Abstract
Tree swallows nesting on mercury-contaminated sites along the South River in Virginia, USA were monitored for reproductive success. The bodies of nestlings found deceased in their nest boxes were collected, along with blood and feather samples from the adult parents and surviving siblings. We also measured hatching and fledging success of the clutches and the annual recapture rate of adults. We found that the body feathers of deceased nestlings contained significantly higher concentrations of mercury (12.89 ± 8.42 μg/g, n = 15) than those of nestlings that survived to fledge (7.41 ± 4.79 μg/g, n = 15). However, mothers of more successful clutches (>75 % hatching) did not differ in mercury concentrations from females with less successful clutches (<50 % hatching). Additionally, adult females breeding for the first time that returned to breed the following year did not differ in blood mercury from females of the same age that bred once but never returned. Our results suggest that mercury had its greatest effect on these songbirds during the nestling stage, whereas for embryos or first-time breeding females, other factors likely played larger roles in mortality.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Mercury (Hg) is an anthropogenic contaminant released by fossil fuel combustion, metal and cement production, waste incineration, and gold mining. Hg accumulates in the sediments of rivers and other bodies of water where it can remain for centuries (Carter 1977). A textile manufacturing plant in Waynesboro, VA used Hg as a catalyst before 1950, and the ecological ramifications continue today (Carter 1977; Jackson et al. 2011b). Once thought to be a problem primarily for aquatic animals, Hg has been shown to move into terrestrial food webs where it can be accumulated by insectivorous songbirds feeding away from the water (Cristol et al. 2008; Keller et al. 2014).
The effects of Hg on birds, including mortality, has been well documented (Heinz and Hoffman 2003; Heinz et al. 2009). Hg, specifically the most bioavailable form: methyl Hg, has been linked to a wide range of reproductive, behavioral, and physiological effects observed in songbirds both in the field and lab (e.g. Hallinger et al. 2010; Henry et al. 2014; Moore et al. 2014). The magnitude and scope of these effects suggest that the mortality of songbirds would be affected in some way by tissue Hg concentration. Among radio-tracked great white herons (Ardea herodias occidentalis), those dying of multiple or chronic diseases had higher liver Hg levels than those experiencing accidental deaths (Spalding et al. 1994), suggesting that Hg may have contributed to the diseases. However, adult tree swallows (Tachycineta bicolor) living on a contaminated site in Virginia exhibited only a 1 % reduction in annual survivorship compared to those on reference sites (Hallinger et al. 2011). Previous studies on adult tree swallows demonstrated a lack of effect on survival from PCB contamination. Females that returned to “moderately contaminated” sites had significantly higher concentrations of PCBs than females that did not return (Custer et al. 2007). Other studies of Hg and survivorship (e.g. loon, Gavia immer: Mitro et al. 2008; ibis, Eudocimus albus: Frederick and Jayasena 2011) have also failed to find a large effect of Hg on mortality, despite evidence for specific health-related effects in these species. However, the low statistical power of these survivorship studies, and logistical difficulties of studying migratory birds over long time spans, make it challenging to demonstrate a relationship between Hg exposure and survivorship in the field. Previous studies have focused primarily on the effects of Hg on survival in adult birds, rather than across the different life stages.
The objective of this study was to examine the relationship between mortality and tissue Hg concentration of tree swallows living on a contaminated site, across discrete parts of the reproductive cycle. We focused on three life stages: embryos from laying through hatching, nestlings from hatching to fledging, and young females who bred once and then did or did not return for their second breeding season. Based on previous literature, we predicted that individual nestling Hg tissue concentration would be negatively related to likelihood of nestling survival and that the blood Hg concentrations of female parents would not be related to hatchability. We also predicted that young adult females returning to breed on our study site would have higher Hg concentrations than females that did not return to breed, as a result of dietary differences.
Methods and Materials
The tree swallow is a cavity-nesting migratory songbird that breeds throughout much of North America. Tree swallows eat insects of both aquatic and terrestrial origin (Mengelkoch et al. 2004; Brasso and Cristol 2008). They also exhibit high levels of nesting site fidelity between years (Winkler et al. 2004) and are hardy to nest intrusion by researchers. For these reasons and others, tree swallows are a frequent study species in ecotoxicology studies (McCarty and Secord 1999; Custer et al. 2007).
The South River in Virginia, USA, was contaminated with Hg from 1929 to 1950 (Carter 1977). In 2005, nest boxes were placed at 19 contaminated sites along the river, and 17 reference sites in the same watershed. Cristol et al. (2008) found significantly higher levels of blood and feather Hg in nearly all songbird species breeding on these contaminated sites. The present experiment took place on the same contaminated sites as Cristol et al. (2008), and a map of the study area can be found in Fig. 1 of that paper.
During the breeding seasons of 2005–2008, the nest boxes along the South River were visited 2–3 times/week. Adult swallows were captured in their boxes either by hand, or using a trap door (Friedman et al. 2008) and sex was determined by external morphology. Young breeding females, i.e., birds in their first year of life, were identified by their variable but distinctive brown back plumage. After applying a unique numbered metal leg band, a blood sample (~75 μL) was taken from the brachial vein following the methods of Brasso and Cristol (2008). Dates of laying, hatching and fledging were recorded for every box once breeding commenced, and a small number of back feathers were taken from each nestling upon banding, 3–4 days before fledging. When a nestling was found dead during the nestling period, or during the nest check after fledging had occurred, the body was removed and frozen at −20°C. These nestlings were classified as “deceased.” Severely decomposed nestlings were discarded and not included in this study.
In June 2014, we removed the frozen nestling tissues from the freezer and sorted them, based on their origin, into two groups: deceased or fledging. For the deceased nestlings we possessed the entire carcass including feathers. For the fledging nestlings we possessed only the feathers that had been sampled a few days before they fledged. Deceased and fledging nestlings did not come from the same nests. Samples in the deceased group (n = 21) had to come from birds that met the following criteria: found dead in a nest box located within 50 m of the contaminated river; nestling old enough to have grown feathers; and at least one brood mate had survived to fledge (i.e., the entire clutch was not a failure). From the frozen nestlings we removed 10–15 back feathers for total Hg analysis.
Samples in the second group, fledging nestlings (n = 72), came from birds that met the following criteria: also hatched on a contaminated site within 50 m of the river, but from a nest box in which the entire brood fledged. Therefore, we classified as fledging only those nestlings from nests in which all hatched nestlings had fledged, making no assumptions as to how long after fledging that individual fledglings survived. Nestlings classified as deceased were from nests that were only partly successful, fledging at least one young. While all of these tissue samples were at least 6 years old, Appelquist et al. (1984) reported that freezing at −20°C results in very little degradation of Hg (~10 %) in feathers and both groups of feathers were stored under the same conditions in the present study. Furthermore, Varian-Ramos et al. (2011) have shown that frozen avian blood samples from these same freezers exhibited a drying-related average increase in Hg concentration of only 6 % after up to 4 years. To determine whether feather Hg correlated with other tissues, livers were dissected from the deceased nestlings, placed in a freeze-dryer for 24 h, and then homogenized by crushing with a small spatula before Hg analysis.
In addition to comparing feather Hg levels of fledging and deceased nestlings, we also used blood total Hg concentrations measured at the time of sampling (2005–2008) to compare the Hg exposure of: (1) more-successful and less-successful clutches; and (2) returning and missing adult female birds. In comparison one, we defined “more-successful” clutches as having a hatching success of over 75 % and “less-successful” as those having a hatch success of below 50 %. These were arbitrary values set to separate the two populations of clutches while maintaining sample size. Hatching success was calculated by dividing number of eggs hatched by number of eggs laid and multiplying by 100. In comparison two, we defined “returning” as females that bred in their first summer of life on our study site and then returned for a second summer. “Missing” birds were defined as birds that bred in their first summer of life, but failed to return the next year. Blood samples were collected from both “missing” and “returning” birds in their first year on our study site to determine if Hg exposure affected later fate. Blood Hg values from some of these groups of birds have been reported in previous publications (Cristol et al. 2008; Brasso et al. 2010; Hallinger and Cristol 2011) but never have the Hg concentrations of surviving/successful birds been compared to those of deceased/less-successful birds.
Feathers from both the fledging and deceased nestling groups were analyzed for total Hg at the College of William and Mary with cold vapor atomic absorbance spectroscopy using a direct Hg analyzer (DMA-80 Milestone, Inc., Shelton, CT, USA). The method that was used followed that in Cristol et al. 2008. Briefly, the feather samples were washed and then dried in a desiccation chamber before being weighed in sample boats. Deionized water was added to the boats after weighing to keep the feathers from falling out. The same Hg analyzing methods were used for blood samples. Ninety-five percent of the Hg present in avian tissue is in the organic form; therefore, measurements of total Hg are indicative of methylated Hg concentration in the tissue (Evers et al. 2005). All Hg concentrations in feathers are presented as wet weight (μg/g).
Nestling feathers from the fledging group were sequentially analyzed by clutch. For example, if four birds hatched and successfully fledged from a particular nest box, the feather samples from all four birds were run back-to-back on the Hg analyzer and a single mean value was generated from these four samples. To further reduce potential for analytic bias, fledging and deceased brood samples were alternated within each run. For quality control, every 20 samples analyzed included two samples each of two standard reference materials (DORM-4, DOLT-4, National Research Council, Canada), a method blank, a sample blank, and a pair of duplicate feather samples (i.e., two aliquots from a coarsely chopped zebra finch primary feather). For all duplicate samples analyzed (n = 9), we had a relative percent difference of 3.27 %. The average percent recovery of Hg from the standard reference materials run over the course of our study was 95.3 % ± 3.00 % for DORM-4 (n = 8) and 99.3 % ± 4.90 % for DOLT-4 (n = 10).
Statistical analysis was performed using the computing environment R (R Core Team 2014). We performed three analyses on the data gathered in 2005–2008. First, to relate nestling mortality and Hg level, average feather Hg concentration of nestlings in the deceased group was compared to that of the fledging group. Fledging birds were combined by brood with a single average value generated for each brood. In the rare event that more than one dead nestling was found in a box, the dead nestlings’ Hg levels were combined to generate one average value. In this way, our sample size of fledging nestlings (n = 72) was reduced to n = 15 broods, and deceased nestlings (n = 21) to n = 15 broods. To put these feather Hg levels into the context of a more well-studied tissue, we removed one deceased nestling’s liver from each brood (n = 15) and analyzed them for Hg content. We then correlated liver Hg (μg/g dry weight) and feather Hg (μg/g wet weight).
Next, for eggs that hatched we had no Hg concentration, because this was a non-invasive study. Therefore, we used average blood Hg level of the female parents as a proxy for the average egg Hg in their clutches (see supporting data for this assumption from the same population in Fig. 1 of Brasso et al. 2010). To relate maternal blood Hg level with egg hatching success we compared the less-successful clutches (<50 % hatched) to the more-successful clutches (>75 % hatched).
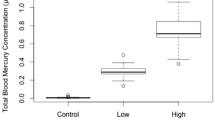
Finally, to compare adult survivorship and Hg level, we used our two groups of young females that had bred in their first year of life: returning and missing. We presumed that missing birds had died due to their expected high level of nest-site fidelity (Winkler et al. 2004), but we acknowledge that they may have survived and bred elsewhere. For both groups, a sample taken in their first year of breeding/life was analyzed for Hg concentration as a predictor of later survival. We used a non-parametric Mann–Whitney Test to compare the two groups in each of the three subsets of data (e.g., returning versus missing birds). Box-and-whisker plots in figures show the distribution of Hg concentrations within each group. Lines denote the median and upper and lower quartiles (50 % of the data points lie between these lines). Dashed lines indicate the minima and maxima of the data sets. Outliers, or values greater than 1.5x the interquartile range, are marked with hollow circles.
Results and Discussion
Nestling tree swallows found dead in their nest boxes at this site had higher levels of Hg (1.7x) in their feathers than nestlings in nests where all survived to fledge. The average feather Hg concentration of deceased nestlings was 12.9 ± 8.42 μg/g (n = 15), compared to 7.41 ± 4.79 μg/g (n = 15) for fledging nestlings (p = 0.004; Fig. 1a). One brood in the deceased group had an uncommonly high feather Hg concentration of 38.2 μg/g (shown as outlier in Fig. 1a). When this brood was removed from analysis, the deceased and fledging groups remained statistically different (p = 0.007). We plotted liver and feather Hg in the deceased group and obtained an r value of 0.93 and a p value of <0.001 from a Spearman correlation test, indicating a strong relationship between feather and liver Hg (Fig. 1b). This relationship between Hg and fledgling mortality is consistent with the finding that adult female tree swallows at this site experienced an approximate 20 % reduction in fledging success of their nestlings, relative to birds on reference sites (Brasso and Cristol 2008). The oft-cited minimum effects level for Hg in feathers is 5–40 μg/g (Cristol et al. 2012). Our results suggest that for nestling tree swallows the lower end of this scale is close to being correct, as nests in which all nestlings fledged averaged just above the suggested lowest effects level (7 μg/g), whereas those that died before fledging were somewhat higher (13 μg/g). Even at a young age, body feather Hg concentration is a consequence of dietary as opposed to egg-derived Hg (Becker et al. 1994). Nestlings that survived to fledge with 7 μg/g in their feathers may have exhibited effects of Hg that were not severe enough to cause immediate mortality. In another songbird species, the Carolina wren (Thryothorus ludovicianus), effects on reproduction were detected at adult feather Hg concentrations as low as 3 μg/g (Jackson et al. 2011a). While adult and nestling feather concentrations cannot be interpreted in the same way, our findings are consistent with previous literature in that relatively low feather Hg concentrations were associated with detectable effects on fitness.
Females with less-successful clutches (≤50 % hatching) had an average blood Hg concentration of 2.15 ± 1.48 μg/g (n = 22), whereas females with more-successful clutches (≥75 % hatching) had an average Hg concentration of 2.60 ± 1.85 μg/g (n = 37). Because maternal blood Hg is highly correlated with average clutch Hg at this site (Brasso et al. 2010), we can reject the hypothesis that high Hg concentration (measured in parent, assumed for egg) was related to low hatching success (p = 0.40; Fig. 2). These data are consistent with some other studies on hatching success and Hg contamination; for example unhatched seabird eggs had lower Hg levels than eggs sampled at random from the colony (Becker et al. 1994). The apparent lack of detectable effect of Hg on hatching success could be due to hormesis, a phenomenon wherein low contaminant exposure levels have a stimulating, rather than an inhibiting, effect. Heinz et al. (2010, 2011) demonstrated hormesis in response to Hg contamination, possibly a result of Hg having mild antibiotic properties.
Adult females that returned to the breeding site after their first year of breeding had previously been measured with average total blood Hg concentrations averaging 3.23 ± 1.94 μg/g (wet weight; n = 43) during their first year. Birds that did not return after breeding in their first year of life had a lower average blood Hg concentration of 2.55 ± 1.32 μg/g (n = 50) but the two means did not differ statistically (p = 0.14; Fig. 3). Our findings on young adults and eggs are in accordance with previous studies at this site in which neither hatching success nor survivorship differed significantly between contaminated and reference sites (Hallinger et al. 2011; Hallinger and Cristol 2011). The possible, albeit statistically nonsignificant, relationship between young adult return rate and Hg concentration could be attributed to the acquisition of large amounts or quality of food the previous year, with concomitant increased Hg concentrations. Swallows at the South River that acquired more aquatic food in their first breeding season might be more likely to return the next year, which would explain the trend toward higher Hg concentrations in returning birds. We however did not measure body condition or other health variables.
Few studies have examined the Hg levels of birds that died versus those that survived. Nestlings that were found dead in their nest boxes had more Hg in their feathers than nestlings that survived to fledging. The deceased nestlings had feather Hg levels above 5 μg/g (except for one with 4.92 μg/g), a level often cited as the lower range of effects for birds (Burger and Gochfeld 1997). Excluding one clutch with an average liver Hg of 11.7 μg/g, the deceased nestlings in our study had an average liver Hg of 2.66 μg/g, well below that suggested by Spalding et al. (1994) to be associated with mortality by chronic disease. Hg can affect songbirds at multiple stages in their life history, and it is important to identify the concentrations associated with fitness effects or mortality at each stage. We have shown, on a contaminated site, that maternal blood levels did not differ between females with >75 % hatching success and those with <50 % (both ~2–3 μg/g). Nor did blood levels differ between young females that went on to return after their first year of breeding and those that did not (both ~2–3 μg/g). But among nestlings, there was an association between living or dying and feather Hg concentrations (7 vs. 13 μg/g). This finding identifies the nestling stage as the most vulnerable life stage for this songbird, with a threshold of 5–10 μg/g feather Hg being associated with nestling mortality.
References
Appelquist H, Asbirk S, Drabaek I (1984) Mercury monitoring: mercury stability in bird feathers. Mar Pollut Bull 15:22–24. doi:10.1016/0025-326X(84)90419-3
Becker PH, Henning D, Furness RW (1994) Differences in mercury contamination and elimination during feather development in gull and tern broods. Arch Environ Contam Toxicol 27:162–167. doi:10.1007/BF00214258
Brasso RL, Cristol DA (2008) Effects of mercury exposure on the reproductive success of tree swallows (Tachycineta bicolor). Ecotoxicology 17(2):133–141. doi:10.1007/s10646-007-0163-z
Brasso RL, Abdel Latif MK, Cristol DA (2010) Relationship between laying sequence and mercury concentration in tree swallow eggs. Environ Toxicol Chem 29:1155–1159. doi:10.1002/etc.144
Burger J, Gochfeld M (1997) Risk, mercury levels, and birds: relating adverse laboratory effects to field biomonitoring. Environ Res 75:160–172. doi:10.1006/enrs.1997.3778
Carter LJ (1977) Chemical plants leave unexpected legacy for two Virginia rivers. Science 198:1015–1020. doi:10.1126/science.198.4321.1015
Cristol DA, Brasso RL, Condon AM et al (2008) The movement of aquatic mercury through terrestrial food webs. Science 320:335. doi:10.1126/science.1154082
Cristol DA, Mojica EK, Varian-Ramos CW, Watts BD (2012) Molted feathers indicate low mercury in bald eagles of the Chesapeake Bay, USA. Ecol Indic 18:20–24. doi:10.1016/j.ecolind.2011.10.007
Custer CM, Custer TW, Hines JE et al (2007) Adult tree swallow (Tachycineta bicolor) survival on the polychlorinated biphenyl-contaminated Housatonic River, Massachusetts, USA. Environ Toxicol Chem 26:1056–1065. doi:10.1897/06-337r.1
Evers DC, Burgess NM, Champoux L et al (2005) Patterns and interpretation of mercury exposure in freshwater avian communities in northeastern North America. Ecotoxicology 14:193–221. doi:10.1007/s10646-004-6269-7
Frederick P, Jayasena N (2011) Altered pairing behaviour and reproductive success in white ibises exposed to environmentally relevant concentrations of methylmercury. Proc Biol Sci 278:1851–1857. doi:10.1098/rspb.2010.2189
Friedman SL, Brasso RL, Condon AM (2008) An improved, simple nest-box trap. J Field Ornithol 79:99–101. doi:10.1111/j.1557-9263.2008.00150.x
Hallinger KK, Cristol DA (2011) The role of weather in mediating the effect of mercury exposure on reproductive success in tree swallows. Ecotoxicology 20:1368–1377. doi:10.1007/s10646-011-0694-1
Hallinger KK, Zabransky DJ, Kazmer KA, Cristol DA (2010) Birdsong differs between mercury-polluted and reference sites. Auk 127:156–161. doi:10.1525/auk.2009.09058
Hallinger KK, Cornell KL, Brasso RL, Cristol DA (2011) Mercury exposure and survival in free-living tree swallows (Tachycineta bicolor). Ecotoxicology 20:39–46. doi:10.1007/s10646-010-0554-4
Heinz GH, Hoffman DJ (2003) Embryotoxic thresholds of mercury: estimates from individual mallard eggs. Arch Environ Contam Toxicol 44:257–264. doi:10.1007/s00244-002-2021-6
Heinz GH, Hoffman DJ, Klimstra JD et al (2009) Species differences in the sensitivity of avian embryos to methylmercury. Arch Environ Contam Toxicol 56:129–138. doi:10.1007/s00244-008-9160-3
Heinz GH, Hoffman DJ, Klimstra JD, Stebbins KR (2010) Enhanced reproduction in mallards fed a low level of methylmercury: an apparent case of hormesis. Environ Toxicol Chem 29:650–653. doi:10.1002/etc.64
Heinz GH, Hoffman DJ, Klimstra JD et al (2011) Teratogenic effects of injected methylmercury on avian embryos. Environ Toxicol Chem 30:1593–1598. doi:10.1002/etc.530
Henry KA, Cristol DA, Varian-Ramos CW, Bradley EL (2014) Oxidative stress in songbirds exposed to dietary methylmercury. Ecotoxicology 24:520–526. doi:10.1007/s10646-014-1400-x
Jackson AK, Evers DC, Etterson MA et al (2011a) Mercury exposure affects the reproductive success of a free-living terrestrial songbird, the Carolina wren (Thryothorus ludovicianus). Auk 128:759–769. doi:10.1525/auk.2011.11106
Jackson AK, Evers DC, Folsom SB et al (2011b) Mercury exposure in terrestrial birds far downstream of an historical point source. Environ Pollut 159:3302–3308. doi:10.1016/j.envpol.2011.08.046
Keller RH, Xie L, Buchwalter DB et al (2014) Mercury bioaccumulation in Southern Appalachian birds, assessed through feather concentrations. Ecotoxicology 23:304–316. doi:10.1007/s10646-013-1174-6
McCarty JP, Secord AL (1999) Reproductive ecology of tree swallows (Tachycineta bicolor) with high levels of polychlorinated biphenyl contamination. Environ Toxicol Chem 18:1433. doi:10.1897/1551-5028
Mengelkoch JM, Niemi GJ, Regal RR (2004) Diet of the nestling tree swallow. Condor 106:423–429. doi:10.2307/1370653
Mitro MG, Evers DC, Meyer MW, Piper WH (2008) Common loon survival rates and mercury in New England and Wisconsin. J Wildl Manag 72:665–673. doi:10.2193/2006-551
Moore CS, Cristol DA, Maddux SL et al (2014) Lifelong exposure to methylmercury disrupts stress-induced corticosterone response in zebra finches (Taeniopygia guttata). Environ Toxicol Chem 33:1072–1076. doi:10.1002/etc.2521
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Spalding MG, Bjork RD, Powell GVN, Sundlof SF (1994) Mercury and cause of death in great white herons. J Wildl Manag 58:735–739. doi:10.2307/3809688
Varian-Ramos CW, Condon AM, Hallinger KK et al (2011) Stability of mercury concentrations in frozen avian blood samples. Bull Environ Contam Toxicol 86:159–162. doi:10.1007/s00128-010-0164-0
Winkler DW, Wrege PH, Allen PE et al (2004) Breeding dispersal and philopatry in the tree swallow. Condor 106:768–776. doi:10.1650/7634
Acknowledgments
Research was completed with oversight from the South River Science Team, which is a collaboration of state and federal agencies, academic institutions, and environmental interests. Thank you to all of the students who collected deceased nestlings from 2005 to 2009. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution at which the studies were conducted.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Taylor, C.E., Cristol, D.A. Tissue Mercury Concentrations and Survival of Tree Swallow Embryos, Nestlings and Young Adult Females on a Contaminated Site. Bull Environ Contam Toxicol 95, 459–464 (2015). https://doi.org/10.1007/s00128-015-1643-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-015-1643-0