Abstract
Purpose
The aim of this work was to retrospectively analyze efficacy, toxicity, and relapse rates of conventional (CRT) and low-dose radiotherapy (LDRT) in patients with indolent orbital lymphomas.
Patients and methods
From 1987–2014, 45 patients (median age 64 years) with 52 lesions of indolent orbital lymphomas were treated with CRT (median dose 36 Gy, range 26–46 Gy) and 7 patients (median age 75 years) with 8 lesions were treated with LDRT (2 fractions of 2.0 Gy).
Results
Median follow-up was 133 months (range 2–329 months) in the CRT group and 25 months (range 10–41 months) in the LDRT group. Overall response rates were 97.7 % (CRT) and 100 % (LDRT). The 2‑ and 5‑year local progression-free survival (PFS) rates were 93.5 and 88.6 %, distant PFS 95.0 and 89.9 %, and overall survival 100 and 85.6 % after CRT. In the LDRT group, 2‑year local PFS and overall survival remained 100 %, respectively, and distant PFS 68.6 %. Acute radiotherapy-related complications (grades 1–2) were detected in virtually all eyes treated with CRT. Cataracts developed in only patients who were irradiated with more than 34 Gy. LDRT was well tolerated with only mild acute and late complications.
Conclusion
Primary radiotherapy of indolent orbital lymphomas is an effective treatment with high response rates and excellent local control in CRT and LDRT. In combination with close follow-up, LDRT may be an attractive alternative since re-irradiation even with conventional doses is still feasible.
Zusammenfassung
Ziel
Ziel der Arbeit war die Analyse von Effektivität, Nebenwirkungen und Rezidivraten nach konventioneller (CRT) und Niedrigdosisbestrahlung (LDRT) indolenter Orbitalymphome.
Patienten und Methoden
Retrospektiv evaluiert wurden 45 zwischen 1987 und 2014 behandelte Patienten (medianes Alter 64 Jahre) mit insgesamt 52 Läsionen indolenter Orbitalymphome, die mittels CRT (mediane Dosis 36 Gy, 26–46 Gy) therapiert wurden.; 7 Patienten mit insgesamt 8 Läsionen erhielten eine LDRT (2 × 2,0 Gy).
Ergebnisse
Das mediane Follow-Up betrug 133 Monate (2–239 Monate) in der CRT- und 25 Monate (10–41 Monate) in der LDRT-Gruppe. Die Ansprechraten lagen bei 97,7 % (CRT) bzw. 100 % (LDRT). Zwei bzw. fünf Jahre nach CRT betrug das lokal progressionsfreie Überleben (PFS) 93,5 bzw. 88,6 %, das distante PFS 95,0 bzw. 89,9 % und das Gesamtüberleben 100 bzw. 85,6 %. Zwei Jahre nach LDRT ließ sich ein lokales PFS und Gesamtüberleben von jeweils 100 % bei einem distanten PFS von 68,6 % beobachten. Akute Grad-1–2-Nebenwirkungen wurden nach CRT an fast allen bestrahlten Augen dokumentiert. Katarakte entstanden nur bei Patienten, die mit einer Dosis > 34 Gy behandelt wurden. Die LDRT wurde sehr gut vertragen und verursachte nur milde Nebenwirkungen.
Schlussfolgerung
Eine primäre und alleinige Radiatio bei indolenten Orbitalymphomen erwies sich als effektive Therapiemethode mit hohen Ansprechraten und ausgezeichneten lokalen Kontrollraten, sowohl nach Bestrahlung mit einer konventionellen Dosis als auch nach einer Radiotherapie mit 4 Gy. Da bei unzureichendem Ansprechen nach LDRT eine Re-Bestrahlung sogar in voller konventioneller Dosierung möglich ist, kann eine Niedrigdosisbestrahlung mit 4 Gy unter engmaschiger, bildmorphologischer Nachsorge bei indolenten Orbitalymphomen in Erwägung gezogen werden.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Primary lymphoma of the ocular adnexa (orbital lymphoma) is a very rare tumor entity that accounts for less than 1 % of all non-Hodgkin lymphomas (NHL) and 8 % of all extranodal NHL [1, 2]. With a proportion of 55 % of all orbital tumors, they are the most frequent malignant tumor of the eye and ocular adnexae [3].
Most common histology subtypes are low-grade (indolent) lymphoma entities like marginal zone or mucosa-associated lymphoid tissue (MALT) lymphoma, follicular lymphoma (FL), or lymphoplasmacytic lymphoma (immunocytoma). However, high-grade (aggressive) lymphomas, e. g., diffuse large B‑cell lymphoma (DLBCL) or mantle-cell lymphoma (MCL), may also occur [4, 5].
These types of cancer typically originate from superficial tissues like conjunctiva, eyelids, or the lacrimal gland. Patients with early conjunctival affection characteristically present with a localized plaque, which is described in the literature as “salmon-pink”. Tumors of the periorbital/retroorbital region may present with swelling, pain, ptosis, exophthalmos, and dysfunction of the extraocular muscles. Bilateral involvement occurs in 10 % of the cases [5] and since there is no lymphatic connection between the orbits, this fact supports the concepts that there are systemic predisposing conditions [6].
The association between gastric MALT lymphoma and Helicobacter pylori infection is already well known and more recently also a connection between orbital MALT lymphoma and Chlamydia psittaci infection was identified. Ferreri et al. [7–9] verified that bacterial eradication with antibiotic therapy was often followed by lymphoma regression.
In contrast, primary intraocular lymphoma (POL) is a subset of primary central nervous system lymphoma with mainly high-grade histology subtypes like DLCBL [10, 11], associated with immune deficiency, e. g., in AIDS patients [12]. Because of the different clinical features and prognosis, therapeutic management is fundamentally different.
Radiotherapy alone is the curative option in patients with indolent lymphoma revealing high response rates and manageable toxicity [13, 14]. Several studies describe high local control rates of 90–100 % after conventional radiation with doses of 30–40 Gy [15, 16]. Radiation-related side effects were tolerable but with increasing severe complications (grade 3–4) when more than 36 Gy is administered [17–19].
The aim of this retrospective study was to review the response rates, toxicity, and relapse rates of conventional radiation doses in the treatment of patients with indolent orbital lymphomas in our institution. In addition, the efficacy and toxicity of a low-dose radiation treatment applied to patients with indolent orbital lymphomas were analyzed.
Patients and methods
Patient characteristics
Patient records of 70 patients (87 lesions) with orbital lymphoma who were treated with radiotherapy from 1987–2014 at the Department of Radiation Oncology at Heidelberg University Hospital were reviewed. Inclusion criteria for this analysis were histologically proven low-grade (indolent) orbital lymphomas treated with radiotherapy alone; high-grade lymphomas or intraocular localization were excluded for reasons explained above.
A total of 52 patients with 60 lesions (3 synchronous bilateral, 2 metachronous bilateral, 3 relapses) of indolent orbital NHL were treated with radiotherapy between 1987 and 2014 (35 left side, 25 right side). As previously described the most common histology subtype was MALT lymphoma with 51.9 %, followed by follicular lymphomas (13.5 %), immunocytomas (9.6 %), and not further classified indolent B‑cell lymphomas (25.0 %). Complete patient characteristics are shown in Tab. 1.
Most of the patients were diagnosed with stage I (82.7 %) or II (7.7 %) orbital lymphoma and radiotherapy of the orbit was therefore the primary treatment (47 patients, 90 %). Three patients with a stage III or IV disease received radiotherapy due to a lack of response after chemotherapy/immunotherapy and because they suffered from local symptoms of the eye. In 2 patients the stage was unknown (Tab. 1).
Synchronous bilateral eye involvement was found in 3 patients (9.6 %), whereas 2 patients had metachronous involvement of the contralateral eye.
Patients presented with typical symptoms like periorbital swelling (33.9 %), exophthalmos (26.8 %), ptosis (16.1 %), diplopia (10.7 %), or visual reduction (7.1 %; Tab. 2). Involved tissues were the retrobulbar region (30 %), the conjunctiva (25 %), the lacrimal gland (18.3 %), and the lid (8.3 %). In 18.4 % of patients, combined involvement of the above mentioned tissues was documented.
Radiotherapy
A conventional radiotherapy (CRT) regime was used to treat 45 patients with 52 lesions. Median age in this cohort was 64 years (range 24–84 years) with 64.4 % being female.
The median dose of 36 Gy (range 26–46 Gy) was applied five times weekly with single doses of 1.8 Gy (11 lesions) or 2.0 Gy (38 lesions). Three lesions were treated with a mixed fractioning scheme. Treatment was done using a linear accelerator: 24 lesions were treated with photons (6 MV), 25 lesions with electrons (7–20 MeV), 2 lesions with combined electron–photon beams and one with cobalt. Lesions treated in the earlier years were usually planned with an anterior photon or electron field with a hanging block for lens shielding, whereas a CT-planned 3D conformal technique was applied in the latter years.
In addition, we reviewed 7 patients with 8 lesions of orbital lymphomas who underwent low-dose radiotherapy (LDRT). Median age was 75 years (range 59–79 years) and slightly older compared to patients treated with CRT. Patients were treated from 2012–2014 with 4 Gy in two single doses of 2.0 Gy. CT-based 3D conformal technique was applied in 2 lesions, while the other 6 lesions were treated with intensity-modulated helical tomotherapy.
Statistical analysis and follow-up
Patient records, planning documents and imaging scans, if present, were reviewed. Patients who were still alive at the beginning of our analysis were contacted and were asked to complete a questionnaire. In addition to the current status of the disease and following therapies, data about toxicity were collected. Adverse events were thereafter classified according to the Common Terminology Criteria for Adverse Events v4.03 (CTCAE).
Overall survival (OS) was calculated in months from the beginning of radiotherapy until the last date of follow-up or death. Local and distant progression-free survivals (LPFS and DPFS, respectively) were calculated in months from the beginning of radiotherapy until the diagnosis of recurrent disease of the orbit or elsewhere.
OS and DPFS were calculated for all patients (45 patients with CRT and 8 patients with LDRT), whereas LPFS refers to the treated lesions (52 lesions after CRT and 8 lesions after LDRT). The survival rates were displayed using the Kaplan–Meier method. All statistical analyses were performed using the software SPSS 20.0 (IBM Corporation, Armonk, NY USA).
Results
The median follow-up period ranged from 2–329 months (median 133 months) in the CRT group and from 10–41 months (median 25 months) in the LDRT group.
Response and survival rates
Conventional radiotherapy
In 6 lesions (4 patients), no response was documented, so they were excluded from further analyses. The response rates in lesions treated with CRT are summarized in Tab. 3: 76.1 % of the irradiated lesions had complete remission (CR) and 21.7 % partial remission (PR) resulting in an overall response rate of 97.8 %. Only for one lesion (2.2 %) was progressive disease (PD) after radiotherapy documented.
One patient with initially bilateral manifestation was treated with radiotherapy of both eyes and had bilateral recurrence 38 months after CRT, which was again treated with CRT. Unfortunately, he again suffered both local and distant relapse 11 months after treatment and is currently stable after receiving immunotherapy with rituximab. A second patient with initial bilateral eye involvement developed a singular relapse on the right eye 14 months after radiotherapy. Therefore, the 2‑ and 5‑year LPFS were 93.5 and 88.6 % (Fig. 1a).
a Kaplan–Meier overall survival (OS; n = 45 patients), local progression-free survival (LPFS; n = 52 lesions), and distant progression-free survival (DPFS; n = 45 patients) in conventional radiotherapy (CRT); b Kaplan–Meier OS (n = 7 patients), LPFS (n = 8 lesions), and DPFS (n = 7 patients) in low-dose radiotherapy (LDRT)
Four patients had systemic relapses, resulting in 2‑ and 5‑year distant progression-free survival (DPFS) of 95.0 and 89.9 %, respectively (Fig. 1a). The OS in the CRT cohort was 100 % at 2 years and 85.6 % at 5 years (Fig. 1a).
Two patients had bilateral, metachronous involvement of the orbit: one patient was treated with CRT on both eyes, while the other patient was first treated with CRT, followed by LDRT. Both have been without any signs of relapse on both eyes at the time that this article was written.
Low-dose radiotherapy
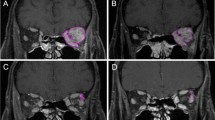
Orbital lymphomas treated with 2 fractions of 2.0 Gy LDRT showed similar response rates (Tab. 3): In the first follow-ups 2 months after radiotherapy, contrast-enhanced MRI scans were performed in all 7 patients and CR was noticed in 3 lesions (37.5 %) and PR in 5 lesions (62.5 %). Interestingly, 4 months later further tumor shrinkage was observed and CR was documented for all 8 lesions (100 %; Fig. 2).
The 2‑year LPFS was 100 % and no local recurrence has been seen in follow-up MRI scans of all 7 patients until October 2015. Both patients with stage III and IV disease, who were treated with LDRT due to local symptoms, suffered from systemic relapses, resulting in a 2‑year DPFS of 68.6 %. The OS in LDRT patients was 100 % at 2 years (Fig. 1b).
Toxicity
Treatment-related toxicity rates in CRT were moderate. Mild transient side effects (grade 1 or 2) in CRT were observed in almost all irradiated eyes with typical symptoms like conjunctivitis, periorbital edema, or dermatitis (Tab. 4). One grade 3–4 acute toxicity (keratitis) was observed. The most common late complication was mild xerophthalmia (39.5 %). Cataracts developed in 25.6 % (11 lesions) of the irradiated eyes, of which 10 lesions were treated with more than 34 Gy. Low-dose radiotherapy was well tolerated with only mild acute and late complications such as xerophthalmia and quick symptom relief after irradiation. No grade 3–4 acute toxicity occurred.
Discussion
Primary radiation therapy with conventional doses (24–36 Gy) is the standard therapy for early stage indolent lymphomas of the orbit resulting in high response rates and outstanding local control [15, 16, 20–22]. Our retrospective analysis shows typical patients characteristics (e. g., older age, typical histology, 10 % bilateral involvement, mostly earlier stages) with similar high response rates (97.8 %) and excellent long-term control rates of 88.6 % 5‑ and 10-year LPFS in CRT. Patients with stage III or IV NHL were additionally treated with local radiotherapy of the orbit, due to a lack of response after chemotherapy/immunotherapy.
Toxicity after CRT is tolerable with an increase of grade 3–4 complications in higher doses. A relatively high cataract incidence of 25 % was observed in our retrospective analysis. All cataracts occurred in patients who received doses higher than 34 Gy. Although a dose dependency in development of cataract is well known [23, 24], treatment independent factors, especially in elderly patients must be considered. Many patients in our analysis either had a history of systemic comorbidities like diabetes mellitus or previous alterations in lens opacity or a history of chronic eye disease (e. g., glaucoma or sicca syndrome). Those predispositions may all independently influence cataract development which makes the relationship to treatment difficult to prove. In addition, the improvement of cataract surgery results in an easily manageable side effect. Furthermore, the evolution of radiation techniques (3D conformal radiotherapy and intensity-modulated radiotherapy) has optimized current treatment and enables homogenous dose delivery and simultaneously precise sparing of organs at risk.
Since indolent lymphomas are highly radiosensitive, low-dose radiation has become increasingly used with promising results regarding local control rates with minimal toxicity. In the first series, published by Ganem et al. [25], 27 patients with 57 sites of lymphoma affection were treated with 2 fractions of 2.0 Gy in a palliative setting with a surprisingly high response rate of 89 %. Several other publications showed comparable results in LDRT even in patients with early stage disease [26–28].
A randomized non-inferiority trial (FORT) compared a conventional radiation scheme (24 Gy) to LDRT (4 Gy) in patients with FL [29]. Despite high response rates and without difference in overall survival in both arms, LDRT remained inferior regarding LPFS [29]. No differentiation in terms of size of affected regions or gradings of FL (study included grade 3b) and inconsistencies concerning the analysis itself are only some pitfalls the study reveals [30]. On account of this, non-inferiority of LDRT of smaller lesions (e. g., orbital lymphoma) is still possible.
Radiotherapy of the orbit may be challenging due to the proximity and sensitivity of surrounding tissues like the lens, lacrimal gland, or retina. Therefore, Fasola et al. [31] treated 27 orbital lymphomas with LDRT. They showed high response rates of 96 % with a 2‑year freedom from local relapse of 100 %, comparable to our data that revealed a similar high response rate of 100 % and 2‑year LPFS of 100 % and no local relapse at the time of manuscript preparation (longest follow-up: 41 months). Although we observed a better 2‑year LPFS and OS after LDRT, the 2‑year DPFS after LDRT was lower compared to CRT (68.6 % vs. 95 %). However, both patients with a systemic relapse after LDRT suffered from higher stages of NHL, which may explain the difference.
Patients receiving LDRT should be closely followed and in the setting of modern and broadly available imaging techniques like MRI scans; another benefit of LDRT also in a curative setting is the fact that further radiotherapy is still possible.
However, the study has its limitations owing to the retrospective analysis and the limited follow-up data and low patient quantity on LDRT. Moreover, a lack of documentation of response and the heterogeneity of radiation treatment techniques in earlier years of CRT are negative aspects of this analysis.
Nevertheless, our data confirm that CRT is a well-established technique with excellent response and control rates in the treatment of orbital lymphomas. Furthermore, LDRT is very convenient for patients due to its short treatment period and the minimal side effects. In addition, we demonstrate that LDRT is effective and—in combination with close follow-up exams—an attractive option also in the curative settings with the possibility of re-irradiation.
By inducing anti-inflammatory effects like activating NF-κB or TGF-β pathway [32], as well as influencing granulocyte/endothelial cell adhesion [33, 34], low-dose radiation is well integrated into the routine treatment of benign diseases. Similar to anti-inflammatory mechanisms, effectiveness in cancer-related diseases is only partly understood: Local effects like overexpression of p53-associated genes, induction of extrinsic and intrinsic apoptosis pathways, and up-regulation of immune reactions via macrophage activation-related genes are induced by LDRT [35]. Besides chemokine induction, activation of dendritic cells and TH1-related genes have been connected to LDRT, suggesting possible systemic (abscopal) effects [35]. Nevertheless, further preclinical studies and clinical prospective studies are needed to elucidate the mechanisms and clinical outcomes of LDRT. We aim to initiate preclinical studies about radiogenic immune responses as well as a multicenter prospective study on LDRT for indolent orbital lymphoma.
Conclusion
Our retrospective analysis shows that primary radiotherapy of indolent orbital lymphomas is a well-established treatment with high response rates and excellent local control in CRT and LDRT. Since radiation-related side effects are more frequent in CRT, LDRT may be a convenient and effective alternative. In combination with close follow-up and modern imaging techniques like MRI, insufficient response or disease progression after LDRT can be detected in a timely manner and re-irradiation after LDRT even with full conventional doses is still feasible. Nevertheless, further prospective trials are needed to prove whether LDRT may also be considered in the curative setting.
References
Freeman C, Berg JW, Cutler SJ (1972) Occurrence and prognosis of extranodal lymphomas. Cancer 29(1):252–260
Fitzpatrick PJ, Macko S (1984) Lymphoreticular tumors of the orbit. Int J Radiat Oncol Biol Phys 10(3):333–340
Margo CE, Mulla ZD (1998) Malignant tumors of the orbit. Analysis of the Florida Cancer Registry. Ophthalmology 105(1):185–190
Ferry JA, Fung CY, Zukerberg L, Lucarelli MJ, Hasserjian RP, Preffer FI, Harris NL (2007) Lymphoma of the ocular adnexa: A study of 353 cases. Am J Surg Pathol 31(2):170–184. doi:10.1097/01.pas.0000213350.49767.46
Fung CY, Tarbell NJ, Lucarelli MJ, Goldberg SI, Linggood RM, Harris NL, Ferry JA (2003) Ocular adnexal lymphoma: clinical behavior of distinct World Health Organization classification subtypes. Int J Radiat Oncol Biol Phys 57(5):1382–1391
Woolf DK, Ahmed M, Plowman PN (2012) Primary lymphoma of the ocular adnexa (orbital lymphoma) and primary intraocular lymphoma. Clin Oncol (R Coll Radiol) 24(5):339–344. doi:10.1016/j.clon.2012.03.001
Ferreri AJ, Dolcetti R, Magnino S, Doglioni C, Ponzoni M (2009) Chlamydial infection: the link with ocular adnexal lymphomas. Nat Rev Clin Oncol 6(11):658–669. doi:10.1038/nrclinonc.2009.147
Ferreri AJ, Ponzoni M, Guidoboni M, De Conciliis C, Resti AG, Mazzi B, Lettini AA, Demeter J, Dell’Oro S, Doglioni C, Villa E, Boiocchi M, Dolcetti R (2005) Regression of ocular adnexal lymphoma after Chlamydia psittaci-eradicating antibiotic therapy. J Clin Oncol 23(22):5067–5073. doi:10.1200/JCO.2005.07.083
Ferreri AJ, Guidoboni M, Ponzoni M, De Conciliis C, Dell’Oro S, Fleischhauer K, Caggiari L, Lettini AA, Dal Cin E, Ieri R, Freschi M, Villa E, Boiocchi M, Dolcetti R (2004) Evidence for an association between Chlamydia psittaci and ocular adnexal lymphomas. J Natl Cancer Inst 96(8):586–594
Cassoux N, Merle-Beral H, Leblond V, Bodaghi B, Milea D, Gerber S, Fardeau C, Reux I, Xuan KH, Chan CC, LeHoang P (2000) Ocular and central nervous system lymphoma: clinical features and diagnosis. Ocul Immunol Inflamm 8(4):243–250
Buggage RR, Chan CC, Nussenblatt RB (2001) Ocular manifestations of central nervous system lymphoma. Curr Opin Oncol 13(3):137–142
Schabet M (1999) Epidemiology of primary CNS lymphoma. J Neurooncol 43(3):199–201
Fakhrian K, Klemm S, Keller U, Bayer C, Riedl W, Molls M, Geinitz H (2012) Radiotherapy in stage I–III follicular non-Hodgkin lymphoma. Retrospective analysis of a series of 50 patients. Strahlenther Onkol 188(6):464–470. doi:10.1007/s00066-011-0057-0
Heinzelmann F, Engelhard M, Ottinger H, Bamberg M, Weinmann M (2010) Nodal follicular lymphoma: the role of radiotherapy for stages I and II. Strahlenther Onkol 186(4):191–196. doi:10.1007/s00066-010-2090-9
Le QT, Eulau SM, George TI, Hildebrand R, Warnke RA, Donaldson SS, Hoppe RT (2002) Primary radiotherapy for localized orbital MALT lymphoma. Int J Radiat Oncol Biol Phys 52(3):657–663 ([pii]) doi:10.1016/s0360-3016(01)02729-8
Bolek TW, Moyses HM, Marcus RB Jr., Gorden L 3rd, Maiese RL, Almasri NM, Mendenhall NP (1999) Radiotherapy in the management of orbital lymphoma. Int J Radiat Oncol Biol Phys 44(1):31–36 ([pii]) doi:10.1016/s0360-3016(98)00535-5
Stafford SL, Kozelsky TF, Garrity JA, Kurtin PJ, Leavitt JA, Martenson JA, Habermann TM (2001) Orbital lymphoma: radiotherapy outcome and complications. Radiother Oncol 59(2):139–144. doi:10.1016/s0167-8140(00)00328-5
Bischof M, Karagiozidis M, Krempien R, Treiber M, Neuhof D, Debus J, Zierhut D (2007) Radiotherapy for orbital lymphoma : outcome and late effects. Strahlenther Onkol 183(1):17–22. doi:10.1007/s00066-007-1627-z
Minehan KJ, Martenson JA Jr., Garrity JA, Kurtin PJ, Banks PM, Chen MG, Earle JD (1991) Local control and complications after radiation therapy for primary orbital lymphoma: a case for low-dose treatment. Int J Radiat Oncol Biol Phys 20(4):791–796. doi:10.1016/0360-3016(91)90025-y
Bhatia S, Paulino AC, Buatti JM, Mayr NA, Wen BC (2002) Curative radiotherapy for primary orbital lymphoma. Int J Radiat Oncol Biol Phys 54(3):818–823. doi:10.1016/s0360-3016(02)02966-8
Zhou P, Ng AK, Silver B, Li S, Hua L, Mauch PM (2005) Radiation therapy for orbital lymphoma. Int J Radiat Oncol Biol Phys 63(3):866–871. doi:10.1016/j.ijrobp.2005.03.005
Esik O, Ikeda H, Mukai K, Kaneko A (1996) A retrospective analysis of different modalities for treatment of primary orbital non-Hodgkin’s lymphomas. Radiother Oncol 38(1):13–18. doi:10.1016/0167-8140(95)01658-9
Ferrufino-Ponce ZK, Henderson BA (2006) Radiotherapy and cataract formation. Semin Ophthalmol 21(3):171–180. doi:10.1080/08820530500351728
Ainsbury EA, Bouffler SD, Dorr W, Graw J, Muirhead CR, Edwards AA, Cooper J (2009) Radiation cataractogenesis: a review of recent studies. Radiat Res 172(1):1–9. doi:10.1667/RR1688.1
Ganem G, Lambin P, Socie G, Girinsky T, Bosq J, Pico JL, Solal-Celigny P, Cosset JM (1994) Potential role for low dose limited-field radiation therapy (2 × 2 grays) in advanced low-grade non-Hodgkin’s lymphomas. Hematol Oncol 12(1):1–8
Haas RL, Poortmans P, de Jong D, Aleman BM, Dewit LG, Verheij M, Hart AA, van Oers MH, van der Hulst M, Baars JW, Bartelink H (2003) High response rates and lasting remissions after low-dose involved field radiotherapy in indolent lymphomas. J Clin Oncol 21(13):2474–2480. doi:10.1200/JCO.2003.09.542
Ganem G, Cartron G, Girinsky T, Haas RL, Cosset JM, Solal-Celigny P (2010) Localized low-dose radiotherapy for follicular lymphoma: history, clinical results, mechanisms of action, and future outlooks. Int J Radiat Oncol Biol Phys 78(4):975–982 (S0360-3016(10)00936-3 [pii]) doi:10.1016/j.ijrobp.2010.06.056
Russo AL, Chen YH, Martin NE, Vinjamoori A, Luthy SK, Freedman A, Michaelson EM, Silver B, Mauch PM, Ng AK (2013) Low-dose involved-field radiation in the treatment of non-hodgkin lymphoma: predictors of response and treatment failure. Int J Radiat Oncol Biol Phys 86(1):121–127. doi:10.1016/j.ijrobp.2012.12.024
Hoskin PJ, Kirkwood AA, Popova B, Smith P, Robinson M, Gallop-Evans E, Coltart S, Illidge T, Madhavan K, Brammer C, Diez P, Jack A, Syndikus I (2014) 4 Gy versus 24 Gy radiotherapy for patients with indolent lymphoma (FORT): a randomised phase 3 non-inferiority trial. Lancet Oncol 15(4):457–463. doi:10.1016/S1470-2045(14)70036-1
Herfarth K, Konig L (2014) Radiation therapy (4 Gy vs. 24 Gy) in patients with indolent non-Hodgkins lymphoma: Results of the FORT Study. Strahlenther Onkol 190(11):1084–1085
Fasola CE, Jones JC, Huang DD, Le QT, Hoppe RT, Donaldson SS (2013) Low-dose radiation therapy (2 Gy x 2) in the treatment of orbital lymphoma. Int J Radiat Oncol Biol Phys 86(5):930–935. doi:10.1016/j.ijrobp.2013.04.035
Rodel F, Schaller U, Schultze-Mosgau S, Beuscher HU, Keilholz L, Herrmann M, Voll R, Sauer R, Hildebrandt G (2004) The induction of TGF-beta(1) and NF-kappaB parallels a biphasic time course of leukocyte/endothelial cell adhesion following low-dose X‑irradiation. Strahlenther Onkol 180(4):194–200. doi:10.1007/s00066-004-1237-y
Rodel F, Hofmann D, Auer J, Keilholz L, Rollinghoff M, Sauer R, Beuscher HU (2008) The anti-inflammatory effect of low-dose radiation therapy involves a diminished CCL20 chemokine expression and granulocyte/endothelial cell adhesion. Strahlenther Onkol 184(1):41–47. doi:10.1007/s00066-008-1776-8
Large M, Hehlgans S, Reichert S, Gaipl US, Fournier C, Rodel C, Weiss C, Rodel F (2015) Study of the anti-inflammatory effects of low-dose radiation: The contribution of biphasic regulation of the antioxidative system in endothelial cells. Strahlenther Onkol 191(9):742–749. doi:10.1007/s00066-015-0848-9
Knoops L, Haas R, de Kemp S, Majoor D, Broeks A, Eldering E, de Boer JP, Verheij M, van Ostrom C, de Vries A, van’t VL, de Jong D (2007) In vivo p53 response and immune reaction underlie highly effective low-dose radiotherapy in follicular lymphoma. Blood 110(4):1116–1122. doi:10.1182/blood-2007-01-067579
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
L. König, R. Stade, J. Rieber, J. Debus, and K. Herfarth state that there are no conflicts of interest.
This analysis was carried out with the approval of the responsible ethics committee of the University of Heidelberg and in accordance with national law and the Helsinki Declaration in its current, revised form.
The accompanying manuscript does not include studies on humans or animals.
Rights and permissions
About this article
Cite this article
König, L., Stade, R., Rieber, J. et al. Radiotherapy of indolent orbital lymphomas. Strahlenther Onkol 192, 414–421 (2016). https://doi.org/10.1007/s00066-016-0962-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-016-0962-3
Keywords
- Low-dose radiotherapy
- Indolent non-Hodgkin lymphoma
- Orbital lymphoma
- Treatment outcome
- Local progression-free survival