Purpose:
To evaluate the impact of neoadjuvant hormonal therapy (NHT) on quality of life after external-beam radiotherapy (EBRT) for prostate cancer.
Patients and Methods:
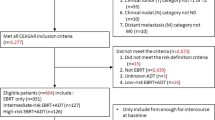
A group of 170 patients (85 with and 85 without NHT) has been surveyed prospectively before EBRT (70.2–72 Gy), at the last day of EBRT, a median time of 2 months and 15 months after EBRT using a validated questionnaire (Expanded Prostate Cancer Index Composite). Pairs with and without NHT (median treatment time of 3.5 months before EBRT) were matched according to the respective planning target volume and prostate volume.
Results:
Before EBRT, significantly lower urinary function/bother, sexual function and hormonal function/bother scores were found for patients with NHT. More than 1 year after EBRT, only sexual function scores remained lower. In a multivariate analysis, NHT and adjuvant hormonal therapy (HT) versus NHT only (hazard ratio 14; 95% confidence interval 2.7–183; p = 0.02) and luteinizing hormone-releasing hormone (LHRH) agonists versus antiandrogens (hazard ratio 3.6; 95% confidence interval 1.1–12; p = 0.04) proved to be independent risk factors for long-term erectile dysfunction (no or very poor ability to have an erection).
Conclusion:
With the exception of sexual function (additional adjuvant HT and application of LHRH analog independently adverse), short-term NHT was not found to decrease quality of life after EBRT for prostate cancer.
Ziel:
Untersuchung des Einflusses einer neoadjuvanten Hormontherapie (NHT) auf die Lebensqualität nach perkutaner Radiotherapie (EBRT) beim Prostatakarzinom.
Patienten und Methodik:
In einer Gruppe von 170 Patienten (85 mit und 85 ohne NHT) wurde die Lebensqualität prospektiv vor EBRT (70,2–72 Gy), am letzten Tag der EBRT, median 2 Monate und 15 Monate nach EBRT mittels eines validierten Fragebogens (Expanded Prostate Cancer Index Composite) erfasst. Paare mit und ohne NHT (median 3,5 Monate vor EBRT verabreicht) wurden entsprechend dem jeweiligen Planungszielvolumen und Prostatavolumen gematcht.
Ergebnisse:
Vor EBRT zeigten sich bei Patienten mit NHT signifikant schlechtere Funktions- und Belastungswerte in den Domänen Wasserlassen, Sexualität und hormonelle Beschwerden. Über 1 Jahr nach Ende der EBRT blieb nur noch die sexuelle Funktion signifikant schlechter. In einer multivariaten Analyse zeigte sich der unabhängige Einfluss der NHT mit adjuvanter Hormontherapie (HT) versus alleiniger NHT (relatives Risiko 14; 95%-Konfidenzintervall 2,7–183; p = 0,02) und der Gonadotropin-releasing-Hormon-(GnRH-)Analoga versus Antiandrogene (relatives Risiko 3,6; 95%-Konfidenzintervall 1,1–12; p = 0,04) als Risikofaktoren für die längerfristige erektile Dysfunktion (keine oder sehr geringe Erektionsfähigkeit).
Schlussfolgerung:
Mit Ausnahme des Einflusses auf die sexuellen Funktionswerte (zusätzliche adjuvante HT und Verwendung von GnRH-Analoga unabhängig voneinander nachteilig) zeigte die kurzzeitige NHT keine negativen Auswirkungen auf die Lebensqualität nach EBRT des Prostatakarzinoms.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References and Recommended Reading
Bolla M, Collette L, Blank L, et al. Long-term results with immediate androgen suppression and external irradiation in patients with locally advanced prostate cancer (an EORTC study): a phase III randmised trial. Lancet 2002;360:103–8.
Bolla M, Gonzalez D, Warde P, et al. Improved survival in patients with locally advanced prostate cancer treated with radiotherapy and goserelin. N Engl J Med 1997;337:295–300.
Crook J, Ludgate C, Malone S, et al. Report of a multicenter Canadian phase III randomized trial of 3 months vs. 8 months neoadjuvant androgen deprivation before standard-dose radiotherapy for clinically localized prostate cancer. Int J Radiat Oncol Biol Phys 2004;60:15–23.
Crook J, McLean M, Catton C, et al. Factors influencing risk of acute urinary retention after TRUS-guided permanent prostate seed implantation. Int J Radiat Oncol Biol Phys 2002;52:453–60.
D’Amico AV. Toward the optimal use of androgen suppression therapy in the radiotherapeutic management of prostate cancer. J Clin Oncol 2007;25: 8–9.
D’Amico AV, Denham JW, Crook J, et al. Influence of androgen suppression therapy for prostate cancer on the frequency and timing of fatal myocardial infarctions. J Clin Oncol 2007;25:2420–5.
D’Amico AV, Manola J, Loffredo M, et al. 6-month androgen suppression plus radiation therapy vs radiation therapy alone for patients with clinically localized prostate cancer: a randomized controlled trial. JAMA 2004;292:821–7.
Davis JW, Kuban DA, Lynch DF, et al. Quality of life after treatment for localized prostate cancer: differences based on treatment modality. J Urol 2001;166:947–52.
Elsayed H, Bolling T, Moustakis C, et al. Organ movements and dose exposures in teletherapy of prostate cancer using a rectal balloon. Strahlenther Onkol 2007;183:617–24.
Feigenberg SJ, Hanlon AL, Horwitz EM, et al. Long-term androgen deprivation increases grade 2 and higher late morbidity in prostate cancer patients treated with three-dimensional conformal radiation therapy. Int J Radiat Oncol Biol Phys 2005;62:397–405.
Guckenberger M, Flentje M. Intensity-modulated radiotherapy (IMRT) of localized prostate cancer. A review and future perspectives. Strahlenther Onkol 2007;183:57–62.
Hanks GE, Pajak TF, Porter A, et al. Phase III trial of long-term adjuvant androgen deprivation after neoadjuvant hormonal cytoreduction and radiotherapy in locally advanced carcinoma of the prostate: the Radiation Therapy Oncology Group protocol 92-02. J Clin Oncol 2003;21:3972–8.
Higano C, Shields A, Wood N. Bone mineral density in patients with prostate cancer without bone metastases treated with intermittent androgen suppression. Urology 2004;64:1182–6.
International Commission on Radiation Units and Measurements (ICRU). Prescribing, recording and reporting photon beam therapy. ICRU report 50. Bethesda: ICRU, 1993:1–8.
Jereczek-Fossa BA, Cattani F, Garibaldi C, et al. Transabdominal ultrasonography, computed tomography and electronic portal imaging for 3-dimensional conformal radiotherapy for prostate cancer. Strahlenther Onkol 2007;183:610–6.
Lawton CA, DeSilvio M, Roach MIII, et al. An update of the phase III trial comparing whole pelvic to prostate only radiotherapy and neoadjuvant to adjuvant total androgen suppression: updated analysis of RTOG 94-13, with emphasis on unexpected hormone/radiation interactions. Int J Radiat Oncol Biol Phys 2007;69:646–55.
Lilleby W, Dale E, Olsen DR, et al. Changes in treatment volume of hormonally treated and untreated cancerous prostate and its impact on rectal dose. Acta Oncol 2003;42:10–4.
Merrick GS, Butler WM, Wallner KE, et al. Long-term urinary quality of life after permanent prostate brachytherapy. Int J Radiat Oncol Biol Phys 2003;56:454–61.
Merrick GS, Butler WM, Wallner KE, et al. Influence of hormonal therapy on late rectal function after permanent prostate brachytherapy with or without supplemental external beam radiotherapy. Int J Radiat Oncol Biol Phys 2004;58:68–74.
Muller AC, Ganswindt U, Bamberg M, et al. Risk of second malignancies after prostate irradiation? Strahlenther Onkol 2007;183:605–9.
Ososba D, Rodrigues G, Myles J, et al. Interpreting the significance of changes in health-related quality-of-life scores. J Clin Oncol 1998;16:139–44.
Padula GDA, Zelefsky MJ, Venkatraman ES, et al. Normalization of serum testosterone levels in patients treated with neoadjuvant hormonal therapy and three-dimensional conformal radiotherapy for prostate cancer. Int J Radiat Oncol Biol Phys 2002;52:439–43.
Pilepich MV, Winter K, Lawton CA, et al. Androgen suppression adjuvant to definitive radiotherapy in prostate carcinoma — long-term results of phase III RTOG 85-31. Int J Radiat Oncol Biol Phys 2005;61:1285–90.
Pinkawa M, Fischedick K, Asadpour B, et al. Toxicity profile with a large prostate volume after external beam radiotherapy for localized prostate cancer. Int J Radiat Oncol Biol Phys 2008;70:83–9.
Pinkawa M, Fischedick K, Gagel B, et al. Association of neoadjuvant hormonal therapy with adverse health-related quality of life after permanent iodine-125 brachytherapy for localized prostate cancer. Urology 2006;68:104–9.
Pinkawa M, Fischedick K, Piroth MD, et al. Health-related quality of life after permanent interstitial brachytherapy for prostate cancer. Correlation with postimplant CT scan parameters. Strahlenther Onkol 2006;182:660–5.
Pinkawa M, Fischedick K, Piroth MD, et al. Prostate-specific antigen kinetics after brachytherapy or external beam radiotherapy and neoadjuvant hormonal therapy. Urology 2007;69:129–33.
Pinkawa M, Gagel B, Piroth MD, et al. Changes of dose delivery distribution within the first month after permanent interstitial brachytherapy for prostate cancer. Strahlenther Onkol 2006;182:525–30.
Potters L, Torre T, Fearn PA, et al. Potency after permanent prostate brachytherapy for localized prostate cancer. Int J Radiat Oncol Biol Phys 2001;50:1235–42.
Roach MIII, Bae K, Speight J, et al. Short-term neoadjuvant androgen deprivation therapy and external-beam radiotherapy for locally advanced prostate cancer: long-term results of RTOG 8610. J Clin Oncol 2008;26:585–91.
Schultheiss TE, Lee WR, Hunt MA, et al. Late GI and GU complications in the treatment of prostate cancer. Int J Radiat Oncol Biol Phys 1997;37:3–11.
Smith MR. Changes in fat and lean body during androgen-deprivation therapy for prostate cancer. Urology 2004;63:742–5.
Vordermark D, Marold D, Wirth S, et al. Patterns of care in the radiotherapy of prostate cancer in Northern Bavaria 1998–2000. Strahlenther Onkol 2007;183:314–20.
Wei JT, Dunn RL, Litwin MS, et al. Development and validation of the expanded prostate cancer index composite (EPIC) for comprehensive assessment of health-related quality of life in men with prostate cancer. Urology 2000;56:899–905.
Weiss W, Horninger W, Forthuber BC, et al. Single-institution results of primary external-beam radiation for the treatment of T1–T3 prostate cancer. Strahlenther Onkol 2007;183:321–6.
Wertz H, Lohr F, Dobler B, et al. Dosimetric impact of image-guided translational isocenter correction for 3-D conformal radiotherapy of the prostate. Strahlenther Onkol 2007;183:203–10.
Wilke DR, Parker C, Andonowski A, et al. Testosterone and erectile function recovery after radiotherapy and long-term androgen deprivation with luteinizing hormone-releasing hormone agonists. BJU Int 2006;97:963–8.
Wirth MP, Hakenberg OW, Froehner M. Antiandrogens in the treatment of prostate cancer. Eur Urol 2007;51:306–14.
Zelefsky MJ, Harrison A. Neoadjuvant androgen ablation prior to radiotherapy for prostate cancer: reducing the potential morbidity of therapy. Urology 1997;49:38–45.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pinkawa, M., Piroth, M.D., Asadpour, B. et al. Neoadjuvant hormonal therapy and external-beam radiotherapy versus external-beam irradiation alone for prostate cancer. Strahlenther Onkol 185, 101–108 (2009). https://doi.org/10.1007/s00066-009-1894-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-009-1894-y