Abstract
Dislocation of the knee is a relatively rare injury with modern arthroscopic techniques, operative reconstruction has become the standard of care. The primary aim of this study was to prospectively follow a large, consecutive series of patients with knee dislocation to document associated injuries, surgical treatment, knee function, and knee osteoarthritis (OA) at a minimum of 2 years follow-up. Hundred and twenty-two consecutive patients with a traumatic knee dislocation (Schenck II–IV) were treated at the Oslo University Hospital, Ulleval, between May 1996 and December 2004. Follow-up evaluation of 85 patients consisted of evaluation of knee joint laxity using the KT1000, the Lachman test, the pivot shift test, the reversed pivot shift, the posterior drawer test, the dial test, and the varus–valgus tests compared to the uninjured knee. Knee function was evaluated using the Lysholm score, the Tegner activity level score, the IKDC2000 score, and four single leg hop tests. Radiographic evaluation was performed using the Kellgren & Lawrence classification grade 0–4. Knee function at a minimum of 2 years after surgery disclosed a Lysholm score of a median of 83, a Tegner activity score of 5, and above 83% on all single leg hop tests compared to the uninjured side. Knee function was lower in the patients with a knee dislocation caused by high-energy trauma compared to low energy trauma. Eighty-seven percent had Kellgren & Lawrence grade 2 or higher for the injured knee compared to 35% for the uninjured knee.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Dislocation of the knee is a relatively rare injury; it constitutes 0.02–0.2% of orthopedic injuries [23, 44, 46]. In fact, before 1999, there were only 270 patients included in published studies [54]. However, the true incidence of this injury is most likely underestimated because a certain number of knee dislocations spontaneously reduce before presentation [49, 58, 60, 63]. Therefore, the definition of knee dislocation has been expanded to include injuries with at least two of the four major ligaments of the knee disrupted from a single traumatic episode [4, 51, 60]. Although dislocations of the knee with the posterior cruciate ligament (PCL) remaining intact have been reported [3, 7, 51], most dislocations involve disruption of both the anterior cruciate ligament (ACL) and PCL.
The first to publish on knee dislocations was Sir Astley Cooper in 1825 [6]. Since that time, the literature has been composed of mostly small case series with poorly controlled variables (Table 1). Because the incidence of this injury is so low, early literature led to controversy regarding the optimal treatment [1]. While some authors recommended cast immobilization [56], others advocated operative repair [38]. With modern arthroscopic techniques, operative reconstruction has become the standard of care [9, 10, 12, 16, 28, 45, 55, 62]. While some authors still recommend staged reconstruction, beginning with the PCL, and later the ACL if necessary [39, 63], today early simultaneous reconstruction of the ACL and PCL, and repair or reconstruction of the medial and lateral structures are recommended [9, 10, 12, 16, 28, 32, 33, 49, 59, 62].
The seriousness of knee dislocations has been well-documented [60]. The rate of life-threatening injuries involving the head, chest or abdomen has been reported to be 27%; the rate of associated fractures ranges from 50 to 60%; and the rate of multiple fractures 41% [58]. The rate of popliteal artery injury ranges between 7 and 48% in the literature [17, 24, 34, 53]. The incidence of injury to the peroneal nerve varies between 25 and 40% [35]. Finally, the rate of amputation following knee dislocation has been reported to be around 10% [54].
The primary aim of this study was to prospectively follow a consecutive series of patients who sustained a knee dislocation to document associated injuries, surgical treatment, knee function and knee osteoarthritis (OA) at a minimum 2 years follow-up. Furthermore, the aim was to examine difference in outcome between patients with knee dislocations from high versus low energy trauma. We hypothesized that patients with knee dislocation would have poor long-term outcome regarding knee function and severity of knee OA. Furthermore, we hypothesized that patients with high-energy trauma would have significantly poorer knee function compared to those with low-energy trauma.
Materials and methods
This is a prospective cohort study with a minimum of 2 years follow-up with the main outcome measures radiographic assessment of knee osteoarthritis (Kellgren & Lawrence) and knee function measured using knee performance tests and questionnaires.
A systematic literature search was carried out in PubMed database from 1985 to 2009. The search strategy for PubMed was done by including the following keywords “knee” AND “dislocation”, AND “surgical outcome”; limited to: human, clinical trials, reviews, and English language. This left 40 studies. Furthermore, patella dislocation, rehabilitation, unicompartmental and knee arthroplasty studies were excluded. One meta-analysis study [8] and one review study [46] that included a summary table of included subjects and outcomes were included. Other reviews were excluded. This left 17 papers (Table 1).
Subjects
As the only level I trauma center in the southeast part of Norway, Oslo University Hospital, Ulleval, receives all major multitrauma including multiple ligament knee injuries directly in the acute phase or through local hospitals. Hundred and twenty-two consecutive patients with a traumatic knee dislocation were treated at the Ullevaal University Hospital between May 1996 and December 2004. The inclusion criteria were injury to both the ACL and PCL according to the classification of Schenck et al. [48] (KD II–KD V) and an injury to the medial and/or lateral side. Patients needed to be skeletally mature and more than 18 years of age. The exclusion criteria were severe intraarticular fracture of the same knee and skeletal immaturity.
These patients were entered into a prospective database and have been followed since the time of surgery. Prior to start of the study, approval was obtained from the Regional Committee for Medical Research Ethics in Eastern Norway. All patients signed a written consent form on involvement in the study, and to participate in the post-operative testing.
At the time of admission, all patients had a thorough history and physical examination. Knee examination was documented, as well as any additional injuries. For the acute setting, the patients’ vascular status was monitored clinically, with serial examination by palpation of the dorsalis pedis and posterior tibial pulses. Additionally, warmth and color of the extremity was monitored. If asymmetry between injured and uninjured limbs was noted during the examination of pulses, skin color or skin temperature, further evaluation with angiography was performed. The status of the peroneal nerve was also documented. All patients underwent standard radiographs of the injured knee, as well as magnetic resonance imaging studies. In the acute patients prior to surgery, a hinged brace and CPM was used in the hospital for approximately 7 days.
Surgical management
Acutely injured patients underwent surgical reconstruction of their injured knee within 2 weeks after the injury, when not contraindicated by other injuries. Waiting for a week allowed time for the capsular structures to seal, in order to minimize chances of fluid extravasation during arthroscopy. Surgical management was based on the preoperative clinical examination, radiographic studies, examination under anesthesia, and arthroscopic findings.
After induction of anesthesia, the patient was positioned supine on the operating room table. Epidural anesthesia was used in addition to facilitate the postoperative pain management. A tourniquet was placed around the proximal thigh. A lateral post was positioned near the level of the tourniquet, and a bump was placed distally, so that when the foot was placed on it, the knee was flexed to ninety degrees.
An examination under anesthesia was performed. The ligamentous status of the injured leg with the uninjured leg was compared. Grade 1+ laxity was considered to be 3–5 mm side-to-side difference; grade 2+, 6–10 mm side-to-side difference; grade 3+, greater than 10 mm side-to-side difference. The ACL was evaluated with the knee at 30° (Lachman) and 90° (anterior drawer). The pivot shift and the reversed pivot shift were documented. The PCL was evaluated at 90° (posterior drawer). Varus and valgus laxity were evaluated at 0° and 30°. The posterolateral corner structures were evaluated with a reverse pivot shift exam, recurvatum and external rotation asymmetry through the dial test.
After a sterile preparation and draping, the diagnostic arthroscopy. The surgery was started without the use of the tourniquet. The status of the cartilage in all compartments were evaluated, and the meniscus in the medial and lateral compartment. The ACL and PCL were evaluated to confirm their injured status. Following completion of the diagnostic arthroscopy, necessary tendon harvests and meniscal repairs were performed. In the early phase of our procedure (1996–1998), Achilles tendon or quadriceps allografts were used for ACL and PCL reconstruction. After 1998, we used autograft tendons for ACL and PCL reconstruction. For the PCL reconstruction, hamstring autograft was used. If there was a medial sided injury, the gracilis and semtendinosus were harvested from the contralateral knee. If the medial side was stable, hamstring harvest was from the ipsilateral side. For the ACL reconstruction, bone–patellar tendon–bone (B–PT–B) was harvested from the ipsilateral side, but in the case of injuries to the extensor apparatus, B–PT–B graft from the contra lateral side was used. These grafts were then prepared on the back table by an assistant, while we proceeded with the surgery.
A standard 5.5 mm shaver to debride the femoral insertion of the ACL and PCL was used. If vision was difficult at this time, due to bleeding, the tourniquet was inflated. After debriding, the femoral notch the tibial footprint of the ACL and tibial portion of the PCL was debrided through a posteromedial portal. A 70° arthroscope was used, and the the shaver was placed into the posteromedial portal. From this portal, the posterior insertion of the PCL on the tibia was debrided, always having the shaver blade facing the tibial bone. Once the PCL was completely debrided, the capsule moved away from the tibia, allowing very good visualization of the posterior proximal tibia.
The tibial tunnel for the PCL was approximated with a Kirschner wire, with the use of an arthroscopic PCL guide. The entrance of the tunnel was placed as far distally as possible within the previously harvested site of the tibial bone plug. The Kirschner wire then entered the joint at the posterior aspect of the tibia, slightly lateral within the notch. The Kirschner wire was visible as it entered the joint. In the first 10 patients, a fluoroscope was used to verify the position of the K-wire. This wire was then over-drilled to a diameter 1 mm larger than the harvested tendon measured.
The tibial tunnel for the ACL was then approximated with a Kirschner wire, using an arthroscopic ACL guide. Since the PCL was not there for reference, the ACL footprint as well as the posterior edge of the anterior horn of the lateral meniscus was used as reference points. This tunnel was then over-drilled to the diameter of the tibial bone plug.
The femoral tunnel for the ACL was then approximated with a Kirschner wire, using an arthroscopic femoral guide, placed through the tibial tunnel. This was then over-drilled to the diameter of the femoral bone plug. The tunnel was placed abuting the posterior cortex, approximately at the 10 or 2 O’clock position.
Finally, the PCL femoral tunnel was approximated using a Kirschner wire, placed through the lateral portal. We placed this at about the 10.30 or 01.30 O’clock position, 5–6 mm deep to the cartilage, to minimize any possibility of necrosis of the subchondral bone. We then over-drilled the femoral tunnel to a diameter 1 mm larger than the PCL graft.
The autograft (hamstrings) was first passed for the PCL reconstruction, followed by the ACL (bone–patellar tendon–bone). For the PCL fixation, the hamstring graft was fixed in the femoral tunnel with the use of an Endobutton (Smith &Nephew®) when the tunnel was long enough. If the femoral tunnel was too short, an RCI screw (Smith &Nephew®) was used. For fixation of the ACL graft in the femoral tunnel, a standard interference screw (Soft Silk, Smith & Newphew®) was used. Fixation of the ACL and PCL grafts in their respective tibial tunnels was delayed until repair of any medial or lateral injuries was complete.
Injuries of the anterior and posterior cruciate ligament, posterolateral corner, and medial collateral ligaments
For combined injuries to ACL, PCL, and PLC treatment began with reconstruction of the ACL and PCL. Following fixation of the femoral side, attention was turned toward the lateral sided injuries. A 15 cm lateral curvilinear incision was made, beginning at Gerdy’s tubercle, and carried proximally centered over the lateral epicondyle. Dissection was carried sharply down to the fascia overlying the iliotibial band. Skin flaps were then elevated anteriorly and posteriorly. We then proceeded to identify the peroneal nerve, lying posterior to the biceps tendon, and protected it with a vessel loop. We then proceed with identifying and treating injured structures on the lateral side.
The surgical approach to anatomically repair and reconstruct the posterolateral knee has been previously described in detail [26]. The lateral collateral ligament (LCL) was best identified by making a 1 cm longitudinal incision over the biceps bursa located at the proximal part of the insertion site of the LCL. Once the LCL was identified at this level, it could be followed distally to the fibula, and proximally to its origin on the femur. The location of the injury was identified. Distal and proximal avulsions were repaired with the use of suture anchors. If the tendon could not be returned to its original site such as with mid substance tears, the repair was augmented with a portion of the distal biceps tendon or a reconstruction using allograft tendons.
The popliteus tendon was identified through the same vertical incision, which exposed the popliteal fossa and hiatus. Avulsions were repaired with a suture anchor. Injuries to the musculotendinous junction were not repaired. Injuries to the poplitefibular ligament (PFL) usually occurred with avulsions of the fibula and were fixed with suture anchors or wires.
Once the lateral sided injuries were repaired, the ACL and PCL were fixed into their tibial tunnels. First the PCL was fixed with the knee joint at approximately 90° of flexion and the normal tibia step off compared to the non-injured knee, in neutral rotation with an interference screw (RCI, Smith & Newphew®) and oftentimes with a double fixation tying the Ethibond sutures from the graft around a biocortical screw with a washer. The ACL was then fixed with an interference screw on the tibia with the knee close to full extension after 20 flexion-extension movements had been carried out.
Following femoral fixation of the ACL and PCL autografts, attention was turned to the medial collateral ligament. A 10 cm incision was made medially, centered over the medial epicondyle. Dissection was carried sharply down to the fascia overlying the medial side. Avulsions or ligament tears to the deep MCL were repaired using suture anchors (Mitek®) (tibia–meniscal and femoral–mensical ligaments) [27].
Distal avulsions of the superficial MCL were repaired with the use of a suture anchor. When the MCL could not be repaired, reconstruction was performed with the use of semitendinosis autograft. The semitendinosus was harvested with the use of a tendon stripper but left attached to its tibial insertion. The tendon was then brought up to the medial epicondyle and fixed to it with the use of a screw and washer and then sutured back to itself.
Following treatment of the medial sided injury, the ACL and PCL grafts were fixed into their tibial tunnels as previously described for the posterolateral side. In Schenck KD-IV injuries, the approach was similar with incisions lateral and medial.
Rehabilitation
After surgery, all patients underwent a standard rehabilitation protocol. The epidural was kept for 2–4 days after surgery. Immediately postoperatively, patients were placed into a knee brace, locked in full extension. During their hospital stay, a continuous passive motion device (CPM) was used twice a day at least 2 h between 0° and 60° of knee flexion from 1996 to 2001. After 2001, CPM was discontinued and passive motion was obtained twice daily with flexion exercises in prone position without a brace. The patients were kept in a brace for the initial 8 weeks, to allow the PCL to begin healing without being stretched. The goal was to reach 90° of knee flexion by 4 weeks postoperatively. During this time, patients were partial weight bearing with the assistance of crutches. Patients also performed isometric quadriceps exercises and straight leg raise exercises with the knee protected in a brace. At 8 weeks, the brace was discontinued and knee range of motion exercises in addition to active-assisted and full active range of motion exercises were continued. The patients progressed from partial weight bearing to full weight bearing and strengthening exercises for quadriceps, hamstrings and calf muscles were introduced. Patients were allowed to return to full activity when they achieved a minimum of 80% quadriceps muscle strength compared to the uninjured limb, and full knee range of motion; usually occurring between 9 and 12 months after surgery.
Follow-up evaluation
Follow-up evaluation consisted of clinical and radiographic evaluation; the self-administered questionnaires; the Lysholm score [30], the Tegner activity level score [57], as well as the International Knee Documentation Committee form (IKDC2000) [19]. Furthermore, knee performance tests included four single leg hop tests.
The Lysholm score was initially designed for use in patients following ACL reconstruction [30]. It has been extensively used in several clinical studies [31], however, recent studies have shown that it is most valid for patient with cartilage injuries [15], but a recently published study [5] have confirmed that Lysholm and Tegner scores could be used for ACL injured subjects and have shown moderate validity and reliability. The new version of the IKDC form, the IKDC2000 has been tested for validity, reliability and responsiveness for ACL injured subjects [20].
Patients underwent physical examination by one of the senior authors. Range of motion was measured with standard goniometric technique [13]. Knee joint laxity was evaluated using the Lachman test, the pivot shift test, the reversed pivot shift, the posterior drawer test, and varus–valgus test compared to the uninjured limb [52]. Laxity was categorized as negative, +, ++, or +++, and presence or absence of an endpoint was recorded. The PCL was examined using the posterior drawer test [52]. The posterolateral corner was evaluated with the reverse pivot shift, the dial test and varus stability at 0° and 30° [52].
Finally, tibial translation in the anterior and posterior direction was measured with the KT-1000 arthrometer (MEDmetric, San Diego, California) [42]. The KT-1000 arthrometer was used to record anterior displacement of the tibia relative to the femur at 134 N, and the manual maximum force. The difference in displacement between the two knees, expressed in millimeters, was used as an index of knee laxity. A side-to-side difference of 3 mm or more has been defined as abnormal [42].
Knee function was evaluated using four single leg hop tests (one leg hop, triple hop test, cross over hop test, and the 6 m timed hop test), as described by Noyes et al. [36] as performance-based measures of knee function. The one leg hop, the triple hop, and the cross over hop tests were recorded in centimeters, and the percent of the injured versus the uninjured was calculated for each test. Finally, the 6 m timed hop test was recorded as the time required to hop 6 m on one leg, and the percent of the uninjured versus the injured leg was calculated. All testing was done by a senior physical therapist.
Radiographic evaluation was performed by one radiologist, who was blinded to the injury pattern and surgery performed. The SynaFlexer™ system (Synarc, San Francisco, USA) for standardized positioning in a non-fluoroscopic fixed-flexion radiographic acquisition was used for the X-rays. A 10° caudal beam angulation was used to ensure alignment of the beam with the medial tibial plateau. A standardized degree of knee flexion (20°) and external foot rotation (5°) were achieved with the use of the SynaFlexer™ calibration and positioning frame [25].
The Kellgren & Lawrence classification system of osteoarthritis was used for evaluation of the radiographs [21]. The severity of radiographic changes was graded from zero to four. Grade one corresponded to doubtful narrowing of the joint space and possible osteophytic lipping and grade four was classified as large osteophytes, marked narrowing of the joint space, severe sclerosis and definite deformity of bone ends. Grade 2 has been used as cut-off for defining knee OA [21, 29]. This classification system has shown to have both high intra- and inter-rater reliability [21, 22].
Statistical analysis
Statistical analyses were performed using NCSS97 software (Number Crunches Statistical System, version 2.0.0.406, NCSS, Kaysville, UT, USA). Mean and standard deviation (SD) were calculated for parametrical data. Median, minimum and maximum values were given for the ordinal data from the Lysholm score, Tegner activity score and age. Frequency tables were used for categorical data. When normality distribution was presumed for parametrical data two sample Student t-tests were used for comparisons between groups (high and low energy trauma). When normality distribution was rejected, Mann–Whitney U tests were used for group comparisons. Mann–Whitney was also used for comparison between groups for the Lysholms score and Tegner activity score, and for comparisons between groups with unequal group sizes (Table 6). One-sample Student t tests were used for comparisons between baseline and follow-up data, but for changes in activity level from baseline to the follow-up Wilcoxons Signed-Rank Test for differences were used. Chi-square test was used for analyzing differences between gender for dropouts and study population. Alpha level was set at 0.05.
Results
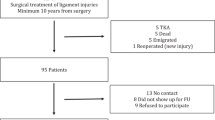
Eighty-five individuals returned for follow-up examination more than 2 years after surgery (Fig. 1). There were no significant differences between the dropouts (n = 36) and the study population, except that there were significant fewer women in the population of dropouts compared to the study population (p < 0.01, Table 2).
From May 1, 1996 to December 2004, 121 knee dislocations KD-II–KD-V were treated at Ullevaal University hospital. These patients were eligible for follow up at a minimum of 2 years after surgery (2.1–9.9 years). Eighty-five of the 121 patients (70%) were seen at follow-up at a mean of 5.3 ± 1.9 years. An overview of the results is presented in Table 3. Median age at injury was 33 years (12–82 years). There were 53% men and 47% females, 50 patients (60%) underwent surgery within 14 days after the injury and there were no differences in patient characteristics or knee function between those who performed surgery within 14 days and the chronic group. Mean weight at follow-up was 81 kg (±17 kg) with a body mass index (BMI) of 26 (±6). The injury occurred on the left side in 55% of the subjects, two patients had bilateral injury. Forty-three (51%) patients sustained a dislocation as a result of high energy trauma. Fifteen (18%) patients were involved in a motorcycle crash; 6 (7%) were involved in an automobile crash; 6 (7%) were pedestrians struck by a car; 40 (47%) were sport injuries, and 18 (21%) were categorized as others. The 40 (49%) patients who sustained a dislocation as a result of low energy trauma were mostly sport injuries (67%).
The injury pattern varied, and was determined by physical examination under anesthesia; the use of MRI, and arthroscopy. Four patients (6%) sustained injuries to the ACL and PCL (KD-II). Forty-two patients (49%) sustained injuries to the ACL, PCL and MCL (KD-III). Twenty-seven patients (32%) sustained injuries to the ACL, PCL and LCL (KD-III). Ten patients (12%) injured their ACL, PCL, MCL and LCL (KD-IV). The popliteus tendon was injured in 33 patients (40%).
Sixty-seven patients (79%) had no nerve injury. 18 patients (21%) sustained an injury to the peroneal nerve. One patient had complete paralysis of the nerve. Eight patients 9%) had paresis (resulting in a foot drop), but intact sensation. Five patients (6%) had paresis and decreased sensation. Four patients (5%,) had a decrease in sensation.
Five patients (6%) sustained injuries to the popliteal artery. All patients were diagnosed initially by asymmetric distal pulses on examination, and were confirmed with angiography. These patients underwent surgical bypass or embolectomy, temporary external fixation followed by ligament reconstruction 3–6 months later.
Five patients (6%) had an associated patella dislocation. Two patients (2%) had an associated patellar tendon rupture. Two patients (2%) sustained a patella fracture. Twelve patients (14%) sustained a fracture of the tibia, femur or fibula.
Twenty-two patients (29%) sustained cartilage injuries. There were 24 meniscal tears (31%). Thirteen patients (17%) injured their medial meniscus; 7 patients (9%) injured their lateral meniscus; 4 patients (5%) injured their lateral and medial meniscus.
The median Lysholm score at follow-up was 83 (15–100), the Tegner activity score was 5 (0–9), and the IKDC2000 score was 64 (±20) (Table 3). The single leg hop tests showed a mean percent difference between injured and uninjured knee of between 83 and 89 (Table 3). There was no significant difference between the early and late time of surgery for the Lysholm, the Tegner, or the IKDC2000 scores.
The clinical exam results are shown in Table 4. Forty-eight patients (57%) had a negative Lachman test, 28 patients (33%) had a 1+ Lachman test, 8 patients (9%) had a 2+ Lachman test, and 1 (1%) patient had a 3+ Lachman test. Seventy-six patients (91%) had a negative pivot shift test, 3 patients (4%) had a 1+ pivot shift test, and 5 patients (6%) had a 2+ pivot shift test. KT-1000 knee arthrometer using the maximum manual side to side test showed a mean difference of 2.7 mm (±3.7); a mean of 11.5 mm (±4.0) mm on injured side and 8.7 mm (±3.1) on uninjured side.
Table 5 shows the results for the high and low energy trauma groups. The low energy trauma group was significantly older and had significantly improved outcome for the triple hop tests (p = 0.02), but no other differences for the functional outcome measurements were disclosed.
Table 6 shows the patient characteristics and knee function for those patients with KD-II and III (n = 72) and those patients with KD-IV (n = 10). The IKDC2000 score for the patients with KD-IV was significantly lower compared to the patients with KD-II and III. But due to very few patients with KD-IV (n = 10), statistical comparison may have limited value.
Seventy-eight patients underwent radiographic examination using the Kellgren & Lawrence grading system. Table 7 shows that there was 87% with knee OA in the injured knee compared to 35% in the uninjured knee (Kellgren & Lawrence grade 2, 3, and 4).
Complications
Complications included post-operative arthrofibrosis in five patients (6%). This occurred primarily in patients with large medial side injury and surgery. The patients were treated with mobilization under anesthesia and in some cases arthroscopic debridement. Four patients developed an infection, three of which were superficial. One infection was deep, requiring irrigation, debridement and a gastrocnemius flap. Three patients developed a deep vein thrombosis (DVT).
Discussion
The most important findings of the present study was that the majority of knee dislocation patients obtained a good Lysholm score (median 83 points), performed activities on a regular basis (median Tegner score of 5), and performed hop tests above 83% of the uninjured knee. A Lysholm score of 83 is in the upper limit for classification of a fair result, and a Lysholm score above 84 points has been considered good knee function [30]. Based on these data, we can say that these patients reported satisfactory results despite the fact that the majority of the patients had radiographic signs of knee OA (87%). Furthermore, knee dislocation caused by high-energy trauma seemed to lead to less favorable results compared to low energy trauma. But the variation in the patients knee function was high within this cohort, with the highest variations within the high energy trauma group.
The systematic treatment of knee dislocations was started at Oslo University Hospital, Ulleval, in May 1996. Initially, the majority of patients were chronic, but as other hospitals in Norway became aware of the approach, an increasing number of knees have been seen in the acute phase. Currently we have 222 knee dislocations in our database. The patients are followed prospectively until they return to work and then after 5 and 10 years with a standard follow-up system [14]. Historically, knee dislocations have been an area of much controversy. The literature on knee dislocation is tainted by many studies with few subjects, no controls and high number of procedures (Table 1). This is in large part due to the relative rarity of this injury. Early controversy focused on whether to treat these injuries conservatively or surgically. The concern regarding operative treatment was that of post-operative stiffness. In 1972, Taylor et al. [56] included 43 patients with knee dislocation, with an emphasis on conservative treatment. Twenty-six of these patients were treated with plaster immobilization. Cast immobilization ranged from 3 to 12 weeks. The remaining patients were treated operatively. They rated the outcome good, fair or poor, based on stability, motion and pain. They concluded that conservative treatment yielded as good or better results than operative, and recommended conservative care for uncomplicated knee dislocation. They also recommended immobilization for no more than 6 weeks. With modern arthroscopic techniques, the ability to treat torn ligamentous structures with less surgical trauma was improved. Wong et al. [62] performed a retrospective study comparing operative treatment versus closed immobilization of knee dislocations. They evaluated 29 consecutive patients, 26 of whom were available for follow-up. 11 patients were treated with closed immobilization, while 15 were treated with surgery. The surgical group had varying methods of treatment, ranging from repair early in the study to reconstruction later in the study period. They reported better range of motion in the non-operative group (137° vs. 129°), however, this difference was not significant. They reported improved stability and improved overall function as measured by the IKDC2000 score in the operative cohort. While the number of patients in the study was small, they concluded that patients did better overall with operative reconstruction than conservative treatment.
Even with arthroscopic techniques, however, there continues to be concern about development of postoperative stiffness. This has led some to advocate staged reconstruction, with early PCL reconstruction and addressing the ACL later, if the patient has persistent instability. Ohkoshi et al. [39] reported on eight patients (9 knees) who underwent staged reconstruction. The PCL was reconstructed at a mean of 12.8 days after injury. The ACL was reconstructed an average of 3.8 months after the PCL. In all knees, they reported full extension with a mean flexion of 140°. The authors concluded that staged reconstruction allowed for use of autograft, with less surgical trauma to the knee, thus minimizing postoperative stiffness.
Yeh et al. reported on 23 patients with traumatic knee dislocation. They reconstructed the PCL at an average of 11 days after injury, but only debrided the ACL. Injured medial and lateral structures were repaired at the time of PCL reconstruction. They reported good results with this technique, with an average Lysholm score of 84.1 and range of motion from 1° to 129°. Three patients required arthroscopic lysis of adhesions at a mean of 3 months postoperatively. KT-1000 measurements at 20 lb of force showed a mean of 4.5 mm side-to-side difference. Despite this, they only performed delayed ACL reconstruction in one patient. They concluded that reconstruction of the PCL with only debridement of the ACL yielded good results.
Others, however, have advocated simultaneous reconstruction of the ACL and PCL after dislocation. Fanelli et al. [9] reported their experience with 35 arthroscopically assisted combined ACL/PCL reconstructions with follow-up ranging from 2 to 10 years. This study population included 19 acute and 16 chronic injuries. PCL injuries were reconstructed with either allograft (in 26 patients) or autograft (in 9 patients). ACL injuries were reconstructed with either allograft (in 18 patients) or autograft (17 patients). MCL injuries were treated with bracing or open reconstruction. Posterolateral instability was treated with primary repair or biceps femoris tendon transfer. They reported very good clinical results with a normal Lachman examination in 33 of 35 patients and a normal posterior drawer in 16 of 35 patients. Postoperative Lysholm, Tegner, and HSS knee ligament rating scale mean values were 91.2, 5.3 and 86.8, respectively. They did not find a statistically significant difference in these scores between the acute and chronic patients. They also did not find a statistically significant difference between patients treated with allograft, and those treated with autograft. Complications and associated injuries were not reported. Postoperative range of motion also was not recorded. The authors concluded that combined arthroscopic ACL/PCL reconstruction is a reliable surgical procedure. They felt that the ACL reconstruction portion of the procedure was more reliable than the PCL reconstruction. They did not feel that multi-ligament reconstruction needed to be staged.
Mariani et al. also looked at combined arthroscopically assisted ACL and PCL reconstruction in 15 patients using bone–patellar–bone autograft for the PCL and double hamstring tendons for the ACL reconstruction. 4 patients had acute injuries, while 11 patients had chronic injuries. Postoperative evaluation revealed full extension in all patients, and an average flexion of 118° (range 105°–135°). One patient required arthroscopic lysis of adhesions. They concluded that combined ACL/PCL reconstruction could be done without significant risk of postoperative stiffness.
More recently, Harner et al. [16] reported their experience with allograft reconstruction of the ACL and PCL. They reported on 31 patients with knee dislocation who were treated with a standard protocol. Avulsed ligaments and injuries to the medial collateral ligament were directly repaired, while complete tears to the cruciate and lateral collateral ligaments were reconstructed with the use of allograft tissue. At follow-up, the mean Lysholm score was 87, mean Knee Outcome Survey Activities of Daily Living was 89 and mean Sports Activity Scale was 89. Four patients required operative treatment for postoperative stiffness. Their overall results were very good. However, this series may not be representative of all dislocations, as it consisted of predominantly low-energy knee dislocations. Higher energy dislocations, open dislocations or those requiring vascular repair (14 total patients) were excluded, leading to perhaps better results than can be expected in all dislocations.
Our study is different from previous studies in the literature (Table 1). One important difference is that all dislocations seen from May 1996 to December 2004 were included in the study. We did not exclude patients with nerve or vascular injuries, high-energy injuries or open injuries. This, in part, explains the large variability in our results. Unfortunately, we were only able to see 85 of the 121 consecutive patients for follow-up. However, the comparison of the drop-outs with the follow-up group in Table 2 shows no systematic difference and thus a reduced chance of drop out bias. Another important difference is the number of patients included. The number of patients was much larger than any previous study; therefore, sub-analyses were performed based on high and low energy trauma, surgery in the acute and chronic phase, and KD-IV versus KD-II and III. In our patient population, the timing of surgery (acute versus chronic) did not have an effect on outcome. This is similar to that reported by Fanelli et al. [9], but different from Harner et al. [16] who found significant improved outcome for those who went through surgery in the acute phase. Furthermore, the presence or absence of a neurovascular injury did not have an effect on the outcome in our study. However, there were significant differences between those individuals who had gone through high versus low energy trauma and those with injury to all four ligaments (KD-IV) versus those categorized as KD-II and III. Patients who sustained a knee dislocation because of high-energy trauma had worse outcomes as measured by Lysholm and triple hop test. Patients who injured all four major ligaments had a worse outcome according to KOS ADL and the Lysholm scale (Table 6). Another area of controversy regarding knee dislocations is the appropriate evaluation for popliteal artery injury following knee dislocation. Some authors have recommended angiograms on all patients with suspected dislocations [16]. Recently, however, several authors have recommended selective angiography, based on serial physical examination or ankle-brachial indexes (ABI). Mills et al. reported their results of a prospective study evaluating 38 patients for popliteal artery injury after knee dislocation. They examined the pulse and obtained an ABI in all patients having a knee dislocation. 11 patients had an ABI lower than 0.90, all of which had an arterial injury requiring surgery. 27 patients had an ABI of 0.90 or higher. None of these patients had a vascular injury detectable by serial clinical examination or duplex ultrasonography. The authors concluded that the use of the ABI is a rapid, reliable tool for determining if an arterial injury is present, and that routine arteriography is not necessary.
Stannard et al. [53] also evaluated the need for routine arteriography. They performed serial pulse examinations on 130 consecutive patients who sustained acute multi-ligamentous knee injuries. Only patients with abnormal findings underwent arteriography. 10 patients had abnormal findings on physical examination. Nine of these patients had flow-limiting popliteal artery damage, requiring surgery. 17 patients had an arteriographic examination despite having normal physical findings. None of these patients had arterial damage requiring surgery. The authors concluded that selective arteriography based on serial physical examination is a safe and effective way to evaluate for arterial injury following knee dislocation.
We follow a similar protocol at our institution. Patients undergo serial physical examination looking for symmetry in the posterior tibialis and dorsalis pedis pulses, color and warmth. Since 2005 we have added the ankle brachial index (ABI). If the ABI is <0.8 an angiogram is carried out. In our series, five patients underwent arteriography based on changes in physical exam. All five were found to have arterial damage significant enough to warrant surgical intervention. No vascular injuries were missed with physical examination. Our conclusions mirror those in the recent literature. Selective arteriography based on serial physical examination is a safe and prudent way to evaluate for arterial injury following knee dislocation.
Eighteen patients (19%) sustained an injury to their peroneal nerve. This is similar to other rates published in the literature. All patients who had complete paresis of the nerve on admission to the hospital did not regain useful motor or sensory function, whereas those with partial injuries regained function. Similar to the results reported by Niall et al., injury to the peroneal nerve was seen most commonly with combined injury to the ACL, PCL and posterolateral corner.
There were significant differences in the radiographic results between the non-injured and the injured knee. Based on the Kellgren & Lawrence analytic system in weight-bearing radiographs, 87% percent had grade II–IV compared to 36% on the non-injured side. This is similar to the long-term results presented by Werrier et al. in 1998, who showed that dislocation was associated with a risk of post-traumatic osteoarthritis of 50% [61].
Conclusion
Dislocation of the knee is a serious injury that requires diligent care from the time of admission. Selective angiography based on serial physical examination is a safe way to evaluate vascular injury following knee dislocation. Good clinical results were obtained with simultaneous reconstruction of the ACL and PCL using autograft, and repair of medial and lateral injured structures. Injuries resulting from high-energy trauma, and those involving all four major ligaments resulted in worse outcomes compared to those with low-energy trauma and those involving two or three ligaments. A significant number of patients had knee OA based on Kellgren & Lawrence grade 2 or higher (87%).
References
Almekinders LC, Logan TC (1992) Results following treatment of traumatic dislocations of the knee joint. Clin Orthop Relat Res 20:3–207
Bin SI, Nam TS (2007) Early results of high-flex total knee arthroplasty: comparison study at 1 year after surgery. Knee Surg Sports Traumatol Arthrosc 15:350–355
Bratt HD, Newman AP (1993) Complete dislocation of the knee without disruption of both cruciate ligaments. J Trauma 34:383–389
Brautigan B, Johnson DL (2000) The epidemiology of knee dislocations. Clin Sports Med 19:387–397
Briggs KK, Lysholm J, Tegner Y, Rodkey WG, Kocher MS, Steadman JR (2009) The reliability, validity, and responsiveness of the Lysholm Score and Tegner Activity Scale for anterior cruciate ligament injuries of the knee: 25 years later. Am J Sports Med 37:898–901
Cooper A (1825) A treatise on dislocations and fractures of the joints, American edn. 1. Wells and Lilly, Boston, pp 142–157
Cooper DE, Speer KP, Wickiewicz TL, Warren RF (1992) Complete knee dislocation without posterior cruciate ligament disruption. A report of four cases and review of the literature. Clin Orthop Relat Res 22:8–233
Dedmond BT, Almekinders LC (2001) Operative versus nonoperative treatment of knee dislocations: a meta-analysis. Am J Knee Surg 14:33–38
Fanelli GC, Edson CJ (2002) Arthroscopically assisted combined anterior and posterior cruciate ligament reconstruction in the multiple ligament injured knee: 2- to 10-year follow-up. Arthroscopy 18:703–714
Fanelli GC, Giannotti BF, Edson CJ (1996) Arthroscopically assisted combined anterior and posterior cruciate ligament reconstruction. Arthroscopy 12:5–14
Frassica FJ, Sim FH, Staeheli JW, Pairolero PC (1991) Dislocation of the knee. Clin Orthop Relat Res 263:200–205
Gaski IA, Martinussen BT, Engebretsen L, Johansen S, Ludvigsen TC (2004) Knee luxation—follow-up after surgery. Tidsskr Nor Laegeforen 124:1078–1080
Gogia PP, Braatz JH, Rose SJ, Norton BJ (1987) Reliability and validity of goniometric measurements at the knee. Phys Ther 67:192–195
Granan LP, Bahr R, Steindal K, Furnes O, Engebretsen L (2008) Development of a national cruciate ligament surgery registry: the Norwegian National Knee Ligament Registry. Am J Sports Med 36:308–315
Hambly K, Griva K (2008) IKDC or KOOS? Which measures symptoms and disabilities most important to postoperative articular cartilage repair patients? Am J Sports Med 36:1695–1704
Harner CD, Waltrip RL, Bennett CH, Francis KA, Cole B, Irrgang JJ (2004) Surgical management of knee dislocations. J Bone Joint Surg Am 86-A:262–273
Hollis JD, Daley BJ (2005) 10-year review of knee dislocations: is arteriography always necessary? J Trauma 59:672–675
Ibrahim SA, Ahmad FH, Salah M, Al Misfer AR, Ghaffer SA, Khirat S (2008) Surgical management of traumatic knee dislocation. Arthroscopy 24:178–187
Irrgang JJ, Anderson AF, Boland AL, Harner CD, Kurosaka M, Neyret P, Richmond JC, Shelborne KD (2001) Development and validation of the international knee documentation committee subjective knee form. Am J Sports Med 29:600–613
Irrgang JJ, Ho H, Harner CD, Fu FH (1998) Use of the International Knee Documentation Committee guidelines to assess outcome following anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 6:107–114
Kellgren JHL, Lawrence JS (1957) Radiological assessment of osteo-arthrosis. Ann Rheum Dis 16:494–502
Kessler S, Guenther KP, Puhl W (1998) Scoring prevalence and severity in gonarthritis: the suitability of the Kellgren & Lawrence scale. Clin Rheumatol 17:205–209
Klimkiewicz JJ, Petrie RS, Harner CD (2000) Surgical treatment of combined injury to anterior cruciate ligament, posterior cruciate ligament, and medial structures. Clin Sports Med 19:479–492 vii
Klineberg EO, Crites BM, Flinn WR, Archibald JD, Moorman CT III (2004) The role of arteriography in assessing popliteal artery injury in knee dislocations. J Trauma 56:786–790
Kothari M, Guermazi A, von IG, Miaux Y, Sieffert M, Block JE, Stevens R, Peterfy CG (2004) Fixed-flexion radiography of the knee provides reproducible joint space width measurements in osteoarthritis. Eur Radiol 14:1568–1573
LaPrade RF (2005) Anatomic reconstruction of the posterolateral aspect of the knee. J Knee Surg 18:167–171
LaPrade RF, Engebretsen AH, Ly TV, Johansen S, Wentorf FA, Engebretsen L (2007) The anatomy of the medial part of the knee. J Bone Joint Surg Am 89:2000–2010
Liow RY, McNicholas MJ, Keating JF, Nutton RW (2003) Ligament repair and reconstruction in traumatic dislocation of the knee. J Bone Joint Surg Br 85:845–851
Lohmander LS, Englund PM, Dahl LL, Roos EM (2007) The long-term consequence of anterior cruciate ligament and meniscus injuries: osteoarthritis. Am J Sports Med 35:1756–1769
Lysholm J, Gillquist J (1982) Evaluation of knee ligament surgery results with special emphasis on use of a scoring scale. Am J Sports Med 10:150–154
Lysholm J, Tegner Y (2007) Knee injury rating scales. Acta Orthop 78:445–453
Mariani PP, Margheritini F, Camillieri G (2001) One-stage arthroscopically assisted anterior and posterior cruciate ligament reconstruction. Arthroscopy 17:700–707
Mariani PP, Santoriello P, Iannone S, Condello V, Adriani E (1999) Comparison of surgical treatments for knee dislocation. Am J Knee Surg 12:214–221
Mills WJ, Barei DP, McNair P (2004) The value of the ankle-brachial index for diagnosing arterial injury after knee dislocation: a prospective study. J Trauma 56:1261–1265
Niall DM, Nutton RW, Keating JF (2005) Palsy of the common peroneal nerve after traumatic dislocation of the knee. J Bone Joint Surg Br 87:664–667
Noyes FR, Barber SD, Mangine RE (1991) Abnormal lower limb symmetry determined by function hop tests after anterior cruciate ligament rupture. Am J Sports Med 19:513–518
Noyes FR, Barber-Westin SD (1997) Reconstruction of the anterior and posterior cruciate ligaments after knee dislocation. Use of early protected postoperative motion to decrease arthrofibrosis. Am J Sports Med 25:769–778
O’Donoghue DH (1955) An analysis of end results of surgical treatment of major injuries to the ligaments of the knee. J Bone Joint Surg Am 37-A:1–13
Ohkoshi Y, Nagasaki S, Shibata N, Yamamoto K, Hashimoto T, Yamane S (2002) Two-stage reconstruction with autografts for knee dislocations. Clin Orthop Relat Res 16:9–175
Owens BD, Neault M, Benson E, Busconi BD (2007) Primary repair of knee dislocations: results in 25 patients (28 knees) at a mean follow-up of four years. J Orthop Trauma 21:92–96
Patterson BM, Agel J, Swiontkowski MF, Mackenzie EJ, Bosse MJ (2007) Knee dislocations with vascular injury: outcomes in the Lower Extremity Assessment Project (LEAP) Study. J Trauma 63:855–858
Pugh L, Mascarenhas R, Arneja S, Chin PY, Leith JM (2009) Current concepts in instrumented knee-laxity testing. Am J Sports Med 37:199–210
Richter M, Bosch U, Wippermann B, Hofmann A, Krettek C (2002) Comparison of surgical repair or reconstruction of the cruciate ligaments versus nonsurgical treatment in patients with traumatic knee dislocations. Am J Sports Med 30:718–727
Rihn JA, Groff YJ, Harner CD, Cha PS (2004) The acutely dislocated knee: evaluation and management. J Am Acad Orthop Surg 12:334–346
Rios A, Villa A, Fahandezh H, de JC, Vaquero J (2003) Results after treatment of traumatic knee dislocations: a report of 26 cases. J Trauma 55:489–494
Robertson A, Nutton RW, Keating JF (2006) Dislocation of the knee. J Bone Joint Surg Br 88:706–711
Roman PD, Hopson CN, Zenni EJ Jr (1987) Traumatic dislocation of the knee: a report of 30 cases and literature review. Orthop Rev 16:917–924
Schenck RC Jr (1994) The dislocated knee. Instr Course Lect 43:127–136
Shapiro MS, Freedman EL (1995) Allograft reconstruction of the anterior and posterior cruciate ligaments after traumatic knee dislocation. Am J Sports Med 23:580–587
Shelbourne KD, Haro MS, Gray T (2007) Knee dislocation with lateral side injury: results of an en masse surgical repair technique of the lateral side. Am J Sports Med 35:1105–1116
Shelbourne KD, Pritchard J, Rettig AC, McCarroll JR, Vanmeter CD (1992) Knee dislocations with intact PCL. Orthop Rev 21:607 1
Solomon DH, Simel DL, Bates DW, Katz JN, Schaffer JL (2001) The rational clinical examination. Does this patient have a torn meniscus or ligament of the knee? Value of the physical examination. JAMA 286:1610–1620
Stannard JP, Sheils TM, Lopez-Ben RR, McGwin G Jr, Robinson JT, Volgas DA (2004) Vascular injuries in knee dislocations: the role of physical examination in determining the need for arteriography. J Bone Joint Surg Am 86-A:910–915
Stayner LR, Coen MJ (2000) Historic perspectives of treatment algorithms in knee dislocation. Clin Sports Med 19:399–413
Talbot M, Berry G, Fernandes J, Ranger P (2004) Knee dislocations: experience at the Hopital du Sacre-Coeur de Montreal. Can J Surg 47:20–24
Taylor AR, Arden GP, Rainey HA (1972) Traumatic dislocation of the knee. A report of forty-three cases with special reference to conservative treatment. J Bone Joint Surg Br 54:96–102
Tegner Y, Lysholm J (1985) Rating systems in the evaluation of knee ligament injuries. Clin Orthop Relat Res 4:3–49
Wascher DC (2000) High-velocity knee dislocation with vascular injury. Treatment principles. Clin Sports Med 19:457–477
Wascher DC, Becker JR, Dexter JG, Blevins FT (1999) Reconstruction of the anterior and posterior cruciate ligaments after knee dislocation. Results using fresh-frozen nonirradiated allografts. Am J Sports Med 27:189–196
Wascher DC, Dvirnak PC, DeCoster TA (1997) Knee dislocation: initial assessment and implications for treatment. J Orthop Trauma 11:525–529
Werier J, Keating JF, Meek RN (1998) Complete dislocation of the knee—the long-term results of ligamentous reconstruction. The Knee 5:255–260
Wong CH, Tan JL, Chang HC, Khin LW, Low CO (2004) Knee dislocations-a retrospective study comparing operative versus closed immobilization treatment outcomes. Knee Surg Sports Traumatol Arthrosc 12:540–544
Yeh WL, Tu YK, Su JY, Hsu RW (1999) Knee dislocation: treatment of high-velocity knee dislocation. J Trauma 46:693–701
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Engebretsen, L., Risberg, M.A., Robertson, B. et al. Outcome after knee dislocations: a 2–9 years follow-up of 85 consecutive patients. Knee Surg Sports Traumatol Arthrosc 17, 1013–1026 (2009). https://doi.org/10.1007/s00167-009-0869-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-009-0869-y




