Abstract
In the present investigation, chromium (VI) induced toxicity on metabolic activity and translocations of nutrients in radish were evaluated under controlled glass house conditions. Chromium was found to induce toxicity and significantly affect plant growth and metabolic activity. Excess of chromium (0.4 mM) caused a decrease in the concentration of iron in leaves (from 134.3 to 71.9 μg g−1 dw) and significant translocation of sulphur, phosphorus and zinc. Translocation of manganese, copper and boron were less affected from root to stem. After 15 days of Cr exposure, maximum accumulation of Cr was found in roots (327.6 μg g−1 dw) followed by stems (186.8 μg g−1 dw) and leaves (116.7 μg g−1 dw) at 0.4 mM Cr concentration. Therefore, Cr may affect negatively not only production, but also the nutritive quality of the radish; likewise, higher Cr content may cause health hazards for humans.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Worldwide, metal contamination has increased in the biosphere as a result of anthropogenic activities. Industrial effluents contain a high concentration of chromium (Cr) with other metals that create a serious problem by polluting the lotic and lentic water bodies, wetlands and agricultural fields in adjoining areas of industrial complexes (Akinola and Ekiyoya 2006; Tiwari et al. 2011). Cr is a transition metal that occurs predominantly in two valence states: hexavelant chromium (VI) and trivalent chromium (III), which display quite different chemical properties (Zou et al. 2009). The global use of Cr in industrial applications and combustion of fuels has led to human occupational exposure and its increased introduction into the environment (Caglieri et al. 2006). It is easily absorbed from the soil and atmosphere by plants where Cr accumulation in plant organs can show toxic effects (Parmar and Chanda 2005). Cr due to its structural similarity with some essential elements can affect mineral nutrition of plants in a complex way. Contamination of agricultural fields with trace metals including Cr has become an important environmental concern throughout the world due to potential adverse effects on human and animal health.
In many parts of India, farmers use industrial discharged wastewater to irrigate their agricultural fields due to the scarcity of available freshwater. Therefore, the present study was undertaken to investigate the visible phytotoxicity symptoms, changes in metabolic activities, accumulation and translocation of some essential nutrients (phosphorus, sulphur, boron, iron, manganese, copper and zinc) in radish (Raphanus sativus) when grown in refined sand under different concentrations of Cr(VI). Radish was chosen for study, as it is a common vegetable crop plant of global importance. World production of radish is estimated to be about 7 million t/year, representing roughly 2 % of all vegetables (Kopta and Pokluda 2013).
Materials and Methods
Raphanus sativus var Pusa chetki was grown in polyethylene pots (5 L capacity) filled with refined sand saturated with nutrient solution and kept in a glass house at ambient temperature, according to the method of Hewitt (1966). The composition of nutrient solution was 4 mM KNO3, 4 mM Ca(NO3)2, 2 mM MgSO4, 1.33 mM NaH2PO4, 100 μM Fe-EDTA, 10 μM MnSO4, 30 μM H3BO3, 1 μM CuSO4, 1 μM ZnSO4, 0.2 μM Na2 MoO4, 0.1 μM NiSO4, 0.1 μM CoSO4, and 0.1 mM NaCl. Nutrient solution was supplied daily except weekends when the pots were flushed with deionised water to remove accumulated salts and roots exudates. On day 41, pots, with four plants in each, were divided into six lots of three pots in each lot. One lot was allowed to grow with full nutrient solution supplied daily to serve as control (without Cr treatment). The remaining lots were supplied with Cr as K2Cr2O7 at 0.05, 0.10, 0.20, 0.30, 0.40 mM concentration superimposed in nutrient solutions and visible toxic effects, change in growth parameters, metabolic activity, uptake and translocation of essential elements was investigated. Leaf area (cm2) was measured by Delta-T leaf area measurement system as an index of plant growth after 15 days of Cr exposure. At day 55 (15 days after beginning of Cr treatment), plants were removed from pots and thoroughly washed with tap water followed by rinsing with deionized water.
Different parameters in harvested plant were examined following the methods provided in Table 1. Oven-dried plant samples were digested 10:1 HNO3:HClO4 (Piper 1942) for metal estimation. Metal concentration in clear digest samples were measured on the Inductively-Coupled Plasma Spectrometer, (ICP Optima 3300 RL) Perkin Elmer Corporation, USA. The standard reference materials of Fe, Zn (BND 1101.02; provided by the National Physical Laboratory, New Delhi, India), Cr and Cu (EPA quality control samples; Lot TMA989) were used for calibration and quality assurance. Analytical data quality of metals was ensured through repeated analysis (n = 3) of standard reference samples and the results were found to be within ±2.03 % to ±2.95 % of certified values. The mean recovery was 96 %–98.5 % for different metals. Blanks were run in triplicate to check the precision of the method with each set of samples. The detection limits for Fe, Zn, Cr and Cu were 0.3, 0.2, 0.5 and 0.9 μg L−1, respectively. The experiment was arranged in a randomized complete block design and duplicated. To confirm the variability of data and validity of results, all data were subjected to two way analysis of variance (ANOVA) to determine significance between treatments. For group wise comparison of means Duncan’s Multiple Range Test (DMRT) was applied to see the significant level (Gomez and Gomez 1984).
Results and Discussion
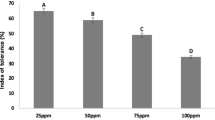
After 5 days of exposure, foliar symptoms of excess Cr were noted as marginal chlorosis from the base of lamina of lower leaves, which gradually spread to the upper part of the leaf. After 15 days of Cr exposure, a significant (DMRT, p < 0.05) depression in growth in terms of height, leaf area and dry weight was observed as compared to controls (Table 2). These symptoms of excess Cr in radish plants are new to literature and somewhat resemble those described by (Tiwari et al. 2009). Similarly, a significant decrease in chlorophyll contents, Hill reaction activity, carotenoid and catalase was observed as compared to control plants after 15 days of Cr exposure (DMRT, p < 0.05). Previous studies have documented these decreases after Cr exposure (Dat et al. 1998; Panda et al. 2003). Excess treatment of Cr up to 0.10 mM caused a slight increase in ribonuclease activity which contradicted earlier findings by Dua and Sawhney 1991. The protein content in leaves of radish decreased from 2.52 to 1.23 (% fw) with increase in Cr(VI) exposure. Degradation of protein in plants can result in the inhibition of nitrate reductase (NR) activity (Solomonson and Barbar 1990). The leaves of radish supplied with excess of Cr increased non-reducing and reducing sugars. Sugar accumulation may be due to a metal induced alternative of carbohydrate metabolism (Rauser and Samarkoon 1980) (Table 3). The concentration of protein-nitrogen increased with increased Cr concentration in nutrient solution as compared to control; however, non-protein nitrogen concentration decreased at higher concentration of Cr as reported earlier (Sharma et al. 1995). Activity of glutathione reductase decreased significantly due to differential Cr(VI) treatment as compared to control plant. Similarly, activity of lipid peroxidation and proline was increased significantly (DMRT, p < 0.05) in leaves of radish by Cr(VI) exposure as compared to that of control plant (Table 4). Lipid peroxidation, which is considered an indication of oxidative stress in plants and proline, is the only amino acid that accumulates to a greater extent in the leaves of many plants under stress (Azmat and Akhter 2010). An excess of Cr (0.4 mM) caused a significant decrease in concentration of Fe in leaves (71.9 μg g−1 dw) as compared to control (134.3 μg g−1 dw) after 15 days of treatment. Similarly, translocation of S, P and Zn from root to leaves decreased significantly (DMRT, p < 0.05). However, translocation of Mn, Cu and B were less affected as reported earlier (Chatterjee and Chatterjee 2000) (Fig. 1). Fe, S and P are known also to compete with Cr for carrier binding (Wallace et al. 1976).
Effect of variable chromium (VI) exposure on a sulphur (% dw), b phosphorus (% dw), c iron (μg g−1 dw), d manganese (μg g−1 dw) translocation in different plant parts of radish (R. sativus), at 55 days after; 15 days of chromium supply. All values are mean ± SD (n = 3). ANOVA significant at p ≤ 0.01. Different letters indicate significantly different value (DMRT, p < 0.05)
After 15 days of Cr exposure, maximum accumulation of Cr was found in root (327.6 μg g−1 dw) followed by stem (186.8 μg g−1 dw) and leaves (116.7 μg g−1 dw) at 0.4 mM Cr concentration (Fig. 2). However, the estimated safe daily dietary intake of Cr is 60 μg day−1 (World Health Organization 1994). The reason of high accumulation in the roots of plant could be due to Cr immobilization in the vacuoles of the root cells, thus rendering it less toxic (Shanker et al. 2004). It may be concluded from the present investigation that exposure of Cr caused toxicity, affected translocation of essential nutrients and resulted in accumulation of high amount of chromium in radish (R. sativus). Visible symptoms and metabolic changes indicated toxic effects of Cr(VI) on plant growth and development. Cr affects plant metabolism through competition in uptake of essential nutrients or inactivation of enzymes by displacement at functional sites. Thus, radish grown in Cr contaminated agricultural fields may be hazardous to humans and animals.
Effect of variable chromium (VI) exposure on a copper, b zinc, c boran, d chromium (μg g−1 dw) translocation in different plant parts of radish (R. sativus), at 55 days after; 15 days of chromium supply. All values are mean ± SD (n = 3). ANOVA significant at p ≤ 0.01. Different letters indicate significantly different value (DMRT, p < 0.05)
References
Akinola MO, Ekiyoya TA (2006) Accumulation of lead, cadmium and chromium in some plants cultivated along the bank of river Ribila at Odo-nla area of lkorodu, Layos state, Nigeria. J Environ Biol 27:597–599
Arnon DI (1949) Copper enzyme in isolated chloroplasts. Polyphenol oxidase in Beta vulgaris. Plant Physiol 24:1–15
Azmat R, Akhter H (2010) Changes in some biophysical and biochemical parameters of mungbean (Vigna radiata L) Wilczek grown on chromium-contaminated soil treated with solid tea wastage. Pak J Bot 42:3065–3071
Barrs HD, Weatherly PE (1973) A reexamination of the relative turgidity technique for estimating water deficits in leaves. Aust J Biol Sci 15:413–428
Bates LS, Waldren RP, Thomas CE (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–207
Bisht SS, Sharma D, Chaturvedi K (1989) Certain metabolic lesions of chromium toxicity in radish. Indian J Agr Biochem 2:109–115
Bradford MN (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Brewer J, Jogendorf AT (1965) Damage to spinach chloroplast induced by dark preincubation with ferricyanide. Plant Physiol 40:303–311
Caglieri A, Goldoni M, Acampa O, Andreoli R, Vettori MV, Corrapadi M, Apostoli P, Mutti A (2006) The effect of inhaled chromium on different exhaled breath condensate biomarkers among chrome-plating workers. Environ Health Perspect 114:542–546
Chatterjee J, Chatterjee C (2000) Phytotoxicity of cobalt, chromium and copper in cauliflower. Environ Pollut 109:69–74
Chesnin I, Yien CH (1951) Turbidimeteric determination of available sulphate. Proc Sci Soc Am 15:149–151
Dat JF, Lopez-Delgado H, Foyer CH, Scott IM (1998) Parallel changes in H2O2 and catalase during thermotolerance induced by salicylic acid or heat acclimation in mustard seedlings. Plant Physiol 116:1351–1357
Dua A, Sawhney SK (1991) Effect of chromium on hydrolytic enzymes in germinating pea seeds. Environ Exp Bot 31:133–139
Gomez KA, Gomez AA (1984) Statistical procedures for agricultural research. Wiley, New York
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Hewitt EJ (1966) Sand and water culture methods used in the Study of Plant Nutrition, Technical Communication No. 22. Commonwealth Agricultural Bureau, London
Kopta T, Pokluda R (2013) Yields, quality and nutritional parameters of radish (Raphanus sativus) cultivars when grown in organically in Czech Republic. Hort Sci 40:16–21
Luck H (1963) Peroxidase. In: Hill B (ed) Methods in enzymatic analysis. Academic, New York, pp 895–897
Montgomery R (1957) Determination of glycogen. Arch Biochem Biophys 67:378–386
Nelson N (1944) Photometric adaptation of Somogyi method for determination of glucose. J Biol Chem 153:375–380
Panda SK, Chaudhury I, Khan MH (2003) Heavy metals induce lipid peroxidation and affects antioxidants in wheat leaves. Biol Plant 46:289–294
Parmar NG, Chanda SV (2005) Effects of mercury and chromium on peroxidase and IAA oxidase enzymes in the seedlings of Phaseolus vulgaris. Turk J Biol 29:15–21
Piper CS (1942) Soil and plant analysis, monograph. Waite Agricultural Research Institute, The University, Adelaide
Rauser WE, Samarkoon AB (1980) Vein loading in seedling of Phaseolus vulgaris exposed to excess cobalt, nickel and zinc. Plant Physiol 65:578–583
Shanker AK, Djanaguiraman M, Sudhagar R, Jayaram K, Pathmanabhn G (2004) Expression of metallothioneins 3-like protein mRNA in sorghum cultivars under Cr(V1) stress. Curr Sci 86:901–902
Sharma DC, Chaterjee C, Sharma CP (1995) Chromium accumulation and its effect on wheat (Triticum aestivum L. cv. HD 2004) metabolism. Plant Sci 111:145–151
Smith IK, Vierhellaer TL, Thorne CA (1988) Assay of glutathione reductase in crude tissue homogenates using 5.5-dithiobis (2-nitrobenzoic acid). Anal Biochem 175:408–413
Solomonson LP, Barbar MJ (1990) Assimilatory nitrate reductase:functional properties and regulation. Ann Rev Plant Mol Biol 41:225–253
Swain T, Hillis WE (1959) The phenolic constituent of Prunus domestica. The qualitative analysis of phenolic constituents. J Sci Food Agri 10:63–68
Tiwari KK, Dwivedi S, Singh NK, Rai UN, Tripathi RD (2009) Chromium (VI) induced phytotoxicity and oxidative stress in pea (Pisum sativum L.): biochemical changes and translocation of essential nutrients. J Environ Biol 30:389–394
Tiwari KK, Singh NK, Patel MP, Tiwari MR, Rai UN (2011) Metal contamination of soil and translocation in vegetables growing under industrial waste water irrigated agricultural field of Vadodara, Gujarat, India. Ecoto Environ Saf 74:1670–1677
Tuve TV, Anfinsen CB (1960) Preparation and properties of spinach ribonuclease. J Biol Chem 235:3437–3441
Wallace T (1951) A colour atlas and guide. 2nd HMSO, London
Wallace A, Soufi SM, Cha JW, Romny EM (1976) Some effects of chromium toxicity on bush been plants grown in soil. Plant Soil 44:471–473
WHO, ICPS (1994) Biological monitoring of heavy metal stress in modulated white lupin: strategies to avoid toxicity. Plant Physiol Biochem 40:1003–1009
Zou J, Yu K, Zhang Z, Jiang W, Liu D (2009) Antioxidant response system and chlorophyll fluorescence in chromium (IV) treated Zea mays L. seedlings. Acta Biol Cracoviensi Ser Bot 51:23–33
Acknowledgments
Authors are thankful to Director, CSIR-NBRI for providing facilities. KKT is grateful to Department of Science and Technology (Govt. of India), New Delhi, and GUJCOST, Gandhinagar for providing financial assistance and Director SICART for encouragements. NKS acknowledge Council of Scientific and Industrial Research, New Delhi for Research Associate Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tiwari, K.K., Singh, N.K. & Rai, U.N. Chromium Phytotoxicity in Radish (Raphanus sativus): Effects on Metabolism and Nutrient Uptake. Bull Environ Contam Toxicol 91, 339–344 (2013). https://doi.org/10.1007/s00128-013-1047-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-013-1047-y