Abstract
Aims/hypothesis
The study aimed to compare participant characteristics, treatment modalities and clinical outcomes in registry participants less than 6 years old.
Methods
Participant characteristics, treatment modalities and clinical outcomes (HbA1c, severe hypoglycaemia [SH] and diabetic ketoacidosis [DKA]) as well as frequencies of attaining HbA1c goals in line with the International Society for Pediatric and Adolescent Diabetes (<7.5% [<58 mmol/mol]) and ADA (<8.5% [<69 mmol/mol]) were compared.
Results
Insulin pump use was more frequent (74% vs 50%, p < 0.001) and HbA1c levels lower in the Prospective Diabetes Follow-up Registry (DPV) than in the T1D Exchange (T1DX) (mean 7.4% vs 8.2%, p < 0.001). A lower HbA1c level was seen in the DPV compared with the T1DX for both pump users (p < 0.001) and injection users (p < 0.001). More children from DPV were meeting the recommended HbA1c goals, compared with children from T1DX (HbA1c <7.5%: 56% vs 22%, p < 0.001; HbA1c <8.5%: 90% vs 66%, p < 0.001). The adjusted odds of having an HbA1c level <7.5% or <8.5% were 4.2 (p < 0.001) and 3.6 (p < 0.001) higher for the DPV than the T1DX, respectively. The frequency of SH did not differ between registries or by HbA1c, whereas the frequency of DKA was higher for the T1DX and greater in those with higher HbA1c levels.
Conclusions/interpretation
DPV data indicate that an HbA1c of <7.5% can frequently be achieved in children with type 1 diabetes who are under 6 years old. An improved metabolic control of type 1 diabetes in young patients appears to decrease the risk of DKA without increasing SH. The greater frequency of suboptimal control in young patients in the T1DX compared with the DPV is not fully explained by a less frequent use of insulin pumps and may relate to the higher HbA1c targets that are recommended for this age group in the USA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The incidence of type 1 diabetes is increasing worldwide by 2–5% annually, the greatest increases being reported in children less than 6 years old [1–4]. Such increases in the prevalence of type 1 diabetes in very young children are worrisome in part because clinical care for diabetes in this age group presents numerous challenges related to erratic eating, inconsistent physical activity, limited ability to report hypoglycaemic symptoms and the need for very small doses of insulin. The best metrics for defining optimal diabetes control and the methods to achieve them in this population have not yet been established. For example, the ADA recommends that clinicians should strive to achieve HbA1c concentrations <8.5% (<69 mmol/mol) in this age group [5], whereas according to the International Society for Pediatric and Adolescent Diabetes (ISPAD) the target HbA1c concentration should be <7.5% (<56 mmol/mol) [6].
A paucity of published data on current clinical outcomes has limited the development of evidence-based guidelines for the management of very young children with type 1 diabetes. Data are even more limited for the acute and long-term benefits and consequences of achieving the currently recommended HbA1c target concentrations in very young patients. Identifying the demographic and treatment factors associated with reaching these HbA1c goals would also provide data to guide clinical care.
The T1D Exchange (T1DX) Registry in the USA and the Prospective Diabetes Follow-up Registry (DPV) in Germany and Austria are two large consortia of paediatric diabetes centres that were established with an objective of improving the care of children with type 1 diabetes through sharing best practices and collecting clinical outcome data from large numbers of patients. In this collaborative study, both the T1DX and the DPV used queries of their databases to describe and compare participant characteristics, treatment modalities and clinical outcomes in registry participants less than 6 years old.
Methods
Participants
The T1DX clinic network includes 70 US-based paediatric and adult endocrinology practices. A registry of more than 26,000 individuals with type 1 diabetes commenced enrolment in September 2010 [7]. Each clinic received approval from a local institutional review board (IRB). Informed consent was obtained according to IRB requirements. Data were collected for the registry’s central database from the participant’s medical record and by having the parent complete a comprehensive questionnaire, as previously described [7]. All young children less than 6 years old with a duration of type 1 diabetes of at least 1 year (n = 2,622) were included in this report; 674 participants enrolled in the T1DX at the 52 sites with paediatric patients from September 2010 to August 2012, and 1,948 patients from the DPV who had at least one office visit in either 2011 or 2012.
Study design
The DPV registry is a prospective longitudinal standardised computer-based documentation system for patients with all types of diabetes. Currently, more than 90% of German and more than 70% of Austrian children with diabetes are included in the registry. Data are documented locally by the participating centres on an electronic health record. Twice yearly, anonymised data are exported and transmitted for central analysis. Missing and inconsistent data are reported back to the centres for correction. Data collection is approved by the ethics committee at Ulm University and by the IRBs at the participating centres [8, 9].
In the T1DX, data were obtained through a combination of clinic and participant report. Method of insulin delivery (pump/injection), height, weight and frequency of severe hypoglycaemia (SH) and diabetic ketoacidosis (DKA) were extracted from the medical chart. The BMI z score was calculated from height and weight and adjusted for age and sex, using WHO reference tables [10, 11]. The number of boluses (or short-acting injections) per day, rates of self-monitoring of blood glucose (SMBG) and continuous glucose monitor (CGM) use were obtained from participant reports via completion of a questionnaire. Conversely, all data from the DPV were extracted from the medical record, as documented by the members of the diabetes team during routine patient care. In the DPV, SMBG was obtained from glucose meters when available and otherwise from participant-reported SMBG diaries. In both studies, prandial and total daily insulin (TDI) were obtained from pump downloads when available and otherwise from participant report.
Outcome measures
The median HbA1c over the year prior to the registry assessment, excluding any values obtained within 3 months of diagnosis, was used to represent HbA1c in this analysis. For both the T1DX and DPV, all HbA1c values were DCCT-standardised [12]. For both the T1DX and DPV, episodes of DKA were defined by: (1) having either an arterial or a venous pH less than 7.30 or a serum bicarbonate less than 15 mmol/l, and (2) receiving treatment provided in a healthcare facility. The T1DX definition required two additional criteria: (3) symptoms such as polyuria, polydipsia, nausea or vomiting, and (4) elevated serum ketone levels or high/moderate urine ketone levels. SH was defined by both registries as a hypoglycaemic event in which seizure or loss of consciousness occurred [13, 14]. Migration history for the DPV was defined as at least one parent born outside of Germany or Austria.
Statistical methods
Summary statistics were calculated within registries and by insulin delivery methods. The proportions of participants with at least one reported SH event and/or at least one DKA event in the previous 12 months were tabulated by HbA1c category (<7.5% [<58 mmol/mol], 7.5 to <8.5% [58 to <69 mmol/mol] and ≥8.5% [≥69 mmol/mol]). Wilcoxon or χ 2 tests were performed to compare the basic demographic and clinical characteristics between registries, and to compare pump and injection users within each registry. Adjusted linear and logistic regression models were run to compare clinical outcomes between the registries. All statistical analyses were performed using SAS 9.3 (SAS Institute, Cary, NC, USA). All p values are two-sided. A priori, in view of the large sample size and multiple comparisons, only p values <0.01 were considered statistically significant.
Results
Young children in the T1DX and DPV had similar clinical characteristics with respect to sex, age and BMI z scores (Table 1). The duration of type 1 diabetes differed between the two registries, as did diabetes management factors such as TDI dose/kg, percentage of prandial insulin and frequency of reported SMBG. The two registries differed substantially with respect to the percentage of young patients who were using an insulin pump: 50% of patients in the T1DX vs 74% in the DPV (p < 0.001). CGM use was uncommon in both cohorts (Table 1). Within the DPV registry, 23% of patients had a history of migration, whereas in the T1DX 19% were not non-Hispanic whites.
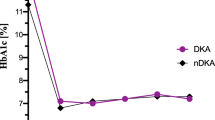
Despite many similarities in terms of clinical characteristics and treatment factors, the mean HbA1c was notably higher (p < 0.001, Table 1, electronic supplementary material [ESM] Fig. 1) and the percentage of very young children with type 1 diabetes meeting the recommended HbA1c goals was lower (p < 0.001, Fig. 1a, b) in the T1DX than the DPV. Children in the T1DX using pump therapy had a lower average HbA1c than those receiving injections and were more likely to have an HbA1c <7.5% (<58 mmol/mol) and <8.5% (<69 mmol/mol) (p < 0.001 for all). In contrast, in the DPV cohort, neither the mean HbA1c nor the likelihood of reaching an HbA1c goal of <7.5% (<58 mmol/mol) or <8.5% (<69 mmol/mol) differed between patients using a pump or injections (p ≥ 0.01 for all).
(a) Percentage with HbA1c <7.5% (<58 mmol/mol) overall and by insulin method. (b) Percentage with HbA1c <8.5% (<69 mmol/mol) overall and by insulin method. White bars, T1DX; black bars, DPV. ***p < 0.001; p values adjusted for age, duration of type 1 diabetes and sex. Error bars show 95% CI. To convert values for HbA1c in % into mmol/mol, subtract 2.15 and multiply by 10.929 or use the conversion calculator at www.HbA1c.nu/eng/
To investigate whether the HbA1c differences between registries could be attributed to measured diabetes management factors, additional linear regression analyses were performed adjusting for pump use and SMBG per day in addition to age, duration of type 1 diabetes and sex. After adjustment, the mean HbA1c was 0.7% (7.5 mmol/mol) higher among patients in the T1DX than the DPV (p < 0.001). The adjusted odds of having an HbA1c <7.5% (<58 mmol/mol) or <8.5% (<69 mmol/mol) were 4.2 (95% CI 3.4, 5.3; p < 0.001) and 3.6 (95% CI 2.9, 4.6; p < 0.001), respectively, higher in the DPV than the T1DX. Among patients using a pump, the mean HbA1c was 0.5% (5.1 mmol/mol) higher in the T1DX than the DPV (p < 0.001). The odds of having an HbA1c <7.5% (<58 mmol/mol) or <8.5% (<69 mmol/mol) were 3.0 (95% CI 2.3, 4.0; p < 0.001) and 2.9 (95% CI 2.1, 4.0; p < 0.001), respectively, higher in the DPV than the T1DX. Among patients on injections, HbA1c was 1.0% (11.1 mmol/mol) higher in the T1DX than the DPV (p < 0.001). The odds of having an HbA1c <7.5% (<58 mmol/mol) or <8.5% (<69 mmol/mol) were 7.2 (95% CI 4.9, 10.6; p < 0.001) and 5.1 (95% CI 3.5, 7.4; p < 0.001), respectively, higher in the DPV than the T1DX. Similar results were obtained when the analyses were restricted to non-Hispanic whites (T1DX) and those without a migration history (DPV).
The clinical characteristics and management factors stratified by insulin delivery method are presented in Table 2. Within each cohort, the frequency of short-acting insulin administration was doubled for pump users vs injection users, and CGM use was more prevalent in pump users. SMBG was higher among pump users for both registries (Table 2).
The frequency of one or more SH events in the previous year was similar between registries (p = 0.19, Fig. 2a), but the frequency of one or more reported DKA events in the past year was higher in the T1DX (p < 0.001, Fig. 2b). The frequency of reported SH was similar between patients using pumps compared with injections in both registries (p > 0.01, Table 2). However, the proportion of those with at least 1 DKA event in the past year was greater among pump users compared with injection users in the DPV registry (p = 0.01, Table 2).
When the frequencies of SH and DKA were compared across HbA1c categories, the results were similar for both the T1DX and DPV (Fig. 3a, b). The proportion of children with an HbA1c <7.5% (<58 mmol/mol) with one or more SH events in the previous year was not significantly different from those with a higher HbA1c in either registry (p > 0.01 for both, Fig. 3a). Conversely, the proportion of those with one or more DKA events did increase, with an increasing HbA1c in both registries (p ≤ 0.004 for both, Fig. 3b).
(a) Percentage with ≥1 SH event in past year by HbA1c. (b) Percentage with ≥1 DKA event in past year by HbA1c. White bars, mean HbA1c <7.5% (<58 mmol/l); black bars, mean HbA1c 7.5 to <8.5% (58 to <69 mmol/l); grey bars, mean HbA1c ≥8.5% (≥69 mmol/l). **p = 0.004; ***p < 0.001; p values unadjusted and testing whether the frequency of SH/DKA differs by HbA1c group, within each registry
Discussion
Despite many similarities in their clinical characteristics and management factors, children with type 1 diabetes who were under 6 years old in the DPV achieved HbA1c concentrations that were on average 0.8% (8.4 mmol/mol) lower, with a much greater proportion achieving the ISPAD HbA1c target of <7.5% (<58 mmol/mol) and the ADA HbA1c target of <8.5% (<69 mmol/mol) compared with children in the T1DX, findings that were similar in pump users and injection users. The measured clinical characteristics and management factors did not fully explain the difference in HbA1c between the cohorts.
We hypothesise that the lower HbA1c target set by ISPAD (<7.5% [<58 mmol/mol]) compared with the age-specific ADA HbA1c target (<8.5% [<69 mmol/mol]) may have contributed to the observed discrepancy, assuming that T1DX providers follow the ADA guidelines and DPV providers follow the ISPAD guidelines. The Hvidoere study found that differences in glycaemic targets played a significant role in explaining HbA1c differences between centres [15]. It has also been previously reported that a lower perceived HbA1c goal is associated with the achievement of lower levels in adolescents with T1D [16]. Thus, it may be more than a coincidence that the proportion of T1DX children meeting the ADA goal of 8.5% (69 mmol/mol) (66%) is close to that of DPV children meeting the 7.5% (58 mmol/mol) ISPAD goal (56%).
The increased use of insulin pumps in the DPV cohort is a potential factor to explain the lower HbA1c levels in the DPV cohort than in the T1DX cohort, since this method of insulin administration may allow for more precision in delivering small doses of insulin and in matching insulin delivery to insulin needs. However, the greater use of insulin pumps did not account fully for the observed HbA1c difference between the T1DX and DPV registries, although the greatest discrepancy in HbA1c levels between the two registries was in injection users. In addition, the mean HbA1c and the proportion of those meeting the HbA1c target did not differ by insulin administration method within DPV.
Differences in race/ethnicity and socioeconomic factors between the participants in the two registries may explain some of the outcome differences. Although this is a potential explanatory factor, a comparison of race and ethnicity between registries is difficult due to differences in how these data are collected by the two registries. However, differences in HbA1c still existed even when the analyses were limited to non-Hispanic whites (T1DX) and those without a migration history (DPV).
As an example of other possible contributing factors, the Hvidoere study reported language differences as a contributing factor for the centre differences in HbA1c [17]. We are unable to evaluate whether language barriers for the participants in each registry contributed to the differences in outcome that were observed. Similarly, comparisons of socioeconomic status between registries are also difficult due to registry-specific methods of collecting such data. However, within the T1DX, higher household income, higher parental education level and having private insurance have been shown to predict better HbA1c levels in children under 18 years of age [18]. In the DPV registry, children of Turkish background were half as likely to use pumps as those without a migration history even after adjustment for factors that differed by migration history (age, sex, BMI standard deviation score (BMI-SDS), outpatient visit and self-control of blood glucose) [19].
Unmeasured factors such as reimbursement issues for pumps, testing strips and other aspects of diabetes care (in the T1DX) and other demographic and socioeconomic status factors in the two registries, for example differences in healthcare systems, child care practices, insurance, pump and CGM access and cost, distance to the treatment centre and processes of care, could also contribute to the differences reported. Furthermore, within each registry there are between-diabetes-centre differences in care and outcomes. Unpublished data from the T1DX have shown a wide variation across T1DX centres in pump use among children under 6 years of age even after adjustment for a longer duration of diabetes, higher parental education and level of household income.
The fear that an increased frequency of SH has adverse neurocognitive effects in very young children was a major rationale for the higher target HbA1c levels advocated by the ADA in patients with type 1 diabetes aged less than 6 years old compared with older age groups. Consequently, one of the most important findings of the study was the demonstration that the lower ISPAD target of HbA1c levels <7.5% (<58 mmol/mol) was achieved in the majority of DPV patients without increasing the risk of SH. Other current reports in children and adults support the achievement of an HbA1c <7% (<53 mmol/mol) or <7.5% (<58 mmol/mol) without an increase in SH [11, 12, 20]. There is an increasing body of evidence that DKA and hyperglycaemia per se, in addition to hypoglycaemia, can have adverse effects on the developing brains of young children [21–23]. Thus, it is particularly noteworthy with respect to safety concerns that better HbA1c outcomes in the DPV registry were associated with a lower frequency of DKA.
Another important observation from this study is that the proportion of those with an SH event did not differ between the two cohorts or between patients treated by pump and by injection within each cohort. This observation is consistent with the findings of randomised clinical trials in older children that the use of insulin analogues, improved insulin pumps and CGM devices, and SMBG have allowed clinicians and families to achieve target HbA1c levels more safely than during the early days of intensive insulin therapy [24–28]. Both ADA and ISPAD guidelines recommend the individualisation of HbA1c guidelines as appropriate to each child and family [5, 6].
As noted above, differences in the methods of data collection between the two registries are one of the limitations of this study, including the fact that clinic-reported rates of SH may be underreported with the T1DX system. In addition, the DPV is a population-based registry that includes 70–90% of potential patients, whereas the T1DX comprises a sample of patients from paediatric diabetes centres primarily staffed by endocrinologists and only captures the families of children who volunteered to participate. Thus, the outcomes in the population aged under 6 years in the T1DX may actually be better than those in patients in this age group in the USA who are not seen at paediatric diabetes treatment centres, or those in patients receiving care at T1DX centres who did not enrol in the registry [29].
As these are registry data, HbA1c was not measured in a central laboratory and this could introduce bias. However, all methods were DCCT-standardised. We also performed three sensitivity analyses to investigate whether the difference in HbA1c between registries was due to differences in laboratory methods (within patients with HbA1c measured by a DCA 2000 analyser, with study site as a random effect and adjusted for laboratory method as a fixed effect). Differences in HbA1c level remained significantly different. Moreover, the observed difference of 0.8% (8.4 mmol/mol) is of sufficient magnitude within a large population, so that it is likely to be a true difference between registries. Despite these limitations, this is the largest sample of children under 6 years old with type 1 diabetes reported, and the magnitude of the HbA1c difference and lack of difference in frequency of SH have important clinical implications.
Clinical outcome data from the two registries do not prove cause and effect relationships, nor do they test specific hypotheses. Instead, they generate new questions and hypotheses in need of further study, for example identifying the obstacles to a more widespread use of pump (and CGM) therapy in young children in the T1DX, followed by testing whether a more widespread use of pumps (and CGM) in this population would lead to lower HbA1c concentrations. While our data provide evidence of the safety of setting lower goals for HbA1c in children under 6 years of age, further study is needed to determine whether improved glucose control in patients in this age group would translate to better glucose control throughout childhood and adolescence and reduced vascular complications [30]. It would also be useful to determine the aetiology of the lower DKA frequency in patients treated by injections in the DPV. Nevertheless, the most important direction of future studies remains the development of new drugs, devices and strategies to further improve the care of all children with type 1 diabetes.
Abbreviations
- CGM:
-
Continuous glucose monitor
- DKA:
-
Diabetic ketoacidosis
- DPV:
-
Prospective Diabetes Follow-up Registry
- IRB:
-
Institutional review board
- ISPAD:
-
International Society for Pediatric and Adolescent Diabetes
- SH:
-
Severe hypoglycaemia
- SMBG:
-
Self-monitoring of blood glucose
- T1DX:
-
T1D Exchange
- TDI:
-
Total daily insulin
References
Stanescu DE, Lord K, Lipman TH (2012) The epidemiology of type 1 diabetes in children. Endocrinol Metab Clin N Am 41:679–694
Patterson CC, Dahlquist GG, Gyurus E, Green A, Soltesz G (2009) Incidence trends for childhood type 1 diabetes in Europe during 1989-2003 and predicted new cases 2005-20: a multicentre prospective registration study. Lancet 373:2027–2033
Maahs DM, West NA, Lawrence JM, Mayer-Davis EJ (2010) Epidemiology of type 1 diabetes. Endocrinol Metab Clin N Am 39:481–497
Imperatore G, Boyle JP, Thompson TJ et al (2012) Projections of type 1 and type 2 diabetes burden in the U.S. population aged <20 years through 2050: dynamic modeling of incidence, mortality, and population growth. Diabetes Care 35:2515–2520
American Diabetes Association (2013) Standards of medical care in diabetes—2013. Diabetes Care 36(Suppl 1):S11–S66
Donaghue KC, Chiarelli F, Trotta D, Allgrove J, Dahl-Jorgensen K (2007) ISPAD Clinical Practice Consensus Guidelines 2006-2007. Microvascular and macrovascular complications. Pediatr Diabetes 8:163–170
Beck RW, Tamborlane WV, Bergenstal RM, Miller KM, Dubose SN, Hall CA (2012) The T1D Exchange Clinic Registry. J Clin Endocrinol Metab 97:4383–4389
Gerstl EM, Rabl W, Rosenbauer J et al (2008) Metabolic control as reflected by HbA1c in children, adolescents and young adults with type-1 diabetes mellitus: combined longitudinal analysis including 27,035 patients from 207 centers in Germany and Austria during the last decade. Eur J Pediatr 167:447–453
Grabert M, Schweiggert F, Holl RW (2002) A framework for diabetes documentation and quality management in Germany: 10 years of experience with DPV. Comput Methods Prog Biomed 69:115–121
Butte NF, Garza C, de Onis M (2007) Evaluation of the feasibility of international growth standards for school-aged children and adolescents. J Nutr 137:153–157
World Health Organization (2013) Growth reference data for 5-19 years. Available from http://www.who.int/growthref/en/. Accessed 12 Dec 2013
Rosenbauer J, Dost A, Karges B et al (2012) Improved metabolic control in children and adolescents with type 1 diabetes: a trend analysis using prospective multicenter data from Germany and Austria. Diabetes Care 35:80–86
Cengiz E, Xing D, Wong JC et al (2013) Severe hypoglycemia and diabetic ketoacidosis among youth with type 1 diabetes in the T1D Exchange clinic registry. Pediatr Diabetes 14(6):447–454
Weinstock RS, Xing D, Maahs DM et al (2013) Severe hypoglycemia and diabetic ketoacidosis in adults with type 1 diabetes: results from the T1D Exchange Clinic Registry. J Clin Endocrinol Metab 98(8):3411–3419
Swift PG, Skinner TC, de Beaufort CE et al (2010) Target setting in intensive insulin management is associated with metabolic control: the Hvidoere childhood diabetes study group centre differences study 2005. Pediatr Diabetes 11:271–278
Clements SA, Anger MD, Bishop FK et al (2013) Lower A1c among adolescents with lower perceived A1c goal: a cross-sectional survey. Int J Pediatr Endocrinol 2013:17
de Beaufort CE, Lange K, Swift PG et al (2013) Metabolic outcomes in young children with type 1 diabetes differ between treatment centers: the Hvidoere Study in Young Children 2009. Pediatr Diabetes 14:422–428
Campbell MS, Schatz DA, Chen V et al (2013) A contrast between children and adolescents with excellent and poor control: the T1D Exchange Clinic Registry experience. Pediatr Diabetes 15:110–117
Icks A, Razum O, Rosenbauer J et al (2012) Lower frequency of insulin pump treatment in children and adolescents of Turkish background with type 1 diabetes: analysis of 21,497 patients in Germany. Diabetes Technol Ther 14:1105–1109
Cooper MN, O'Connell SM, Davis EA, Jones TW (2013) A population-based study of risk factors for severe hypoglycaemia in a contemporary cohort of childhood-onset type 1 diabetes. Diabetologia 56:2164–2170
Perantie DC, Lim A, Wu J et al (2008) Effects of prior hypoglycemia and hyperglycemia on cognition in children with type 1 diabetes mellitus. Pediatr Diabetes 9:87–95
Barnea-Goraly N, Raman M, Mazaika P et al; Diabetes Research in Children Network (DirecNet) (2014) Alterations in white matter structure in young children with type 1 diabetes. Diabetes Care 37:332–340
Cato MA, Mauras N, Ambrosino J et al (2014) Cognitive functioning in young children with type 1 diabetes. J Int Neuropsychol Soc 20:238–247
Bergenstal RM, Tamborlane WV, Ahmann A et al (2011) Sensor-augmented pump therapy for A1C reduction (STAR 3) study: results from the 6-month continuation phase. Diabetes Care 34:2403–2405
Garg S, Ampudia-Blasco FJ, Pfohl M (2010) Rapid-acting insulin analogues in Basal-bolus regimens in type 1 diabetes mellitus. Endocr Pract 16:486–505
Singh SR, Ahmad F, Lal A, Yu C, Bai Z, Bennett H (2009) Efficacy and safety of insulin analogues for the management of diabetes mellitus: a meta-analysis. CMAJ 180:385–397
Juvenile Diabetes Research Foundation Continuous Glucose Monitoring Study Group (2008) Continuous glucose monitoring and intensive treatment of type 1 diabetes. N Engl J Med 359:1464–1476
Diabetes Control and Complications Trial Research Group (1994) Effect of intensive diabetes treatment on the development and progression of long-term complications in adolescents with insulin-dependent diabetes mellitus: Diabetes Control and Complications Trial. J Pediatr 125:177–188
Miller KM, Xing D, Tamborlane WV, Bergenstal RM, Beck RW (2013) Challenges and future directions of the T1D Exchange Clinic Network and Registry. J Diabetes Sci Technol 7:963–969
Diabetes Control and Complications Study Group (2003) Sustained effect of intensive treatment of type 1 diabetes mellitus on development and progression of diabetic nephropathy: the Epidemiology of Diabetes Interventions and Complications (EDIC) study. JAMA 290:2159–2167
Acknowledgements
A list of clinical sites in the T1DX and DPV registries contributing data to this manuscript can be found in ESM Text 1 and ESM Text 2.
Funding
The T1D Exchange is supported through the Leona M. and Harry B. Helmsley Charitable Trust. The DPV is supported through the German BMBF Competence Network Diabetes Mellitus (FKZ 01GI1106).
Duality of interest
DMM, JMH, SND, KMM, BH, LAD, BRM, ES, TMK, RWH declare that they have no duality of interest associated with this manuscript. RWB’s non-profit employer has received consultant payments on his behalf from Sanofi and Animas and a research grant from NovoNordisk with no personal compensation to RWB. WVT has received consulting fees or honorariums from Medtronic and Animas.
Contribution statement
DMM and BH researched the data and contributed to data interpretation and the writing of the manuscript. JMH, SND, RWB, WVT and RWH researched the data and contributed to data interpretation, data analysis and the writing of the manuscript. KMM, LAD, BRM and TMK contributed to data interpretation and the writing of the manuscript. ES contributed to data collection in Austria and contributed to data interpretation and the writing of the manuscript. All authors approved the final version.
Author information
Authors and Affiliations
Consortia
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM Fig. 1
(PDF 9 kb)
ESM Text 1
(PDF 114 kb)
ESM Text 2
(PDF 10 kb)
Rights and permissions
About this article
Cite this article
Maahs, D.M., Hermann, J.M., DuBose, S.N. et al. Contrasting the clinical care and outcomes of 2,622 children with type 1 diabetes less than 6 years of age in the United States T1D Exchange and German/Austrian DPV registries. Diabetologia 57, 1578–1585 (2014). https://doi.org/10.1007/s00125-014-3272-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-014-3272-2