Background and Purpose:
In patients with head-and-neck cancer treated with chemoradiotherapy (CRT), a cisplatin-based regimen is often used. Several treatment schedules are accepted with a cumulative cisplatin dose of 200 mg/m2 (CisCD200) given during radiotherapy. The aim of this analysis was to investigate feasibility and efficacy of a weekly cisplatin 40 mg/m2 regimen.
Patients and Methods:
During 08/2001 and 12/2006, 103 patients with squamous head-and-neck cancer received concurrent CRT with intended weekly cisplatin 40 mg/m2 and were analyzed retrospectively. CRT was definitive for a newly diagnosed primary in 62, postoperative in 16, and for recurrence in 25 patients. Most patients had carcinoma of the hypo- and oropharynx (81%). Patients received a median total dose of 70 Gy (range, 42–71.2 Gy).
Results:
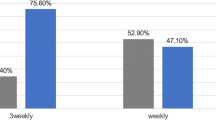
Only 42 patients (41%) received a CisCD200 predominantly due to hematotoxicity. Actuarial 12- and 18-month overall survival (OS) for patients with and without CisCD200 was 83.3% versus 72.1% (p = 0.19) and 66.7% versus 67.2% (p = 0.86), the 12- and 18-month locoregional control (LRC) 66.7% versus 78.7% (p = 0.325) and 59.5% versus 78.7% (p = 0.109), respectively. Multivariate analysis revealed only type of CRT (definitive vs. recurrent) and T-classification as significant variables predicting OS and LRC.
Conclusion:
Feasibility and efficacy of CRT with weekly cisplatin 40 mg/m2 were suboptimal in this analysis. However, the prospects of weekly cisplatin may be its more suitable integration into emerging trimodality concepts combining CRT with molecularly targeted agents.
Hintergrund und Ziel:
Bei der Radiochemotherapie (CRT) von Patienten mit Kopf-Hals-Tumoren wird haufig eine platinhaltige Chemotherapie gewahlt. Verschiedene Behandlungsschemata zielen auf eine kumulative Dosis von mindestens 200 mg/m2 Cisplatin (CisCD200) simultan zur Bestrahlung. Ziel dieser Studie war es, die Wirksamkeit und Vertraglichkeit wochentlicher Cisplatingaben von 40 mg/m2 zu untersuchen.
Patienten und Methodik:
Im Zeitraum von 08/2001 bis 12/2006 erhielten 103 Patienten mit Kopf-Hals-Tumoren eine CRT mit geplanten wöchentlichen Cisplatingaben von 40 mg/m2 und wurden retrospektiv ausgewertet. 62 Patienten bekamen eine definitive CRT bei primar diagnostizierten Tumoren, 16 Patienten wurden postoperativ und 25 Patienten in einer Rezidivsituation behandelt. Hypo- oder Oropharynxkarzinome lagen in 81% (Tabelle 1) der Falle vor. Die mediane Gesamtdosis betrug 70 Gy (Streubreite 42–71,2 Gy, Tabelle 2).
Ergebnisse:
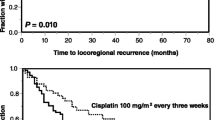
Nur 42 Patienten (41%) erreichten die CisCD200 (Tabelle 3), hauptsachlich aufgrund erhohter Hamatotoxizitat. Das Gesamtuberleben (OS) nach 12 und 18 Monaten (Abbildung 1) betrug fur die Patienten mit und ohne CisCD200 83,3% versus 72,1% (p = 0,19) und 66,7% versus 67,2% (p = 0,86). Die lokoregionare Kontrolle (LRC) betrug nach 12 und 18 Monaten (Abbildung 2) 66,7% versus 78,7% (p = 0,325) und 59,5% versus 78,7% (p = 0,109; Tabelle 4). In der Multivarianzanalyse hatten nur die Behandlungsart der CRT (Primar- vs. Rezidivbehandlung, Abbildung 3) und das T-Stadium signifikanten Einfluss auf OS und LRC (Tabelle 5).
Schlussfolgerung:
Die Durchfuhrbarkeit und Wirksamkeit der CRT mit wochentlichen Cisplatingaben erscheinen in dieser Analyse suboptimal. Der Vorteil wochentlicher Cisplatingaben liegt vor allem in der besseren Integrierbarkeit in Konzepte, die eine CRT mit einer zielgerichteten Therapie verbinden.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adelstein DJ, Li Y, Adams GL, et al. An Intergroup phase III comparison of standard radiation therapy and two schedules of concurrent chemoradiotherapy in patients with unresectable squamous cell head and neck cancer. J Clin Oncol 2003;21:92–8.
Adelstein DJ, Saxton JP, Rybicki LA, et al. Multiagent concurrent chemoradiotherapy for locoregionally advanced squamous cell head and neck cancer: mature results from a single institution. J Clin Oncol 2006;24:1064–71.
Al-Sarraf M, LeBlanc M, Giri PG, et al. Chemoradiotherapy versus radiotherapy in patients with advanced nasopharyngeal cancer: phase III randomized Intergroup study 0099. J Clin Oncol 1998;6:1310–7.
Bachaud JM, Cohen-Jonathan E, Alzieu C, et al. Combined postoperative radiotherapy and weekly cisplatin infusion for locally advanced head and neck carcinoma: final report of a randomized trial. Int J Radiat Oncol Biol Phys 1996;36:999–1004.
Beckmann GK, Hoppe F, Pfreundner L, et al. Hyperfractionated accelerated radiotherapy in combination with weekly cisplatin for locally advanced head and neck cancer. Head Neck 2005;27:36–43.
Bernier J, Domenge C, Ozsahin M, et al. European Organization for Research and Treatment of Cancer trial 22931. Postoperative irradiation with or without concomitant chemotherapy for locally advanced head and neck cancer. N Engl J Med 2004;350:1945–52.
Bölke E, Gerber PA, Lammering G, et al. Development and management of severe cutaneous side effects in head-and-neck cancer patients during concurrent radiotherapy and cetuximab. Strahlenther Onkol 2008;184:105–10.
Bottke D, Bathe K, Wiegel T, et al. Phase I trial of radiochemotherapy with bendamustine in patients with recurrent squamous cell carcinoma of the head and neck. Strahlenther Onkol 2007;183:128–32.
Bourhis J, Le Maître A, Baujat B, et al., Meta-Analysis of Chemotherapy in Head, Neck Cancer Collaborative Group, Meta-Analysis of Radiotherapy in Carcinoma of Head, Neck Collaborative Group, Meta-Analysis of Chemotherapy in Nasopharynx Carcinoma Collaborative Group. Individual patients’ data meta-analyses in head and neck cancer. Curr Opin Oncol 2007;19:188–94.
Browman GP, Hodson DI, Mackenzie RJ, et al., Cancer Care Ontario Practice Guideline Initiative Head and Neck Cancer Disease Site Group. Choosing a concomitant chemotherapy and radiotherapy regimen for squamous cell head and neck cancer: a systematic review of the published literature with subgroup analysis. Head Neck 2001;23:579–89.
Cancer Therapy Evaluation Program. Common Terminology Criteria for Adverse Events, Version 3.0, DCTD, NCI, NIH, DHHS March 31, 2003 (http://ctep.cancer.gov). Bethesda, MD, USA: National Cancer Institute, August 9, 2006
Cooper JS, Pajak TF, Forastiere AA, et al. Radiation Therapy Oncology Group 9501/Intergroup. Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck. N Engl J Med 2004;350:1937–44.
Eich HT, Löschcke M, Scheer M, et al. Neoadjuvant radiochemotherapy and radical resection for advanced squamous cell carcinoma of the oral cavity. Outcome of 134 patients. Strahlenther Onkol 2008;184:23–9.
Einstein MH, Novetsky AP, Garg M, et al. Survival and toxicity differences between 5-day and weekly cisplatin in patients with locally advanced cervical cancer. Cancer 2007;109:48–53.
Forastiere AA, Goepfert H, Maor M, et al. Concurrent chemotherapy and radiotherapy for organ preservation in advanced laryngeal cancer. N Engl J Med 2003;349:2091–8.
Green J, Kirwan J, Tierney J, et al. Concomitant chemotherapy and radiation therapy for cancer of the uterine cervix. Cochrane Database Syst Rev 2005;3:CD002225.
Ho KF, Swindell R, Brammer CV. Dose intensity comparison between weekly and 3-weekly cisplatin delivered concurrently with radical radiotherapy for head and neck cancer: a retrospective comparison from New Cross Hospital, Wolverhampton, UK. Acta Oncol 2008;47:1513–8.
Jeremic B, Milicic B, Dagovic A, et al. Radiation therapy with or without concurrent low-dose daily chemotherapy in locally advanced, nonmetastatic squamous cell carcinoma of the head and neck. J Clin Oncol 2004;22:3540–8.
Kuhnt T, Sandner A, Wendt TG, et al. Concomitant hyperfractionated accelerated radiotherapy (HART) with cisplatin and concurrent cetuximab for locoregionally advanced squamous cell head and neck cancer: a phase I dose escalation trial. J Clin Oncol 2008;26:abstract 6029.
Marcial VA, Pajak TF, Mohiuddin M, et al. Concomitant cisplatin chemotherapy and radiotherapy in advanced mucosal squamous cell carcinoma of the head and neck. Long-term results of the Radiation Therapy Oncology Group study 81-17. Cancer 1990;66:1861–8.
Pfister DG, Su YB, Kraus DH, et al. Concurrent cetuximab, cisplatin, and concomitant boost radiotherapy for locoregionally advanced, squamous cell head and neck cancer: a pilot phase II study of a new combined-modality paradigm. J Clin Oncol 2006;24:1072–8.
Pignon JP, le Maître A, Bourhis J, MACH-NC Collaborative Group. Meta-Analyses of Chemotherapy in Head and Neck Cancer (MACH-NC): an update. Int J Radiat Oncol Biol Phys 2007;69:112–4.
Rades D, Fehlauer F, Sheikh-Sarraf M, et al. Toxicity of two cisplatin-based radiochemotherapy regimens for the treatment of patients with stage III/IV head and neck cancer. Head Neck 2008;30:235–41.
Rades D, Stoehr M, Meyners T, et al. Evaluation of prognostic factors and two radiation techniques in patients treated with surgery followed by radio(chemo)therapy or definitive radio(chemo)therapy for locally advanced head-and-neck cancer. Strahlenther Onkol 2008;184:198–205.
Torres MA, Jhingran A, Thames HD Jr, et al. Comparison of treatment tolerance and outcomes in patients with cervical cancer treated with concurrent chemoradiotherapy in a prospective randomized trial or with standard treatment. Int J Radiat Oncol Biol Phys 2008;70:118–25.
Wolff HA, Overbeck T, Rödel R, et al. Effektivität und Toxizität niedrig - do sierter, täglicher Cisplatin-Applikation (6 mg/m2 Körperoberfläche) bei der kombinierten Radiochemotherapie lokal fortgeschrittener Kopf-Hals-Tumore. Strahlenther Onkol 2008;184:Sondernr 1:134.
Author information
Authors and Affiliations
Corresponding author
Additional information
*Both authors contributed equally to the study.
Rights and permissions
About this article
Cite this article
Steinmann, D., Cerny, B., Karstens, J.H. et al. Chemoradiotherapy with Weekly Cisplatin 40 mg/m2 in 103 Head-and-Neck Cancer Patients. Strahlenther Onkol 185, 682–688 (2009). https://doi.org/10.1007/s00066-009-1989-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-009-1989-5