muscle strength, the time between injury and reconstruction and the pre- and post-surgery rehabilitation [8, 22, 55]. In addition, changes in the sensory system with ACL reconstruction, such as alterations in the somatosensory evoked potentials or the development of inconsistent postural synergies, may also influence muscle [18].
Although many authors have compared lower limb muscle strength in patients with ACL reconstruction after either BPTB or HST grafts, the plethora of information is difficult to interpret. Therefore, a systematic review of the literature is warranted to synthesize reported findings of the isokinetic muscle strength in studies comparing ACL reconstruction using either BPTB or HST autografts. Clarification of muscle strength recovery after ACL reconstruction using either graft type will enhance decision-making with regard to graft choice and rehabilitation.
Materials and methods
A thorough search of the databases MEDLINE, Cinahal and EMBASE for articles that compared muscle strength using isokinetic dynamometry between patients that had undergone ACL reconstruction with either BPTB or HST autograft was completed in September 2009. Full text articles published in English were searched using variations and combinations of the following terms: anterior cruciate ligament reconstruction, knee reconstruction, dynamometry, strength, weakness, and torque.
To be included in this review, articles must have:
-
compared two groups of patients that had undergone ACL reconstruction: one of the groups must have received BPTB autograft and one HST autograft;
-
evaluated knee flexor and extensor isokinetic muscle strength between 4 and 24 months after ACL reconstruction surgery;
-
been published in English language.
The following criteria were used to exclude articles from the systematic review. Studies were not included if:
-
studies did not include original data;
-
any participants had undergone revision of ACL;
-
participants had undergone multiple-ligament reconstruction.
Studies of different methodological design were included in this systematic review and are subject to different biases. Therefore, multiple tools were used to assess the quality of included studies. Randomized Control Trials (RCT) were assessed for quality using the PEDRO scale [20] which assesses the quality of studies based on 11 criteria. All other study designs were assessed using the tool described by Downs and Black [21]. The assessment of methodological quality was completed by 2 reviewers independently. Disagreement was resolved by discussion with a 3rd reviewer.
Extraction of data
Two independent reviewers read all of the articles in the final yield and systematically extracted pre-defined relevant data. Demographic details of participants were extracted from all articles in addition to the descriptive variables of isokinetic strength assessment at all speeds.
A meta-analysis was conducted on the findings of isokinetic evaluations at testing speeds of 60°/s and 180°/s, an average of 12 months after ACL reconstruction surgery. To be included in the meta-analysis, the mean and measures of variability must have been reported. Wherever the outcomes were not presented in a form suitable for direct inclusion in the meta-analysis, the corresponding authors were contacted by email in an attempt to obtain the data required for meta-analysis (numbers of participants, mean scores and SDs).
Statistical analysis
Muscle strength of the operated limb was extracted when reported either as a percentage of the uninvolved limb (i.e. Limb Symmetry Index) or as a percentage deficit of the uninvolved limb (100 × deficit of injured leg/deficit of uninjured leg). Mean differences and 95% confidence intervals were calculated from the extracted data. Random-effects models were used to pool data. Review Manager 5 (Version: 5.0.24) software was used for the calculation of effect sizes.
Results
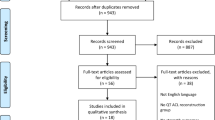
A total of 1,532 published studies were identified in the original search of databases. Following the application of inclusion and exclusion criteria, a final yield of 14 studies were included in this systematic review as presented in the flow chart (Appendix). Of the 14 included studies, eight were RCT and six non-Randomized Control Trials (non-RCT).
The study design and the characteristics of each study included in this review are presented in Table 1.
Quality assessment of the RCTs and the non- RCTs is presented in Tables 2 and 3. Inadequate randomization may allow the introduction of bias; however, only 3 of the 8 RCTs reported the process of patient randomization. Although blinding of the patient and surgeon is not always possible in this field of research, only 2 studies reported that assessors were blinded to the group allocation of patients.
Muscle strength outcomes
The muscle strength outcomes that were reported from all studies are presented in Tables 4 (for RCTs) and 5 (for non-RCTs).
Six studies [5, 7, 11, 12, 16, 69] found no significant difference between BPTB and HST for isokinetic muscle strength for knee extensors or knee flexors at follow-up times between 4 and 24 months after reconstruction.
Four studies [10, 26, 35, 42] found significant extensor muscle strength deficit in the operated limb in the BPTB group compared to the HST group at different follow-up times between 4 and 24 months. In addition, six studies [10, 13, 26, 31, 42, 71] found significant deficits of the flexor muscles in the operated limb in HST group compared to the BPTB group at different follow-up times between 4 and 24 months.
Sufficient data were provided in only four of the RCTs [11, 16, 26, 42] and three of the non-RCTs [13, 69, 71] to conduct a meta-analysis on findings 12 months after ACLR.
Figures 1 and 2 show forest plots that summarize quadriceps and hamstring strength for patients at a speed of 60°/s. There were 3 articles where muscle strength of the operated limb was reported as a percentage of the uninvolved limb. For patients with HST graft, quadriceps strength was an average of 9% stronger and hamstrings strength was 8% weaker than patients with BPTB graft. Two articles reported muscle strength of the operated limb as percentage deficit of the uninvolved limb. Similarly, patients with HST graft showed a 3% lower deficit in quadriceps strength and 9% greater deficit in hamstrings strength than patients with BPTB.
Figures 3 and 4 show forest plots that summarize quadriceps and hamstring strength for patients at a speed of 180°/s. There were 2 articles where muscle strength of the operated limb was reported as a percentage of the uninvolved limb. For patients with HST graft, quadriceps strength was an average of 7% stronger and hamstrings strength was 9% weaker than patients with BPTB graft. Two articles reported muscle strength of the operated limb as percentage deficit of the uninvolved limb. Similarly, patients with HST graft showed a 1% lower deficit in quadriceps strength and 20% greater deficit in hamstrings strength than patients with BPTB.
Discussion
The most important finding of the present study was the apparent trend for muscle weakness that is specific to the graft donor site following ACL reconstruction. The meta-analysis performed showed that extensor muscle strength deficit exists in ACL reconstructed knees using BPTB autograft and that flexor muscle strength deficit exists in ACL reconstructed knees using HST autografts, 12 months post-operatively.
Not all studies reported muscle weakness in one group of patients or the other. Six studies [5, 7, 11, 12, 16, 69] did not find significant differences in extensor or flexor muscle strength between BPTB and HST groups, at any testing speed (60°/s, 120°/s, 180°/s, 240°/s, 300°/s). In contrast, eight studies found differences between groups. Significant quadriceps muscle strength deficit in BPTB group was observed in four studies [10, 26, 35, 42] and six studies found significant hamstrings muscle deficits in HST group [10, 13, 26, 31, 42, 71]. All of the studies evaluated patients between 4 and 24 months after surgery and muscle weakness was found to persist throughout this period. These findings are in agreement with other reviews [19, 57] that have concluded that the graft site affects muscle strength.
There is an obvious trend for quadriceps deficit at BPTB group compared to HST group and a trend for hamstrings deficit in HST group compared to BPTB group at 12 months post-operative. The results of the meta-analysis showed that difference between BPTB and HST group for extensor muscle strength was nearly 10% at the speed of 60°/s and 180°/s and that for flexor muscle strength was 20% at 180°/s. It is clinically accepted that anything less than a 10% difference between limbs is considered inconsequential [39]. Although the difference in quadriceps strength between sides was not greater than 10%, the difference in hamstring strength exceeded this clinical limit. It is difficult to know what the implications for this asymmetry between limbs are, given that most research has focused on investigating asymmetrical quadriceps weakness. Further research is, therefore, needed to establish whether such a large hamstring weakness in the operated limb of patients with HST graft has any clinical relevance.
The apparent trend for muscle strength weakness related to the donor site may be explained by previous research. It seems that harvesting the patellar tendon autograft during the ACL reconstruction may alter the length–tension relationship of the extensor mechanism [32] and consequently contribute to extensor muscle strength deficit. It is also described that muscle function might be altered due to the attenuation of the gamma loop function caused by the initial ACL injury and that is not restored after the ACL reconstruction. The mechanoreceptors located within the ACL play an important role in enhancing the activity of gamma motor neurons (contributing, to a normal muscle function) [36, 38, 63]; however, this mechanism is not restored with ACL reconstruction, and may, therefore, also play a role in the extensor muscle weakness seen after harvesting the BPTB graft. Furthermore, knee pain and effusion have been documented up to 12 months following ACL reconstruction and could alter the neural control of the quadriceps [37, 65, 68].
Strength deficits in the knee flexor muscles may be more easily explained. There is evidence that tendon fibers can regenerate following harvesting of the hamstring tendon to become similar to healthy and non-harvested fibers [25, 28]. However, Hioki et al. [34] found an atrophy of hamstrings’ muscle fibers as well as hypertrophy of the semimembranosus and biceps muscles, after harvesting the hamstrings tendon. Moreover, they demonstrated that after harvesting the hamstrings tendon the semitendinosus muscle assumes different shapes and movements and that each pattern was related to different knee flexor strength. It is not clear how these changes in morphology affect muscle and knee function.
Regardless of the physiological explanations for muscle weakness, it is clear that restoration of muscle strength must focus on increasing muscle strength following ACL reconstruction to maximize functional outcomes. In particular, it appears that patients with different graft types may be susceptible to muscle strength that is specific to graft type. These findings suggest that rehabilitation that addresses muscle weakness specific to graft type may enhance strength outcomes after ACL reconstruction.
The findings of muscle weakness related to graft donor site were not consistent throughout all of the studies included in this review. There were some methodological differences between these studies that may explain this inconsistency. The method of randomization was not reported or was insufficient for the most of the RCTs. Only three [11, 26, 42] used a specific random allocation, which verifies that allocation was concealed. This allows for a bias that potentially could alter the findings of these studies. Although almost all RCTs assessed patients with the same activity level, three did not report the sex of the patients despite the plethora of information that gender influences outcome after ACL reconstruction. Therefore, the generalizability of the findings reported in these studies may be limited [1, 64]. Although blinding is one of the most important factors to limit bias in a RCT, no patients or therapist and only 2 studies reported that assessors were blinded to patient group allocation. Only in the trials of Aglietti et al. [5] and Maletis et al. [42], the assessors were blinded. Again, the potential for bias in the findings of those studies that did not blind assessors needs to be considered. The studies that were not RCTs were subjected to different biases. Because patients in these studies were not randomized to receive either a BPTB or HST graft, it is important that both groups be similar at baseline on factors that may confound muscle strength findings. However, 3 studies did not adequately describe that groups were similar on important demographic characteristics such as height and weight. These limitations need to be considered when interpreting the findings of this review. Future work that compares the muscle strength outcomes between patients with either BPTB or HST ACL reconstruction needs to consider these factors when designing future research.
There are some limitations that need to be considered when interpreting the findings of this review. The meta-analysis was limited to only half of the studies included in the review because of disparity in the parameters of isokinetic testing (e.g., the speed of testing, and the time since surgery). Nevertheless, studies that did not evaluate muscle strength according to the strict criteria were still included in the systematic review and contribute significantly to the information that details recovery of muscle strength following ACL reconstruction.
Conclusions
Although not all studies reported muscle strength differences between patients with either BPTB or HST graft ACL reconstruction, there was an obvious trend toward greater muscle weakness that was dependent on the graft donor site. Rehabilitation that is specific to this difference in muscle strength between graft types is needed.
Furthermore, more high quality studies need to be conducted assessing the muscle strength recovery after the reconstruction of the torn ACL, in order to reveal a potential superiority of a graft type over the other graft options.
References
Ageberg E, Forssblad M, Herbertsson P, Roos EM (2010) Sex differences in patient-reported outcomes after anterior cruciate ligament reconstruction: data from the Swedish knee ligament register. Am J Sports Med 38:1334–1342
Ageberg E, Roos HP, Silbernagel KG, Thomee R, Roos EM (2009) Knee extension and flexion muscle power after anterior cruciate ligament reconstruction with patellar tendon graft or hamstring tendons graft: a cross-sectional comparison 3 years post surgery. Knee Surg Sports Traumatol Arthrosc 17:162–169
Aglietti P, Buzzi R, Giron F, Simeone AJ, Zaccherotti G (1997) Arthroscopic-assisted anterior cruciate ligament reconstruction with the central third patellar tendon. A 5–8-year follow-up. Knee Surg Sports Traumatol Arthrosc 5:138–144
Aglietti P, Buzzi R, Zaccherotti G, De BP (1994) Patellar tendon versus doubled semitendinosus and gracilis tendons for anterior cruciate ligament reconstruction. Am J Sports Med 22:211–217
Aglietti P, Giron F, Buzzi R, Biddau F, Sasso F (2004) Anterior cruciate ligament reconstruction: bone-patellar tendon-bone compared with double semitendinosus and gracilis tendon grafts. A prospective, randomized clinical trial. J Bone Joint Surg Am 86-A:2143–2155
Anderson AF, Snyder RB, Lipscomb AB Jr (2001) Anterior cruciate ligament reconstruction. A prospective randomized study of three surgical methods. Am J Sports Med 29:272–279
Anderson JL, Lamb SE, Barker KL, Davies S, Dodd CA, Beard DJ (2002) Changes in muscle torque following anterior cruciate ligament reconstruction: a comparison between hamstrings and patella tendon graft procedures on 45 patients. Acta Orthop Scand 73:546–552
Andersson D, Samuelsson K, Karlsson J (2009) Treatment of anterior cruciate ligament injuries with special reference to surgical technique and rehabilitation: an assessment of randomized controlled trials. Arthroscopy 25:653–685
Augustsson J, Thomeé R, Karlsson J (2004) Ability of a new hop test to determine functional deficits after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 12:350–356
Aune AK, Holm I, Risberg MA, Jensen HK, Steen H (2001) Four-strand hamstring tendon autograft compared with patellar tendon-bone autograft for anterior cruciate ligament reconstruction. A randomized study with 2-year follow-up. Am J Sports Med 29:722–728
Beard DJ, Anderson JL, Davies S, Price AJ, Dodd CA (2001) Hamstrings vs. patella tendon for anterior cruciate ligament reconstruction: a randomised controlled trial. Knee 8:45–50
Beynnon BD, Johnson RJ, Fleming BC, Kannus P, Kaplan M, Samani J, Renstrom P (2002) Anterior cruciate ligament replacement: comparison of bone-patellar tendon-bone grafts with two-strand hamstring grafts. A prospective, randomized study. J Bone Joint Surg Am 84-A:1503–1513
Bizzini M, Gorelick M, Munzinger U, Drobny T (2006) Joint laxity and isokinetic thigh muscle strength characteristics after anterior cruciate ligament reconstruction: bone patellar tendon bone versus quadrupled hamstring autografts. Clin J Sport Med 16:4–9
Bollen SR, Scott BW (1996) Rupture of the anterior cruciate ligament—a quiet epidemic? Injury 27:407–409
Brand J Jr, Weiler A, Caborn DN, Brown CH Jr, Johnson DL (2000) Graft fixation in cruciate ligament reconstruction. Am J Sports Med 28:761–774
Carter TR, Edinger S (1999) Isokinetic evaluation of anterior cruciate ligament reconstruction: hamstring versus patellar tendon. Arthroscopy 15:169–172
Childs SG (2002) Pathogenesis of anterior cruciate ligament injury. Orthop Nurs 21:35–40
Courtney C, Rine RM, Kroll P (2005) Central somatosensory changes and altered muscle synergies in subjects with anterior cruciate ligament deficiency. Gait Posture 22:69–74
Dauty M, Tortellier L, Rochcongar P (2005) Isokinetic and anterior cruciate ligament reconstruction with hamstrings or patella tendon graft: analysis of literature. Int J Sports Med 26:599–606
de Morton NA (2009) The PEDro scale is a valid measure of the methodological quality of clinical trials: a demographic study. Aust J Physiother 55:129–133
Downs SH, Black N (1998) The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health 52:377–384
Eitzen I, Holm I, Risberg MA (2009) Preoperative quadriceps strength is a significant predictor of knee function 2 years after anterior cruciate ligament reconstruction. Br J Sports Med 43:371–376
Ejerhed L, Kartus J, Sernert N, Kohler K, Karlsson J (2003) Patellar tendon or semitendinosus tendon autografts for anterior cruciate ligament reconstruction? A prospective randomized study with a 2-year follow-up. Am J Sports Med 31:19–25
Eriksson K, Anderberg P, Hamberg P, Olerud P, Wredmark T (2001) There are differences in early morbidity after ACL reconstruction when comparing patellar tendon and semitendinosus tendon graft. A prospective randomized study of 107 patients. Scand J Med Sci Sports 11:170–177
Eriksson K, Kindblom LG, Hamberg P, Larsson H, Wredmark T (2001) The semitendinosus tendon regenerates after resection: a morphologic and MRI analysis in 6 patients after resection for anterior cruciate ligament reconstruction. Acta Orthop Scand 72:379–384
Feller JA, Webster KE (2003) A randomized comparison of patellar tendon and hamstring tendon anterior cruciate ligament reconstruction. Am J Sports Med 31:564–573
Feller JA, Webster KE, Gavin B (2001) Early post-operative morbidity following anterior cruciate ligament reconstruction: patellar tendon versus hamstring graft. Knee Surg Sports Traumatol Arthrosc 9:260–266
Ferretti A, Conteduca F, Morelli F, Masi V (2002) Regeneration of the semitendinosus tendon after its use in anterior cruciate ligament reconstruction: a histologic study of three cases. Am J Sports Med 30:204–207
Finsterbush A, Frankl U, Matan Y, Mann G (1990) Secondary damage to the knee after isolated injury of the anterior cruciate ligament. Am J Sports Med 18:475–479
Fischer-Rasmussen T, Jensen PE (2000) Proprioceptive sensitivity and performance in anterior cruciate ligament-deficient knee joints. Scand J Med Sci Sports 10:85–89
Gobbi A, Mahajan S, Zanazzo M, Tuy B (2003) Patellar tendon versus quadrupled bone-semitendinosus anterior cruciate ligament reconstruction: a prospective clinical investigation in athletes. Arthroscopy 19:592–601
Herzog W, Longino D, Clark A (2003) The role of muscles in joint adaptation and degeneration. Langenbecks Arch Surg 388:305–315
Hiemstra LA, Webber S, MacDonald PB, Kriellaars DJ (2000) Knee strength deficits after hamstring tendon and patellar tendon anterior cruciate ligament reconstruction. Med Sci Sports Exerc 32:1472–1479
Hioki S, Fukubayashi T, Ikeda K, Niitsu M, Ochiai N (2003) Effect of harvesting the hamstrings tendon for anterior cruciate ligament reconstruction on the morphology and movement of the hamstrings muscle: a novel MRI technique. Knee Surg Sports Traumatol Arthrosc 11:223–227
Jansson KA, Linko E, Sandelin J, Harilainen A (2003) A prospective randomized study of patellar versus hamstring tendon autografts for anterior cruciate ligament reconstruction. Am J Sports Med 31:12–18
Johansson H, Sjolander P, Sojka P (1991) Receptors in the knee joint ligaments and their role in the biomechanics of the joint. Crit Rev Biomed Eng 18:341–368
Kartus J, Movin T, Karlsson J (2001) Donor-site morbidity and anterior knee problems after anterior cruciate ligament reconstruction using autografts. Arthroscopy 17:971–980
Konishi Y, Fukubayashi T, Takeshita D (2002) Mechanism of quadriceps femoris muscle weakness in patients with anterior cruciate ligament reconstruction. Scand J Med Sci Sports 12:371–375
Kvist J (2004) Rehabilitation following anterior cruciate ligament injury: current recommendations for sports participation. Sports Med 34:269–280
Lautamies R, Harilainen A, Kettunen J, Sandelin J, Kujala UM (2008) Isokinetic quadriceps and hamstring muscle strength and knee function 5 years after anterior cruciate ligament reconstruction: comparison between bone-patellar tendon-bone and hamstring tendon autografts. Knee Surg Sports Traumatol Arthrosc 16:1009–1016
Lyman S, Koulouvaris P, Sherman S, Do H, Mandl LA, Marx RG (2009) Epidemiology of anterior cruciate ligament reconstruction: trends, readmissions, and subsequent knee surgery. J Bone Joint Surg Am 91:2321–2328
Maletis GB, Cameron SL, Tengan JJ, Burchette RJ (2007) A prospective randomized study of anterior cruciate ligament reconstruction: a comparison of patellar tendon and quadruple-strand semitendinosus/gracilis tendons fixed with bioabsorbable interference screws. Am J Sports Med 35:384–394
Marumoto JM, Mitsunaga MM, Richardson AB, Medoff RJ, Mayfield GW (1996) Late patellar tendon ruptures after removal of the central third for anterior cruciate ligament reconstruction. A report of two cases. Am J Sports Med 24:698–701
Mastrokalos DS, Springer J, Siebold R, Paessler HH (2005) Donor site morbidity and return to the preinjury activity level after anterior cruciate ligament reconstruction using ipsilateral and contralateral patellar tendon autograft: a retrospective, nonrandomized study. Am J Sports Med 33:85–93
Matsumoto A, Yoshiya S, Muratsu H, Yagi M, Iwasaki Y, Kurosaka M, Kuroda R (2006) A comparison of bone-patellar tendon-bone and bone-hamstring tendon-bone autografts for anterior cruciate ligament reconstruction. Am J Sports Med 34:213–219
McDaniel WJ Jr, Dameron TB Jr (1983) The untreated anterior cruciate ligament rupture. Clin Orthop Relat Res 172:158–163
Meunier A, Odensten M, Good L (2007) Long-term results after primary repair or non-surgical treatment of anterior cruciate ligament rupture: a randomized study with a 15-year follow-up. Scand J Med Sci Sports 17:230–237
Myer GD, Paterno MV, Ford KR, Quatman CE, Hewett TE (2006) Rehabilitation after anterior cruciate ligament reconstruction: criteria-based progression through the return-to-sport phase. J Orthop Sports Phys Ther 36:385–402
Natri A, Jarvinen M, Latvala K, Kannus P (1996) Isokinetic muscle performance after anterior cruciate ligament surgery. Long-term results and outcome predicting factors after primary surgery and late-phase reconstruction. Int J Sports Med 17:223–228
Nelson F, Billinghurst RC, Pidoux I, Reiner A, Langworthy M, McDermott M, Malogne T, Sitler DF, Kilambi NR, Lenczner E, Poole AR (2006) Early post-traumatic osteoarthritis-like changes in human articular cartilage following rupture of the anterior cruciate ligament. Osteoarthritis Cartilage 14:114–119
Noyes FR, Matthews DS, Mooar PA, Grood ES (1983) The symptomatic anterior cruciate-deficient knee. Part II: the results of rehabilitation, activity modification, and counseling on functional disability. J Bone Joint Surg Am 65:163–174
Noyes FR, Barber SD, Mangine RE (1991) Abnormal lower limb symmetry determined by function hop tests after anterior cruciate ligament rupture. Am J Sports Med 19:513–518
Otero AL, Hutcheson L (1993) A comparison of the doubled semitendinosus/gracilis and central third of the patellar tendon autografts in arthroscopic anterior cruciate ligament reconstruction. Arthroscopy 9:143–148
Papadonikolakis A, Cooper L, Stergiou N, Georgoulis AD, Soucacos PN (2003) Compensatory mechanisms in anterior cruciate ligament deficiency. Knee Surg Sports Traumatol Arthrosc 11:235–243
Petersen W, Laprell H (1999) Combined injuries of the medial collateral ligament and the anterior cruciate ligament. Early ACL reconstruction versus late ACL reconstruction. Arch Orthop Trauma Surg 119:258–262
Pua YH, Bryant AL, Steele JR, Newton RU, Wrigley TV (2008) Isokinetic dynamometry in anterior cruciate ligament injury and reconstruction. Ann Acad Med Singapore 37:330–340
Samuelsson K, Andersson D, Karlsson J (2009) Treatment of anterior cruciate ligament injuries with special reference to graft type and surgical technique: an assessment of randomized controlled trials. Arthroscopy 25:1139–1174
Shelbourne KD, Foulk DA (1995) Timing of surgery in acute anterior cruciate ligament tears on the return of quadriceps muscle strength after reconstruction using an autogenous patellar tendon graft. Am J Sports Med 23:686–689
Shelbourne KD, Gray T (1997) Anterior cruciate ligament reconstruction with autogenous patellar tendon graft followed by accelerated rehabilitation. A two- to nine-year followup. Am J Sports Med 25:786–795
Shelbourne KD, Nitz P (1990) Accelerated rehabilitation after anterior cruciate ligament reconstruction. Am J Sports Med 18:292–299
Smith BA, Livesay GA, Woo SL (1993) Biology and biomechanics of the anterior cruciate ligament. Clin Sports Med 12:637–670
Snyder-Mackler L, Fitzgerald GK, Bartolozzi AR III, Ciccotti MG (1997) The relationship between passive joint laxity and functional outcome after anterior cruciate ligament injury. Am J Sports Med 25:191–195
Sojka P, Sjolander P, Johansson H, Djupsjobacka M (1991) Influence from stretch-sensitive receptors in the collateral ligaments of the knee joint on the gamma-muscle-spindle systems of flexor and extensor muscles. Neurosci Res 11:55–62
Stijak L, Radonjic V, Nikolic V, Blagojevic Z, Aksic M, Filipovic B (2009) Correlation between the morphometric parameters of the anterior cruciate ligament and the intercondylar width: gender and age differences. Knee Surg Sports Traumatol Arthrosc 17:812–817
Torry MR, Decker MJ, Viola RW, O’Connor DD, Steadman JR (2000) Intra-articular knee joint effusion induces quadriceps avoidance gait patterns. Clin Biomech (Bristol, Avon) 15:147–159
Tow BP, Chang PC, Mitra AK, Tay BK, Wong MC (2005) Comparing 2-year outcomes of anterior cruciate ligament reconstruction using either patella-tendon or semitendinosus-tendon autografts: a non-randomised prospective study. J Orthop Surg (Hong Kong) 13:139–146
Tsepis E, Giakas G, Vagenas G, Georgoulis A (2004) Frequency content asymmetry of the isokinetic curve between ACL deficient and healthy knee. J Biomech 37:857–864
Van d V, Gill TJ, DeFrate LE, Papannagari R, Li G (2008) The effect of anterior cruciate ligament deficiency and reconstruction on the patellofemoral joint. Am J Sports Med 36:1150–1159
Webster KE, Wittwer JE, O’Brien J, Feller JA (2005) Gait patterns after anterior cruciate ligament reconstruction are related to graft type. Am J Sports Med 33:247–254
West RV, Harner CD (2005) Graft selection in anterior cruciate ligament reconstruction. J Am Acad Orthop Surg 13:197–207
Witvrouw E, Bellemans J, Verdonk R, Cambier D, Coorevits P, Almqvist F (2001) Patellar tendon vs. doubled semitendinosus and gracilis tendon for anterior cruciate ligament reconstruction. Int Orthop 25:308–311
Yunes M, Richmond JC, Engels EA, Pinczewski LA (2001) Patellar versus hamstring tendons in anterior cruciate ligament reconstruction: a meta-analysis. Arthroscopy 17:248–257
Zatterstrom R, Friden T, Lindstrand A, Moritz U (1994) The effect of physiotherapy on standing balance in chronic anterior cruciate ligament insufficiency. Am J Sports Med 22:531–536
Acknowledgments
The authors acknowledge the support from the Greek General Secretariat for Research and Technology and the European Union (Operative Program Competitiveness; AKMON) to the Orthopaedic Sports Medicine Center, and the Hellenic Association of Orthopaedic Surgery & Traumatology.
Author information
Authors and Affiliations
Corresponding author
Appendix
Rights and permissions
About this article
Cite this article
Xergia, S.A., McClelland, J.A., Kvist, J. et al. The influence of graft choice on isokinetic muscle strength 4–24 months after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 19, 768–780 (2011). https://doi.org/10.1007/s00167-010-1357-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-010-1357-0