Abstract
Estrogenic potency of water samples from various rivers and wastewater treatment plants (WWTPs) in Shanghai was determined to assess the estrogenic pollution in typical Chinese urban regions. While the estrogenic activity, calculated as estradiol equivalents (EEQs), in surface water was found to be 2.48–41.9 ng/L, it was 47.7–80.1 ng/L in the influents from WWTPs. After treatment (removal rate of 62.3%–83.6%), the EEQ level in effluents was 7.82–24.8 ng/L, which was within the range of the corresponding receiving rivers. Our findings suggest that WWTP effluents may not be the predominate source of potential aquatic estrogenic contamination in Shanghai.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Endocrine disrupting compounds (EDCs) from natural and synthetic sources have received a great deal of attention since they are potential environmental contaminants capable of interfering with the functions of the endocrine system (Colborn and Clement 1992). In particular, EDCs belonging to the esgtrogenic class have been studied the most, because they can mimic, increase, or inhibit the action of endogenous estrogens by a number of divergent pathways (Gillesby and Zacharewski 1998). The lowest observable effect concentration (LOEC) of estrogens affecting fish production in the laboratory was found to be 0.2–1.0 ng/L, which is lower than the concentration detected in some contaminated water (Zha et al. 2008), suggesting a potential health risk and a huge challenge for aquatic animals.
The presence of estrogens in aquatic environment is a growing global problem, and it has been reported in a number of Asian countries (Duong et al. 2010). In China, researchers have begun to evaluate the estrogenic potency in water samples from areas such as Taihu Lake (Shen et al. 2001), Beijing (Ma et al. 2007), Tianjin (Lei et al. 2009), Pearl River, South China (Gong et al. 2009) and Nanjing (Lu et al. 2010), but the data are still limited. Shanghai is one of the ten biggest cities in the world with a population of nearly 20 millions. It is also China’s largest industrial base and foreign trade port located in central China’s Yangtze River estuary coastline. In this study, surface water from four rivers as well as the influents and effluents from four wastewater treatment plants (WWTPs) in Shanghai were collected in the rainy season and dry season for assessment of estrogenic potency using Yeast estrogen screen (YES). This study was conducted to evaluate estrogenic activity in the aquatic environment in China, and to gain insight into the possible pollution levels of estrogenic chemicals in the municipal area of Shanghai.
Materials and Methods
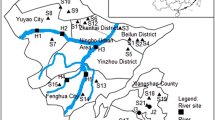
Surface water samples from 38 sites along Huangpu River, Suzhou Creek, Yunzao Brook and Yangtze River in and around Shanghai were collected during both the rainy season (April) and the dry season (November) of 2010 (Fig. 1). Influents and effluents, as well as different processed waste water samples, were collected from four municipal WWTPs in Shanghai.
A recombinant yeast strain (Saccharomyces cerevisiae) was kindly provided by Water Environment Technology Co. Ltd., Wuxi Chinese Academy of Sciences. All water samples were filtered through a 0.45 μm membrane for the YES assay, and in some cases, the influents from WWTPs were diluted with ultra-pure water due to the high value of estradiol equivalents (EEQ). A serial dilution of 17β-estradiol (E2, 0.272–272 ng/L) was used to plot an estrogenic concentration–response standard curve. The yeast cells were cultured at 29–30°C with shaking at 200 rpm for 48 h to the logarithmic phase with an optical density value of about 0.7 at 600 nm (OD600). An aliquot of yeast culture (1 mL) was then added to a water sample (8 mL) and incubated at 30°C with shaking at 200 rpm for 2 h in the presence of culture medium. All the water samples were assayed in a minimum of triplicates, and each included a positive control (E2, 27.2 ng/L) and a negative control (DMSO). After 2-h exposure, the yeast cells were measured for their OD600 value and harvested by centrifugation, followed by a β-galactosidase assay. The EEQ values were determined by comparing with E2 induction rates. The matrix spike recoveries were at a range of 66.7%–78.7%. Samples were negative in terms of cytotoxicity toward yeast cells.
Results and Discussion
An S-shape curve of the concentration of E2 versus β-galactosidase activity was plotted (Fig. 2). The calculated EC50 of E2 was found to be 33.1 ± 7.95 ng/L, which is comparable to a value reported from another study (Ma et al. 2007).
A standard dose–response curve for 17β-estradiol. The β-galactosidase activity (U) was calculated according to an equation described by Li et al. (2009)
The estrogenic activities in the water samples are summarized in Fig. 3. In the rainy season, EEQ values ranged from 2.49 to 40.3 ng/L, with a mean of 18.4 ng/L; in the dry season, EEQ values ranged from 4.51 to 41.9 ng/L, with a mean of 21.2 ng L−1. These data are similar to those found in other Asian countries (Duong et al. 2010), in which the estimated estrogenic activity in the river water ranged from values below detection limits to EEQ values of 66.9 ng/L. Table 1 shows the comparison of estrogenic activity in this study with previously reported data. It can be seen that estrogenic contamination in aquatic environment is a global phenomenon, especially in Asian countries. Our data revealed a relatively higher level of estrogenic activity in surface water and WWTPs water of Shanghai as compared to the estrogenic pollution in other regions of China from previous reports (Ma et al. 2007; Lu et al. 2010). This could be attributed to huge anthropogenic discharges into the aquatic environment as a consequence of Shanghai having the largest population density, industrial base and foreign trade port in China.
In both seasons, the sampling sites with the highest EEQs were h6 (40.3 ± 7.01 ng/L in the rainy season and 39.3 ± 9.65 ng/L in the dry season), which is located at one of the biggest chemical industry areas of the city, and s8 (38.6 ± 10.8 ng/L in the rainy season and 41.9 ± 3.44 ng/L in the dry season) near the downstream outlet of a municipal sewage discharge. Site h8 located near the Shanghai Expo Park area had the lowest EEQ value of 2.49 ± 1.22 ng/L in the rainy season; this site also received several rounds of comprehensive enviromental rehabilitation since 2000. However, in the dry season, the estrogenic activity at h8 was increased to 21.2 ± 2.63 ng/L, indicating an increase in estrogenic pollution in that area after the Expo. In contrast, water from the upper reaches of Hangpu River (h1 site, located close to the water resource conversation area) collected in the dry season was found to have a low EEQ value that was below the average.
In general, the average EEQ values of river water samples collected in the rainy season are all very similar, except for Yunzao brook, which is slightly lower (Table 2). Moreover, the EEQ levels were slightly increased in Huangpu River and Suzhou Creek in the dry season. Based on sampling areas, the surface water with the highest EEQ value was found in the neighborhoods of industrial factories and drainage outlets, indicating possible point-source estrogenic pollution from industrial and municipal effluents. It is known that most estrogenic steroids have relatively short half-lives (estimated 2–6 days) in water and sediment, while microorganisms in water are known to be capable of degrading some estradiols (Williams et al. 1999). Owing to constant dilution and the presumed biodegradability of river water, we concluded that there is not a significant tendency for accumulation of EDCs in the four rivers surveyed in Shanghai along the river flow path from upstream to downstream.
The estrogenic activity in influents and effluents from the four WWTPs are shown in Fig. 4a. The EEQ level in influents was relatively high at 47.7–80.1 ng/L, but in effluents it was reduced to 7.82–24.8 ng/L. The estrogenic removal efficiencies of WWTP1, WWTP2, WWTP3 and WWTP4 were 71.0%, 62.3%, 65.9% and 83.6%, respectively. For WWTP1 and WWTP3, industrial waste was a partial source of the wastewater, and thus the EEQ values of influents were higher than in the other two plants, but their effluent EEQ values are still within the range reported in previous studies (Cargouët et al. 2004). Estrogenic activity in different processed water from WWTP4 is shown in Fig. 4b. It was found that after successive treatment processes, estrogenic potency in wastewater had been reduced significantly, which was consistent with previous studies on the removal rate of estrogenic potency in WWTPs from Germany (Körner et al. 2000), France (Cargouët et al. 2004) and USA (Tilton et al. 2002).
a Estrogenic activity in the influents and effluents from different WWTPs in Shanghai; b estrogenic activity in the water samples after successive treatment processes from WWTP4. I, the influent; II, the water following primary sedimentation process; III & IV, the water samples following biological phosphorus and nitrogen removal process; V, the water following aerated activated sludge and UV process (the effluent). Samples labeled with the same letters are not significantly different from each other, while those labeled with different letters indicate a statistically significant difference between samples. (p < 0.05, one way ANOVA followed by LSD test). Values are means ± S.D. (n = 4)
Waste water treatment plants receive a large spectrum of molecules from domestic and/or industrial waste that have been shown not to be totally eliminated during the treatment processes (Ternes et al. 1999). The ability of WWTPs to remove steroid estrogens and other EDCs from influents is crucial in maintaining the water quality of the receiving river. A correlation between reproductive abnormalities in fish and exposure to WWTP effluents has been reported (Jobling et al. 1998). One study estimated that the WWTP treatment processes globally eliminated only about 50% of the total estrogens in wastewater (Cargouët et al. 2004). Therefore, the fates of estrogens remaining in the effluents and their possible effects on health of humans and aquatic animals should not be ignored.
The untreated wastewater from the four WWTPs had high average EEQ value of 61.9 ± 12.4 ng/L; after a series of treatment processes, the effluents had an average EEQ value of 19.1 ± 6.32 ng/L, which is similar to the estrogenic activity in surface water of the receiving rivers (18.4 ng/L in the rainy season and 21.2 ng/L in the dry season). Our data suggest that the effluents with incomplete removal of estrogenic compounds from WWTPs might be a minor source of estrogenic pollution in the surface water, but direct discharge of untreated water into the receiving river, which accounts for at least 20% of the total discharge of municipal wastewater in the city, might be a major source of estrogenic pollution in the Shanghai municipal area. Although our results indicated the presence of estrogenic contamination in the surface water of Shanghai city, further investigation is required to determine the source and cause of estrogenic pollution.
References
Cargouët M, Perdiz D, Mouatassim-Souali A, Tamisier-Karolak S, Levi Y (2004) Assessment of river contamination by estrogenic compounds in Paris area (France). Sci Total Environ 324:55–66
Colborn T, Clement C (1992) Chemically-induced alterations in sexual and functional development: the wildlife/human connection. Princeton Scientific Publishing Co Inc, Princeton
Duong CN, Ra JS, Cho J, Kim SD, Choi HK, Park JH, Kim KW, Lnam E, Kim SD (2010) Estrogenic chemicals and estrogenicity in river waters of South Korea and seven Asian countries. Chemosphere 78:286–293
Gillesby BE, Zacharewski TR (1998) Exoestrogens: mechanisms of action and strategies for identification and assessment. Environ Toxicol Chem 17:3–14
Gong J, Ran Y, Chen DY, Yang Y, Ma XX (2009) Occurrence and environmental risk of endocrine-disrupting chemicals in surface waters of the Pearl River, South China. Environ Monit Assess 156:199–210
Hashimoto S, Horiuchi A, Yoshimoto T, Nakao M, Omura H, Kato Y (2005) Horizontal and vertical distribution of estrogenic activities in sediments and waters from Tokyo Bay, Japan. Arch Environ Contam Toxicol 48:209–216
Jobling S, Nolan M, Tyler CR, Brighty G, Sumpter JP (1998) Widespread sexual disruption in wild fish. Environ Sci Technol 32:2498–2506
Körner W, Bolz U, Süssmuth W, Hiller G, Schuller W, Hanf V, Hagenmaier H (2000) Input/output balance of estrogenic active compounds in a major municipal sewage plant in Germany. Chemosphere 40:1131–1142
Lei BL, Huang SB, Zhou YQ, Wang DH, Wang ZJ (2009) Levels of six estrogens in water and sediment from three rivers in Tianjin area, China. Chemosphere 2:36–42
Li F, Chen J, Wang Z, Li J, Qiao X (2009) Determination and prediction of xenoestrogens by recombinent yeast-based assay and QSAR. Chemosphere 74:1152–1157
Lu GH, Song WT, Wang C, Yan ZH (2010) Assessment of in vivo estrogenic response and the identification of environmental estrogens in the Yangtze River (Nanjing section). Chemosphere 9:982–990
Ma M, Rao KF, Wang ZJ (2007) Occurrence of estrogenic effects in sewage and industrial wastewaters in Beijing, China. Environ Pollut 147:331–336
Shen JH, Gutendorf B, Vahl HH, Shen L, Westendorf J (2001) Toxicological profile of pollutants in surface water from an area in Taihu Lake, Yangtze Delta. Toxicology 166:71–78
Snyder SA, Keith TL, Verbrugge DA, Snyder EM, Gross TS, Kannan K (2001) Identification and quantification of estrogen receptor agonists in wastewater effluents. Environ Sci Technol 35:3620–3625
Ternes TA, Stumpf M, Mueller J, Haberer K, Wilken RD, Servos M (1999) Behavior and occurrence of estrogens in municipal sewage treatment plants-I. Investigations in Germany, Canada and Brazil. Sci Total Environ 225:81–90
Tilton F, Benson WH, Schlenk D (2002) Evaluation of estrogenic activity from a municipal wastewater treatment plant with predominantly domestic input. Aquat Toxicol 61:211–224
Williams RJ, Jurgen MD, Johnson AC (1999) Initial predictions of the concentrations and distribution of 17β-oestradiol, oestone and ethinyl oestradiol in 3 English rivers. Water Res 33:1663–1671
Zha JM, Sun LW, Ma M, Wang ZJ (2008) Assessment of 17α -ethinylestradiol effects and underlying mechanisms in a continuous, multigeneration exposure of the Chinese rare minnow (Gobiocypris rarus). Toxicol Appl Pharmacol 226:298–308
Acknowledgments
This work was supported by National Natural Science Foundation of China (No. 40830744) and Shanghai Leading Academic Discipline Project (No. S30109).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, M., Wang, K., Shen, Y. et al. Evaluation of Estrogenic Activity in Surface Water and Municipal Wastewater in Shanghai, China. Bull Environ Contam Toxicol 87, 215–219 (2011). https://doi.org/10.1007/s00128-011-0334-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-011-0334-8