Abstract
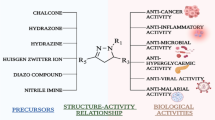
In the last few years, the pyridazine derivatives have been found to exhibit a wide range of pharmacological activities. A large number of research articles and patents described them, and several drugs based on its nucleus have come into light. Pyridazinone, a derivative of pyridazine, was initially exploited in search of cardiovascular drugs and for its use in agrochemicals, but later on this nucleus was found to be associated with plethora of activities. In this article, we have reviewed the influence of structural changes on the pharmacodynamic profile of the pyridazinone moiety.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pyridazines are the organic compounds derived by replacing the two carbons in the benzene ring by nitrogen atoms. This kind of replacement can have three isomeric forms depending on the position of nitrogen with respect to each other in the ring, which can be 1–2, 1–3, or 1–4 relationship, giving rise to the pyridazines (1), pyrimidine (2), and pyrazine (3), respectively. Out of these pyridazinone are those category of pyridazine which contain nitrogen atoms at positions 1 and 2 in the ring along with a keto functionality (Asif, 2010). In the past, a lot of research work has been done on pyridazinones. This nucleus is also known as “wonder nucleus” as it has given many compounds with a varied range of pharmacodynamic profile (Abubshait, 2007; Coudert et al., 1991; Heinisch and Frank, 1990; Pahernik et al., 1995; Sotelo et al., 2000; Wang et al., 2007). A variety of approaches to use pyridazinone system in drug design have been well described in number of recent articles (Asif, 2012, 2013a, b; Bansal and Thota, 2013; Lee et al., 2004).

A substantial number of pyridazinones in the recent past have been reported to possess antimicrobial (Kassab, 2002; Mojahidul et al., 2008; Sayed et al., 2002), antitubercular (Sayed et al., 2002), analgesic (Piaz et al., 1996; Giovannoni et al., 2003; Gokce et al., 2001; Dogruer et al., 2000; Biancalani et al., 2009a, b; Pieretti et al., 1999; Rubat et al., 1992), anti-inflammatory (Hallas, 1995; Takaya et al., 1979; Dogruer et al., 2003; Flouzat et al., 1993; Gökce et al., 2009; Santagati et al., 1985a, b; Sato et al., 1981; Raskin, 1999; Cignarella et al., 1978; Deniz and Fethi, 2003; Banoglu et al., 2004; Ayla et al., 2003), cyclooxygenase inhibitor (Okcelik et al., 2003; Chintakunta et al., 2002), antidiabetic (Rathish et al., 2009), antihypertensive (Barbaro et al., 2001; Seki et al., 1996; Siddiqui and Wani, 2004; Bansal et al., 2009; Verdouw et al., 1986; Korvald et al., 2002; Rüegg et al., 1984; Schneider et al., 1979; Schudt et al., 1991; Ranju et al., 2008; Kubo, 1997; Kumar et al., 2008; Wang et al., 2008; Uhlmann et al., 1995; Bowman et al., 1999; Ishimori et al., 1994; Iwamoto, 1998), antiplatelet (Cherng et al., 2006; Coelho et al., 2004; Sotelo et al., 2002; Zeng et al., 1993; Mikashima et al., 1984; Pham et al., 1988), anticancer (Miguel et al., 2005; Malinka et al., 2004), antifungal (Sayed et al., 2002; Siddiqui et al., 2008; Anwair et al., 2003), antidepressant–anxiolytic (Griebel et al., 1999), anticonvulsant (Xu et al., 1991), bronchodilatory (for asthma) and anti-allergic (Nagai et al., 1992; Yamamoto et al., 1995; Hibi et al., 1989), antifeedant (Cao et al., 2003), inhibition of linolenic acid (Willemot, 1997), activity for neurological disorders (Sato, 1979; Laura et al., 2006), and many other properties.
Some of the major pyridazinone derivatives which have appeared in market are indolidan (4) (Abouzid and Bekhit, 2008), bemoradan (Combs et al., 1990), pimobendan (5) (Robertson et al., 1987a, b), levosimendan (6) as antihypertensive (Archan and Toller, 2008), minaprine as antidepressant (Sotelo et al., 2003), emorfazone (7) as anti-inflammatory (Bowman et al., 1999), and azanrinone as a cardiotonic (Siddiqui and Wani, 2004).




In this review, we explore the various substitutions and modifications in the derivatives possessing pyridazinone moiety and exhibiting various pharmacological activities.
Antihypertensive
It was observed from the literature that among the oldest activity of pyridazinone nucleus is its antihypertensive activity. The pyridazinone derivatives exhibit antihypertensive activity by virtue of their vasorelaxant property. In 1980s, 6-arylpyridazinones were synthesized, and the compounds such as 7-fluoro and 5-keto-5H-indeno(1,2-c)pyridazines (8) (Cignarella et al., 1982) were found to be antihypertensive. Around the same time, many other pyridazinone derivatives were synthesized and evaluated for various pharmacodynamic properties. Few of those compounds were found to posses antiplatelet aggregating activity along with antihypertensive activity, e.g., 7-amino or 7-acetyl-aminosubstituted-4,4a-dihydro-5H-indeno(1,2-c)pyridazin-3-ones (9, 10) (Cignarella et al., 1986, 1998) and 6-[p-[(chloroalkanoyl)amino]phenyl]-4,5-dihydo-3(2H)-pyridazinones (11) (Thyes et al., 1983). Some 2-non-substituted/2-methyl-/2-(2-acetyloxyethyl)-6-[4-(substitutedpyrrol-1-yl)phenyl]-4,5-dihydro-3(2H) pyridazinone derivatives (12) were also synthesized and evaluated for their vasorelaxant effect. Some of these compounds have shown good activity on isolated rat aorta (Demirayak et al., 2004a, b).





Siddiqui et al. (2010) synthesized various 6-(substituted phenyl)-2-(substituted methyl)-4,5-dihydropyridazin-3(2H)-ones (13). These compounds were compared for their antihypertensive property against hydralazine, as standard drug using tail cuff method. Compounds containing piperazine or its derivatives attached through nitrogen and bearing phenyl ring substituted with CH3, C2H5, or OCH3 were found to exhibit highly significant reduction in mean arterial BP. But the doses responsible for this effect were higher in comparison with hydralazine. Based on these results, it was inferred that groups such as p-CH3 and p-C2H5 in phenyl ring at position 6 increase the activity, whereas the various cyclic secondary amines at position 2 in a methylene group have no influence on the activity. The influence on nitrogen-containing group at position 2 was also observed, and it was found out that N-piperazine, N-phenothiazine, N-(1,2,4-triazole), N-morpholine, N-(4-N-methylpiperazine), and N-pyrrolidine moieties when present in the structure showed good antihypertensive activity.

Adrenoceptor antagonists
The adrenoceptors (AR) which belong to superfamily of G-protein-coupled receptors are categorized into two types α1 and α2. They have many common structural features which could relate to the similarity in their mechanism of action (Strader et al., 1989a, b). α1 AR are postsynaptically located, whereas α2 AR are located in presynaptic neuronal junction. Due to their importance in the treatment for hypertension (Curran and Ross, 1974) and benign prostatic hypertrophy (BPH; Hieble et al., 1986), the search for new selective α1-adrenoceptor antagonists has increased. The antihypertensive activity of compounds depends on post-junctional α1-adrenoceptor blockage which causes peripheral vasodilatation.
4-Chloro-5-{4-[2-(2-methoxyphenoxy)-ethyl]-1-piperazinyl}-3(2H)-pyridazinone derivatives (14) were synthesized by Strappaghetti et al. which were good α1 antagonists. These compounds had four or five carbon linkers in them, which have made them good toward activity and selectivity for α-AR (Strappaghetti et al., 2000).

Synthesis of a series of 3(2H)-pyridazinone derivatives (15) was taken up, and the compounds were evaluated in vitro using radioligand receptor binding assays for their affinity toward adrenoceptors (α1 and α2) (Corsano et al., 1999). The target compounds have shown good affinity toward α1 AR having Ki values in sub-nanomolar range. An increase in affinity was observed with increase in the polymethylene chain length in the series with maximum activity up to six and seven carbon chain lengths, and 4-[2-(2-methoxyphenoxy)-ethyl]-1-piperazinyl moiety was linked at position 5 of the 3(2H)-pyridazinone ring. A further increase in the chain length in the homologous series led to decrease in the activity. When 4-[2-(2-methoxyphenoxy)-ethyl]-1-piperazinyl group was linked at position 6, a different effect was observed. In this case, the highest affinity was observed when the polymethylene chain length was of four carbon atoms. It was also observed that this polymethylene chain length which acts as spacer between the two major constituents of the molecule can influence the affinity as well as the selectivity of the compounds.

In another study, piperazine was linked from N-1 nitrogen to positions 4, 5, or 6 of pyridazinone ring, and the N-4 nitrogen was linked to a suitable adrenoceptor pharmacophore (1,4-benzodioxanyl, 2- methoxy-phenoxyethyl or phenoxyethyl groups) by a suitable spacer (sp). The acidic hydrogen N-2 of pyridazinone was also substituted by phenyl or methyl groups. The α1-adrenoceptor antagonist activity was obtained in terms of pkb. The general structure of this class 16 was build by COMFA (comparative molecular field analysis). The SAR and COMFA results of the study had put forward certain characteristic features required for good α1-adrenoceptor antagonist activity; these are: the presence of the benzodioxanyl nucleus or its 2-methoxyphenoxyethyl analog, the phenyl ring of the adrenoceptor pharmacophore moieties must be at a specific distance from the protonated piperazine nitrogen atom, the oxygen atom in the 2-methoxy-phenoxyethyl or benzoxanyl groups enhances the activity due to formation of hydrogen bond with the receptor. The modifications of 4- and 5-(1-piperazinyl)pyridazinone isomers did not have much effect on the activity, while a decrease in activity was observed in 6-substituted isomer. The substitution of N-2 acidic proton in 5-(1-piperazinyl) isomers lowered the activity, whereas it increased the activity in case of 4-(1-piperazinyl) isomers (Cinone et al., 1999).

Cardiotonic
Congestive heart failure is a common, costly, disabling, and potentially deadly condition. Digoxin is widely used in the treatment for various heart conditions, namely atrial fibrillation, atrial flutter, and sometimes heart failure that cannot be controlled by other medication. Digitalis/digoxin has recently fallen out of favor because it did not demonstrate a mortality benefit in patients with congestive heart failure, and due to its toxic side effects, the scientists have been encouraged to develop an alternative therapy (Braunwald, 1981; Packer, 1988).
Inhibition of the phosphodiesterase (PDE) isoenzyme III leads to an increase in intracellular concentrations of the second messenger cAMP, which mediates the phosphorylation of protein kinases, which in turn activates cardiac calcium channels. An increased calcium influx from the sarcoplasmic reticulum (SR) during phase 2 (the plateau phase) of the cardiac action potential leads to a positive inotropic effect of PDE III inhibitors: they increase the force of cardiac contraction. This increased reflux of calcium into the SR is also responsible for myocardial relaxation. In addition to that, PDE III inhibitors also act as vasodilators (Osadchii, 2007). Amirinone a pyridine PDE III inhibitor was the first drug in this category (Farah and Alousi, 1978). A further research in this field in search of safer and more potent compounds led to the discovery of milrinone and its analogs (Bekhit and Baraka, 2005; Abadi et al., 1999). To develop SAR in the series, several pyridazinone derivatives of amrinone were also synthesized (Combs et al., 1992; Bakewell et al., 1990; McCall et al., 1986; Pastelin et al., 1983). Their mechanism of action was established, and a five-point model and pharmacophore were identified (Leclerc et al., 1986; Erhardt et al., 1988; Bristol et al., 1984). It was found out from the study that most of the compounds acting as c-AMP PDE III inhibitors contained pyridazinone ring attached to an aromatic nucleus. In the sub category of these compounds were indolidan (4) (van Meel, 1985), bemoradan (Combs et al., 1990), and pimobendan (5) (Robertson et al., 1987a, b), in which the pyridazinone ring was attached to a benzo-fused heterocycle. This benzo-fused heterocyclic system governs the pharmacokinetics of the molecule (Abouzid et al., 1999; Demirayak et al., 2004a, b).
A series of compounds designed by Abouzid et al. (2008) had pyridazinone attached directly to the quinolone nucleus. In next series, an aminophenyl spacer was inserted between the pyridazinone and quinoline molecules (17). In the third series, they had engaged pyridazinone and pyridone ring systems via the same spacer moiety. All the compounds were evaluated for their vasorelaxant activity in vitro on isolated main pulmonary artery of the rabbit. The highest vasorelaxant activity was observed by two compounds in the 4-amino quinoline series when compared with milrinone. Both these compounds had higher values of pharmacophoric fit in the molecular mapping study as they possessed phenolic OH group and alkoxy group at position 6 on the quinoline ring.

In another study, arylidene substituted meldrum’s acid and 5-amino-6-phenyl-3(2H)-pyridazinone were used to synthesize 4-aryl-2,5-dioxo-8-phenylpyrido[2,3-d]pyridazines (Coelho et al., 2004). In these compounds, it was revealed that the pyridine system is in skew boat conformation, the phenyl ring is in pseudo-axial position, and the pyridazinone ring is planar. Thus, these compounds possessed all the requirements for cardio tonic activity from structural point of view (Pita et al., 2000).
Anti-inflammatory/analgesic/antipyretic
The literature reference of pyridazinones as analgesic, anti-inflammatory, and antipyretic is almost around four decades old. Various 2-alkyl or 2-alkenyl-4-alkoxy-5-(substituted amino)-3(2H)-pyridazinones (18) were synthesized and evaluated for analgesic and anti-inflammatory activities. The compound 4-ethoxy-2-methyl-5-morpholino-3(2H)-pyridazinone (emorfazone, 7) was the most potent in this category. It was found to possess higher potency and lesser toxicity than aminopyrine and phenylbutazone (Takaya et al., 1979).

There are reports of synthesis of 4,6-diaryl-3-pyridazinones and their evaluation as anti-inflammatory, analgesic, and antipyretic. It was observed that inclusion of arylpiperazinomethyl moiety at position 2 in the pyridazinone ring enhanced the potency of the compounds (Rubat et al., 1989; Santagati et al., 1985a, b).
Among the earlier study on pyridazinone derivatives related to aminopyrine containing pyridazinone nucleus was the synthesis of 4-alkoxy-2-methyl-5-morpholino-3(2H)-pyridazinones (19). These compounds were found to possess good anti-inflammatory and analgesic properties (Sato, 1979).

Synthesis of 3(2H)-pyridazinone derivatives was reported possessing good analgesic, antipyretic, and anti-inflammatory activities. The analgesic activity of these compounds was higher than phenylbutazone and was devoid of ulcerogenic action (Santagati et al., 1985a, b).
A series of 4-carbomyl-5-aryl-6-methyl-4,5-dihydropyridazin-3(2H)-ones (20) (Wang et al., 2007) were also synthesized and evaluated for anti-inflammatory and analgesic properties. Only one compound showed anti-inflammatory activity but had shorter duration than that of indomethacin, which was taken as reference drug. On the other hand, many derivatives had displayed significant analgesic properties.

Synthesis of various 6-substituted-3(2H)-pyridazinones was carried out, and the compounds were evaluated for anti-inflammatory and analgesic activities. The compound 6-[4-(2-fluorophenyl)piperazin-1-yl]-3(2H)-pyridazinone (21) exhibited anti-inflammatory activity similar to indomethacin. The SAR of these compounds confirmed the influence of substitution at position 6 in the 3(2H)-pyridazinone ring toward the anti-inflammatory–analgesic potency of the molecules (Gokce et al., 2004).

Literature reports synthesis of some pyridazinone derivatives and biheterocyclic derivatives made with pyridazinone ring having 4-arylpiperazinylalkyl substituents attached to the lactam nitrogen of pyridazinone (22) (Biancalani et al., 2009a, b; Giovannoni et al., 2003; Dogruer et al., 2000). These compounds were found to have notable analgesic property. To further investigate the role of heterocyclic substituent attached to the pyridazinone lactam nitrogen, toward their analgesic property, Malinka et al. (2011) carried out design and synthesis of compounds containing arylpiperazinylpropyl chain linked to 4-O atom of a tautomeric form of pyrrolopyridazinone. These compounds were evaluated for their analgesic potency using hot plate method (for central analgesia) and writhing test (for peripheral analgesia). ASA (aspirin) and morphine were used as standards in these tests. From the results, it was revealed that substitution of pyrrolidinone ring with pyridazinone ring in pyrrole-3,4-dicarboximides increased the peripheral analgesic property of the compound, whereas replacement of the 4-arylpiperazinylalkyl moiety from lactam N-2 to 4-O atom of the hydroxyl group of tautomeric form of pyrrolopyridazinone diminishes the analgesic activity.

In one of the recent studies carried out by Syed et al., some 6-aryl-2-(p-(methanesulfonyl)phenyl)-4,5-dihydropyridazi-3(2H)-one derivatives (23) were synthesized and evaluated in vivo for anti-inflammatory activity in carrageenan-induced rat paw edema, using etoricoxib as standard drug. The compounds have shown mild-to-moderate anti-inflammatory activity. These compounds were also tested for their ulcerogenic potential and were found to be safe from the point of view of ulcer induction (Ovais et al., 2013). The same group has also evaluated 6-aryl-2-(p-sulfamoylphenyl)-4,5-dihydropyridazin-3(2H)-ones for similar anti-inflammatory activity and their ulcerogenic potential, and this series was also found to be potent anti-inflammatory and low ulcer induction potential (Bashir et al., 2012).

COX inhibitors
Cyclooxygenase (COX) is the key enzyme in the manifestation of inflammation and pain, and it catalyzes the conversion of arachidonic acid into prostaglandin H2. NSAIDS (nonsteroid anti-inflammatory drugs) which are COX inhibitors are widely used in the treatment for pain and inflammation. The major side effects of these drugs are ulceration in gastrointestinal tract and hemorrhage (Allison et al., 1992). The cyclooxygenase exists in two isoforms, COX-1 and COX-2. COX-2 is the enzyme which is activated in response to various pro-inflammatory stimuli (Kurumbail et al., 1996). The major structural difference in the two isoforms of the enzyme lies in the active site where COX-1 contains isoleucine residue (Ile523) which is replaced by a valine residue in COX-2 (Val523). This difference in single amino acid accounts for the differences in the polarity, binding kinetics, and selectivity of the COX-2 inhibitors toward its isoenzyme (Bombardier, 2002). Thus, selective COX-2 inhibitors have been found to have lesser gastrointestinal side effects, and since COX-1 is not inhibited, its cytoprotective action helps in healing (Ferreira et al., 1971; Murry and Brater, 1993; Dannhardt and Kiefer, 2001). In the last decade, many COX-2 inhibitors have reached the market (Penning et al., 1997), few of them are rofecoxib (Talley et al., 2000), celecoxib (Li et al., 1996), valdecoxib (Friesen et al., 1998), and etoricoxib (Li et al., 2003).
In search of novel selective cyclooxygenase inhibitors, the compounds bearing pyridazinone moiety such as the syntex compound (24) RS-57067 (25) and benzylpyridazinone derivatives (26) (Biancalani et al., 2006; Beswick et al., 2004; Chintakunta et al., 2002) have come into light. The various derivatives such as 3-o-substituted benzyl pyridazinone (Li et al., 2003; Okcelik et al., 2003), 4-phenyl, and 4-(2-chlorophenyl)-6-(5-chloro-2-oxo-3H-benzoxazol-7-yl)-3(2H)-pyridazinone (27) (Harris et al., 2004) were also found to be selective cyclooxygenase inhibitors.




In 2004, Harris et al. reported vicinally disubstituted pyridazinones as potent and selective COX-2 inhibitors. The compound A-241611 (28) was discovered as an early lead from the series, and it showed both acute and chronic anti-inflammatory profiles in rat models. But this compound suffered from low aqueous solubility and hence lower rate of elimination. A further research in this field gave a 4-butoxy-substituted compound, A-282904 (29), which had good anti-inflammatory and analgesic potency as a COX-2 inhibitor in comparison with A-241611. But it still suffered from poor solubility. Synthesis of another derivative using additional alcohol to the terminal carbon of the alkoxy side chain gave a good compound ABT-963 (([2-(3,4-difluoro-phenyl)-4-(3-hydroxy-3-methyl-butoxy)-5-(4-methanesulfonyl-phenyl)-2H-pyridazin-3-one], 30). This compound was a selective and potent COX-2 inhibitor with a better solubility profile when compared with the other two previous compounds and even with established drugs celecoxib and rofecoxib. Because of its improved solubility, it had shown improved pharmacokinetic profile. ABT-963 reduced the prostaglandin E2 levels after oral administration, which is responsible for its anti-inflammatory effect, and also reduced nociception in hyperalgesic models in dose-dependent manner. Its high selectivity toward COX-2 inhibitors showed its effectiveness as anti-inflammatory in arthritic models also.



In one of the studies, a series of pyridazinone derivatives were synthesized by linking the aryl and biphenyl moieties at position 6 using two carbon spacer in between (31). Such compounds exhibited good anti-inflammatory activity along with better gastrointestinal protectivity. The results had shown that using ethyl spacer between two dihydropyridazinone ring and aryl moiety gave the compounds with higher activity when compared to the ethenyl analogs (Gokce et al., 2001).

Some of the pyrazolone derivatives have been found to possess analgesic and anti-inflammatory activities such as dipyrone and phenylbutazone, but the side effects associated with them have limited their clinical usefulness. The 3(2H)-pyridazinone derivatives which are structurally related to pyrazolone have been widely reported in the literature as analgesic and anti-inflammatory compounds devoid of side effects (Gokce et al., 2005; Dündar et al., 2007; Sato et al., 1981). One such compound is emofazone (7), it possess anti-nociceptive property which is neither mediated by prostaglandin nor through opiod receptors (Viaud et al., 1995; Rohet et al., 1996). A number of studies have confirmed that the attachment of aryl piperazinyl side chain to the lactam nitrogen in the pyridazinone ring lends the molecule analgesic activity (Giovannoni et al., 2003; Rubat et al., 1992; Piaz et al., 2003; Santagati et al., 1985a, b; Banoglu et al., 2004). This analgesic potency can further be increased by inserting a spacer carbon chain between the lactam nitrogen and the amine moiety (Gökce et al., 2009b). Keeping these points in mind, Gokce et al., (2009a) designed and synthesized 6-(substituted-arylpiperazinyl)-3(2H)-pyridazinone derivatives bearing different substituents at position 2 in the pyridazinone ring. The compounds belonging to 6-substituted-3(2H)-pyridazinone-2-acetyl-2-(p-substituted benzal)hydrazone derivatives (32) were evaluated for analgesic and anti-inflammatory activities using carrageenan-induced writhing and rat paw edema tests, respectively, taking phenylbenzoquinone as standard. They were also tested for their irritative and ulcerogenic potential on gastric mucosa. 6-[4-(3-Chlorophenyl)piperazine]-3(2H)-pyridazinone-2-acetyl-2-benzal hydrazone, 6-[4-(4-chlorophenyl)piperazine]-3(2H)-pyridazinone-2-acetyl-2-benzalhydrazone, and 6-[4-(pyridyl)piperazine]-3(2H)-pyridazinone-2-acetyl-2-benzal hydrazone derivatives were found to possess better analgesic–anti-inflammatory activity, and cytoprotective effect in comparison with the reference compound acetyl salicylic acid (Piaz et al., 1998).

Asthma/COPD
The treatment regimen for chronic obstructive pulmonary disease (COPD) and asthma, although dependent on the severity of the diseases, mainly depends on corticosteroids and β2-agonists (Gaga et al., 2007). In cases of mild-to-moderate asthma, a combination of inhalation corticosteroids and long-acting β2-agonist (LABA) is employed now days. However, corticosteroids when taken in high doses over a prolonged period of time can produce several severe side effects (US FDA Web site). Similarly, LABA has also been reported to possess certain potential adverse effects in their post-approval clinical trials from 2011 by US FDA (Chung, 2006). Thus, there is a dire need of some other category drug in the management of these respiratory diseases. Inhibitors for the phosphodiesterase (PDE) IV enzyme family have stolen a considerable interest as compounds with the mixed action having anti-inflammatory and bronchodilatory activities; they act by increasing the intracellular c-AMP levels and inducing smooth muscle relaxation (Torphy et al., 1998). A good PDE IV inhibitor can be an appropriate substitute for β2-agonist and corticosteroids. Rolipram was one such compound but could not make it to the market because of low potency and side effects such as nausea, vomiting, and headache. Roflumilast (35) is another drug which has been approved by US FDA in 2011 and acts as selective, long-acting PDE IV inhibitor. Pyridazinones have also been developed as PDE IV inhibitors, particularly compounds such as syntex agents 24, 33, and 34 (Giembycz et al., 2008).



Another inhibitor in phosphodiesterase series is PDE III inhibitors, which have been found to be superior to PDE IV inhibitors in mediating the relaxation of airway passage (Ochiai et al., 2011). Hence, either a combination of PDE III and IV inhibitors or dual inhibitors (PDE III/IV) offer a better therapeutic response in comparison with the individual selective agents.
Working on these lines, Ochiai et al. (2012) have synthesized (−)-6-[7-methoxy-2- (trifluoromethyl)pyrazolo[1,5-a]pyridin-4-yl]-5-methyl-4,5-dihydro-3(2H)-pyridazinone (KCA-1490, 36) as a dual PDE III/IV inhibitor. This compound possessed anti-inflammatory and bronchodilatory activity and had an improved therapeutic window over the existing drug roflumilast (Bristol et al., 1984). When the compounds in KCA-1490 series were studied for SAR, it was observed that 5-methyldihydropyridazinone subunit was essential for the PDE III inhibitory activity.

Anticancer activity
Pyrrole[2,3-d]pyridazine is one of the rarely reported moiety. Pyrrole[2,3-d]pyridazin-4-ones (37) (Marquet and Bisagni, 1968) have been reported in the literature as inhibitors of human cancer cell proliferation and pyrrole[2,3-d]pyridazin-7-ones (38) as carbohydrate-modified nucleosides which are active as antiviral and antiproliferative agents (Meade et al., 1997). Another work has been reported by Murineddu et al. (2002) on 1-methyl-2-phenyl- and 1,3-dimethyl-2-phenyl-substituted pyrrole[2,3-d]pyridazinones as cytotoxic agents were tested in vitro at NCI in preclinical antitumor screening program against 60 human tumor cell lines. Significant activity was found for indenopyrrole[2,3-d]pyridazinone 39 (X=CH2) and for benzo[g]pyridazin[4,5-b]indol-7-one, 40 (X=CH=CH).



A new class of 6-aryl-2-(p-sulfamylphenyl)-pyridazin-3(2H)-ones (41) was designed and synthesized. They were tested against human cancer cell line. Of all the compounds evaluated, the compound bearing R1=R2=H and R3=C2H5 substitution was found to possess remarkable activity against non-small-cell lung and leukemia cell lines. In acute toxicity studies also, this compound has shown good intraperitoneal tolerance (Rathish et al., 2012).

In another study, the antiproliferative potential of a series of 23 compounds bearing substituted aryl-pyridazin-3(2H)-one moiety was tested against 60 human cancer cell lines. One of the compounds bearing an ethyl substitution at position R2 in the ring displayed promising antiproliferative activity against 20 cell lines (Ovais et al., 2013).
Antimicrobial activity
Various metal complexes of 5-benzoyl-4-hydroxy-2-methyl-6-phenyl-2H-pyridazin-3-one (42) were synthesized using Cd(II) and Ni(II) by Sonmez et al. These complexes have shown activity against gram-positive S. aureus and gram-negative Pseudomonas putida and two yeasts (Candida albicans and C. tropicalis). Sortase A (SrtA) is an enzyme which attaches the surface proteins in S. aureus to the cell wall. This enzyme helps in the transpeptidation process of the cell wall synthesis. Any molecule which inhibits this enzyme would hence be a powerful anti-infective agent as it would inhibit transpeptidation ultimately not allowing cell wall synthesis of the microorganism. In an attempt to identify SrtA inhibitors, high throughput screening of around 30,000 compounds was carried out, which lead to the identification of three molecules 43–45 that could be developed into anti-infective agents. The pyridazinone and pyrazolethione analogs were found to be significant in structure–activity relationship studies. These molecules showed better activity than the already existing synthetic or natural compounds and held good potential to be explored further (Sonmez et al., 2006).




Antifungal agent
β-1,3-Glucan synthase (GS) is the target enzyme for the development of many antifungal agents. Echinocandins are a class of semisynthetic natural products that act as GS inhibitor but can only be given parenterally (Walker et al., 2011). In an attempt to develop novel small molecules as antifungal agents, a class of piperazinyl–pyridazinones (46–49) was discovered as GS inhibitor. These classes of compounds have shown oral efficacy against C. glabrata infection in murine model and were found to be superior than echinocandins (Ton-That et al., 2004).




Antiplatelet activity
Formation of thrombus in the coronary artery is the primary cause for the development of angina, stroke, myocardial infarction, and peripheral artery disease (Sureea et al., 2009). Owing to the better understanding of the role of platelets in vascular injuries, new approaches for the better and efficacious antiplatelet agents are coming into light. Aspirin, clopidogrel, ticlopidine, and sulfinpyrazole are among various therapeutic strategies developed to inhibit platelet function. A series of 5-aryl-6-methyl-4,5-dihydropyridazin-3(2H)ones (50) and related 5-aryl-6-methyl-pyridazin-3(2H)ones (51) were synthesized and evaluated for their pharmacological profile. Some of them displayed significant antithrombotic and antiulcer properties (Pinna et al., 1988).


In late 90s, a lot of work can be found on pyridazinones acting as platelet aggregation inhibitors. A series of 4,5-functionalized-2-methyl-6-(substituted phenyl)-3(2H)-pyridazinones (52) were synthesized and evaluated as platelet aggregation inhibitors in human platelet-rich plasma. The compounds had shown good activity in sub-micromolar range (Fuster et al., 1992).

In search of new antiplatelet agents in a series, 6-phenyl-3(2H)-pyridazinones were developed with a diverse range of substituent in the position 5. These compounds were evaluated for their potential to inhibit platelet aggregation. These modifications confirmed that the group at position 5 in 6-phenyl-3(2H)-pyridazinones system influences both variations in antiplatelet activity and mechanism of action (Coelho et al., 2004). In another work, 6-phenyl-5-hydroxymethyl-4,5-dihydro-3(2H)-pyridazinone (53) and 6-thienyl-5-hydroxymethyl-4,5-dihydro-3(2H)-pyridazinones (54) were found to inhibit platelet aggregation induced by thrombin (IC50 = 0.25 and 0.26 mm, respectively) or by the calcium ionophore ionomycin (IC50 = 0.42 and 0.43 mm, respectively; Pinna et al., 1988).


Glycoprotein (IIB/IIIA) receptor antagonist is a new class which has been identified as antiplatelet agents. Amrinone and milrinone (2(1H)-pyridones), zardaverine (55), and pimobendan (5) (3(2H)-pyridazinones) are clinically used PDE III inhibitors (56). A number of 6-aryl-3(2H)-pyridazinone derivatives substituted at position 5 have been synthesized, and their mechanism of action is found to be based on their capacity to inhibit calcium ion influx, which is required for the activation of platelet aggregation. The substitutions at position 5 greatly influence the potency of the molecule, like the presence of electron-withdrawing groups at position 5, e.g., CHO, COOME, and CH2OCOCH3 enhance the activity (Dal Piaz et al., 1997).

Coelho et al. have prepared 5-substituted-6-phenyl-3(2H)-pyridazinones (57) and reported antiplatelet activity along with SAR. The compounds such as “6c” and “6f” were found to be particularly interesting (Schneider et al., 1979). In a study conducted by Cherng et al., a new pyridazinone derivative PC-09 (58) was synthesized and found to be a good antiplatelet aggregating agent. Upon investigation, it was revealed that PC-09 has multiple modes of action, i.e., it causes inhibition of thromboxane A2 formation, reduction in mobilization of intracellular calcium, and platelet surface GPIIB/IIIA expression along with increasing cyclic AMP levels by inhibiting cyclic AMP phosphodiesterase (Cherng et al., 2006).

Antidiabetic
Benzenesulfonylurea substituted 15 novel pyridazinone derivatives “(3a–o)” were synthesized from their corresponding sulfonamides derivatives via novel carbamates. Their blood sugar lowering effect was evaluated using glucose tolerance test at the dose of 20 mg/kg (p.o.) Thirteen of these compounds have shown good decrease in blood sugar levels of the rat models (Rathish et al., 1997).
Acetylcholinesterase inhibitors
Alzheimer’s disease (AD) is a complex neurodegenerative disorder of the central nervous system. Acetylcholinesterase (AchE), a serine protease, is responsible for acetylcholine hydrolysis. The hydrolysis of acetylcholine results in the termination of nerve impulses at the cholinergic synapses and neuromuscular junction. Among the various approaches for treating AD, inhibition of AchE is still prevailing in treating or alleviating the symptoms of AD. Xing et al. have identified 2,6-disubstituted pyridazinone by HTS as a novel acetylcholinesterase (AchE) inhibitor. By SAR development, compound 59 stood out as displaying high ache inhibitory activity and AchE/butyrylcholinesterase (BuchE) selectivity in vitro. Docking studies revealed that 59 might interact with the catalytic active site (CAS) and the peripheral anionic site (PAS) simultaneously. Based on this novel binding information, 6-o-tolylamino and N-ethyl-N-isopropylacetamide substituted piperidines were disclosed as new PAS and CAS binder (Xing et al., 2013).

HT agonist
5-HT is instrumental in regulating mood, sleep, sexuality, and appetite. The 5-HT receptor subtype 5HT2c is present in brain, and their agonists have potential for the treatment for sexual dysfunction, obesity, schizophrenia, and urinary incontinence. Certain pyridazinone derivatives were synthesized, and their SAR was established as 5HT2c receptor agonist for urinary incontinence. The SAR was developed by using the known non-selective 5-HT2c agonist meta-chlorophenyl piperidine (60). The compound 61 has shown good in vivo efficacy in preclinical models of stress urinary incontinence (Charlotte et al., 2009).

Antifeedant
Oxadiazolyl 3(2H)-pyridazinone (ODP, 62) was developed and evaluated as antifeedant and compared with toosendanin, an insecticide extracted from the bark of Melia toosendan. The insect growth regulatory and antifeedant activity were checked on the larvae of the armyworm, Pseudaletia separata walker (Lepidoptera: Noctuidae). This compound was found to inhibit the activities of the enzymes such as trypsin, chymotrypsin, and alpha amylase in the fifth instar of the larvae (Cao et al., 2003; Asif, 2013a, b).

Conclusion
Pyridazinone, although a simple basic nucleus, holds a lot of potential. With basic prior understanding of SAR, this moiety can be utilized effectively as a lead in many of known pharmacodynamic fields such as antihypertensives, COX inhibitors, antimicrobial, antitubercular, antidiabetic, antiplatelet, anticancer, antidepressant–anxiolytic, anticonvulsant, bronchodilatory, antifeedant, 5-HT agonists, and many other properties. A number of drugs in the market have pyridazinone nucleus/moiety in their structures such as indolidan, pimobendan, levosimendan, emorfazone, and zardaverine. The molecule has been studied well, and a good amount of SARs has been established for various series, but it still holds potential for further exploitation in various known and unknown fields.
References
Abadi A, Al-Deeb O, Al-Afify A, El-Kashef H (1999) Synthesis of 4-alkyl (aryl)-6-aryl-3-cyano-2(1H)-pyridinones and their 2-imino isosteres as nonsteroidal cardiotonic agents. Il Farmaco 54:195–201
Abouzid K, Bekhit SA (2008) Novel anti-inflammatory agents based on pyridazinone scaffold design synthesis and in vivo activity. Bioorg Med Chem 16:5547–5556
Abouzid KA, Youssef KM, Amine FM, Botros S, Isaac Z (1999) Synthesis of 14-disubstituted piperazines as potential antihypertensive agents Egypt. J Pharm Sci 30:429–436
Abouzid K, Hakeem MA, Khalil O, Maklad Y (2008) Pyridazinone derivatives: design synthesis and in vitro vasorelaxant activity. Bioorg Med Chem 16:382–389
Abubshait SA (2007) An efficient synthesis and reactions of novel indolylpyridazinone derivatives with expected biological activity. Molecules 12(1):25–42
Allison MC, Howatson AG, Torrance CJ, Lee FD, Russel RI (1992) Gastrointestinal damage associated with the use of nonsteroidal antiinflammatory drugs. N Engl J Med 327:749–756
Anwair MA, Károlyházy L, Szabó D, Balogh B, Kövesdi I, Harmat V, Krenyácz J, Gellért A, Takács-Novák K, Mátyus P (2003) Lipophilicity of aminopyridazinone regioisomers. J Agric Food Chem 51(18):5262–5270
Archan S, Toller W (2008) Levosimendan: current status and future prospects. Curr Opin Anesthesiol 21(1):78–84
Asif M (2010) Exploring potential synthetic methods and general chemistry of pyridazine and pyridazinone: a brief introduction. Int J Chem Tech Res 2(2):1112–1128
Asif M (2012) Some recent approaches of biologically active substituted pyridazine and phthalazine drugs. Curr Med Chem 18:2984–2991
Asif M (2013a) Antifeedant herbicidal and molluscicidal activities of pyridazinone compounds. Mini-Rev Org Chem 10:113–122
Asif M (2013b) Recent approaches toward anticonvulsant activity of pyridazine compounds open. J Org Chem. doi:10.12966/ojoc06.01.2013
Ayla B, Fügen Ö, Oya Ü, Zafer G, Berna T (2003) Studies on some 3-oxo-5-benzylidene-6-methyl-(4H)-2-substitutedpyridazines with antinociceptive and antiinflammatory activities FABAD. J Pharm Sci 28:19–25
Bakewell SJ, Coates WJ, Comer MB, Reeves ML, Warrington BH (1990) Inotropic vasodilator and low Km cAMP-selective cGMP-inhibited phosphodiesterase (PDE III) inhibitory activities of 4a-methyl-44a-dihydro-5H-indeno[12-c]pyridazin-3(2H)-ones and 4a-methyl-44a56-tetrahydrobenzo[h]cinnolin-3(2H)-ones. Eur J Med Chem 25:765–774
Banoglu E, Akoglu C, Unlu S, Kupeli E, Yesilada E, Sahin MF (2004) Amide derivatives of [6-(5-methyl-3-phenylpyrazole-1-yl)-3(2H)-pyridazinone-2-yl] acetic acids as potential analgesic anti-inflammatory compounds. Arch Pharm Pharm Med Chem 337:7–14
Bansal R, Thota S (2013) Pyridazin-3(2H)-ones: the versatile pharmacophore of medicinal significance. Med Chem Res 22:2539–2552
Bansal R, Kumar D, Carron R, de la Calle C (2009) Synthesis and vasodilatory activity of some amide derivatives of 6-(4-carboxymethyloxyphenyl)-45-dihydro-3(2H)-pyridazinone. Eur J Med Chem 44(11):4441–4447
Barbaro R, Betti L, Botta M, Corelli F, Giannaccini G, Maccari L, Manetti F, Strappaghetti G, Corsano S (2001) Synthesis biological evaluation and pharmacophore generation of new pyridazinone derivatives with affinity toward α1- and α2-adrenoceptors. J Med Chem 44:2118–2132
Bashir R, Yaseen S, Ovais S, Ahmad S, Hamid H, Alam MS, Samim M, Javed K (2012) Synthesis and biological evaluation of some novel sulfamoylphenyl-pyridazinone as anti-inflammatoryagents (part-II*). J Enzyme Inhib Med Chem 27(1):92–96
Bekhit AA, Baraka AM (2005) Novel milrinone analogs of pyridine-3-carbonitrile derivatives as promising cardiotonic agents. Eur J Med Chem 40:1405–1413
Beswick P, Sharon B, Bountra C, Brown T, Browning K, Campbell I, Chessell I, Clayton N, Collins S, Corfield J, Guntrip S, Haslam C, Lambeth P, Lucas F, Mathews N, Murkit G, Naylor A, Pegg N, Pickup E, Player H, Price H, Stevens A, Stratton S, Wiseman J (2004) Identification of 23-diaryl-pyrazolo[15-b]pyridazines as potent and selective cyclooxygenase-2 inhibitors. Bioorg Med Chem Lett 14:5445–5458
Biancalani CN, Vergelli CC, Piaz VD, Graziano A, Biagini P, Ghelardini C, Galeotti N, Giovannoni MP (2006) Arylpiperazinylalkylpyridazinones and analogues as potent and orally active antinociceptive agents: synthesis and studies on mechanism of action. J Med Chem 49:7826–7835
Biancalani C, Giovannoni MP, Pieretti S, Cesari N, Graziano A, Vergelli C, Cilibrizzi A, Di Gianuario A, Colucci M, Mangano G, Garrone B, Poenzani L, Dal Piaz V (2009) J Med Chem 52:7397–7409
Biancalani C, Giovannoni MP, Pieretti S, Cesari N, Graziano A, Vergelli C, Cilibrizzi A, DiGianuario A, Colucci M, Mangano G, Garrone B, Polenzani L, Dal Piaz V (2009) Further studies on arylpiperazinyl alkyl pyridazinones: discovery of an exceptionally potent orally active antinociceptive agent in thermally induced pain (dagger). J Med Chem 52:7397–7409
Bombardier C (2002) An evidence-based evaluation of the gastrointestinal safety of coxibs. Am J Cardiol 89:3D–9D
Bowman P, Haikala H, Paul RJ (1999) Levosimendan a calcium sensitizer in cardiac muscle induces relaxation in coronary smooth muscle through calcium desensitization. J Pharmacol Exp Ther 288(1):316–325
Braunwald E (1981) Heart failure: pathophysiology and treatment. Am Heart J 102:486–490
Bristol JA, Sircar I, Moos WH, Evans DB, Weishaar RE (1984) Cardiotonic agents. 1. 45-dihydro-6-[4-(1H-imidazol-1-yl)phenyl]-3 (2H)-pyridazinones: novel positive inotropic agents for the treatment of congestive heart failure. J Med Chem 27:1099–1101
Cao S, Qian X, Song G, Chai B, Jiang Z (2003) Synthesis and antifeedant activity of new oxadiazolyl 3(2H)-pyridazinones. J Agric Food Chem 51:152–155
Charlotte MN, Mark DA, Blagg J, Ellis D, Evrard E, Martin P, Green MP, Liu KK-C, McMurray G, Ralph M, Sanderson V, Ward R, Watson L (2009) Design and synthesis of pyridazinone-based 5-HT2C agonists. Bioorg Med Chem Lett 19:5791–5795
Cherng SC, Huang WH, Shiau CY, Lee AR, Chou TC (2006) Mechanisms of antiplatelet activity of PC-09 a newly synthesized pyridazinone derivative. Eur J Pharmacol 532(1–2):32–37
Chintakunta VK, Akella V, Vedula MS, Mamnoor PK, Mishra P, Casturi SR, Vangoori A, Rajagopalan G (2002) R 3-O-substituted benzyl pyridazinone derivatives as COX inhibitors. Eur J Med Chem 37(4):339–347
Chung KF (2006) Phosphodiesterase inhibitors in airways disease. Eur J Pharmacol 533:110–115
Cignarella G, Grella G, Loriga M, Curzu MM, Schiatti G (1978) Unexpected anti-inflammatory activity of rigid structure derivatives of 6-arylpyridazinone antihypertensives. I. Synthesis and activity of 44a-dihydro-5H-indeno[12c]pyridazin-3-ones. Farmaco [Sci] 33(11):866–874
Cignarella G, Lorgia M, Pinna GA, Pirisi MA, Schiatti P, Selva D (1982) Unexpected anti-inflammatory activity of rigid structures derived from antihypertensive 6-arylpyridazinones. III. Synthesis and activity of 7-fluoro- and 5-keto-5H-indeno(12c)pyridozines. Farmaco 37(2):133–144
Cignarella G, Barlocco D, Landriani L, Folloni M, Pinna GA, Sala F, Germini M (1986) 6-Aryl-45-dihydro-3(2H)-pyridazinones derived from 5H-indeno(12c)pyridazine. J Med Chem 29:2191–2194
Cignarella G, Barlocco D, Landriani L, Folloni M, Pinna GA, Sala F, Germini M (1998) New congeners of the antihypertensive and antithrombotic 7-amino(Ib) and 7-acetyl amino-44a-dihydro-5H-indeno(12c)pyridazin-3-ones. Farmaco 43:169–179
Cinone N, Carrieri A, Strappaghetti G, Corsano S, Barbaro R, Carotti A (1999) Comparative molecular field analysis of some pyridazinone-containing α1-antagonists. Bioorg Med Chem 7:2615–2620
Coelho A, Sotelo E, Fraiz N, Yanez M, Laguna R, Cano E, Ravina E (2004) Pyridazines part36: synthesis and antiplatelet activity of 5-substituted-6-phenyl-3(2H)-pyridazinones. Bioorg Med Chem Lett 4:321–324
Combs DW, Rampulla MS, Bell SC, Klaubert DH, Tobia AJ, Falotico R, Haertlein B, Moore JB (1990) 6-Benzoxazinylpyridazin-3-ones: potent long-acting positive inotrope and peripheral vasodilator agents. J Med Chem 33:380–387
Combs DW, Rampulla MS, Demers JP, Falotico R, Moore JB (1992) Heteroatom analogues of bemoradan: chemistry and cardiotonic activity of 14-benzothiazinylpyridazinones. J Med Chem 35:172–176
Corsano S, Strappaghetti G, Barbaro R, Giannaccini G, Betti L, Lucacchini A (1999) Synthesis of new pyridazinone derivatives and their affinity towards α1–α2-adrenoceptors. Bioorg Med Chem 7(5):933–941
Coudert P, Duroux E, Bastide P, Couquelet J, Tronche P (1991) Synthesis and evaluation of the aldose reductase inhibitory activity of new diaryl pyridazine-3-ones. J Pharm Belg 46(6):375–380
Curran WV, Ross A (1974) 6-Phenyl-45-dihydro-3(2H)-pyridazinones. Series of hypotensive agents. J Med Chem 17:273–281
Dal Piaz V, Ciciani G, Giovannoni MP (1997) Synthesis of 45-functionalized-2-methyl-6-(substituted aryl)-3(2H)-pyridazinones: a new group of potent platelet aggregation inhibitors. Farmaco 52:173–178
Dannhardt G, Kiefer W (2001) Cyclooxygenase inhibitors—current status and future prospects. Eur J Med Chem 36:109–126
Demirayak S, Karaburun AC, Beis R (2004a) Some pyrrole substituted aryl pyridazinone and phthalazinone derivatives and their antihypertensive activities. Eur J Med Chem 39:1089–1095
Demirayak S, Karaburn AC, Kayagil I, Erol K, Basar S (2004b) Some pyridazinone and phthalazinone derivatives and their vasodilator activities. Arch Pharm Res 1:13–21
Deniz SDM, Fethi S (2003) Synthesis and analgesic and anti-inflammatory activity of new pyridazinones. Turk J Chem 27:727–738
Dogruer DS, Fiahin MF, Ünlü S, Ito S (2000) Studies on some 3(2H)-pyridazinone derivatives with antinociceptive activity. Arch Pharm Med Chem 333:79–86
Dündar Y, Gökçe M, Küpeli E, Sahin MF (2007) Synthesis and analgesic and antiinflammatory activity of ethyl 6-substituted-3 (2H)-pyridazinone-2-ylacetate derivatives. Arzneim Forsch Drug Res 57:777–781
Erhardt PW, Hagdorn AH, Sabio M (1988) Cardiotonic agents. 3. A topographical model of the cardiac cAMP phosphodiesterase receptor. Mol Pharmacol 33:1–13
Farah AE, Alousi AA (1978) New cardiotonic: agents a search for a digitalis substitute. Life Sci 22:1139–1148
Ferreira SH, Moncada S, Vane JR (1971) Indomethacin and aspirin abolish prostaglandin release from the spleen. Nature 231:237–239
Flouzat C, Bresson Y, Mattio A (1993) Novel non opioid non antiinflammatory analgesics: 3-(aminoalkyl) and 3-[(4-aryl-1-piperazinyl)alkyl]oxazolo [45-b] pyridin-2(3H)ones. J Med Chem 36(4):497–503
Friesen RW, Brideau C, Chan C-C, Charleson S, Deschenes D, Dube D, Ethier D, Fortin T, GauthierJY Girard Y, Gordon R, Greig G, Riendeau D, Savoie C, Wang Z, Wong E, Visco D, Xu L-J, Young RN (1998) 2-Pyridinyl-3-(4-methylsulfonyl)phenylpyridines: selective and orally active cyclooxygenase-2 inhibitors. Bioorg Med Chem Lett 8:2777–2791
Fuster V, Badimon L, Badimon JJ, Chesebro JH (1992) The pathogenesis of coronary artery disease and the acute coronary syndromes (part II). N Engl J Med Chem 326:242–256
Gaga M, Zervas E, Grivas S, Castro M, Chanez P (2007) Evaluation and management of severe asthma. Curr Med Chem 14:1049–1060
Giembycz MA, Kaur M, Leigh R, Newton RA (2008) Holy Grail of asthma management: toward understanding how long-acting beta(2)-adrenoceptor agonists enhance the clinical efficacy of inhaled corticosteroids. Br J Pharmacol 153:1090–1104
Giovannoni MP, Vergelli C, Ghelardini C, Galeotti N, Bartolini A, Piaz VD (2003) [(3-Chlorophenyl) piperazinylpropyl] pyridazinones and analogues as potent antinociceptive agents. J Med Chem 46:1055–1059
Gokce M, Dogruer D, Sahin F (2001) Synthesis and antinociceptive activity of 6-substituted-3-pyridazinone derivatives. Farmaco 56:233–237
Gokce M, Sahin MF, Kupeli E, Yesilada E (2004) Synthesis and evaluation of the analgesic and anti-inflammatory activity of new 3(2H)-pyridazinone derivatives. Arzneimittelforschung 54:396–401
Gokce M, Bakir G, Sahin MF, Kupeli E, Yesilada E (2005) Synthesis of new Mannich bases of arylpyridazinones as analgesic and antiinflammatory agents. Drug Res 55:318–325
Gökçe M, Utku S, Kupeli E (2009a) Synthesis and analgesic and anti-inflammatory activities 6-substituted-3(2H)-pyridazinone-2-acetyl-2-(p-substituted/nonsubstituted benzal) hydrazone derivatives. Eur J Med Chem 44:3760–3764
Gökçe M, Colak MS, Küpeli E, Sahin MF (2009b) Synthesis and analgesic and anti-inflammatory activity of 6-phenyl/(4-methylphenyl)-3(2H)-pyridazinon-2-propionamide derivatives. Arzneimittelforschung 59(7):357–363
Griebel G, Perrault G, Tan S, Schoemaker H, Sanger DJ (1999) Comparison of the pharmacological properties of classical and novel BZ-omega receptor ligands. Behav Pharmacol 10(5):483–495
Hallas J, Lauritsen JK, Villadsen HD, Gram LF (1995) Nonsteroidal anti-inflammatory drugs and upper gastrointestinal bleeding identifying high-risk groups by excess risk estimates. Scand J Gastroenterol 30:438–444
Harris RR, Black L, Surapaneni S, Kolasa T, Majest S, Namovic MT, Grayson G, Komater V, WilCOX D, King L, Marsh K, Jarvis MF, Nuss M, Nellans H, Pruesser L, Reinhart GA, Cox B, Jacobson P, Stewart A, Coghlan M, Carter G, Bell RL (2004) ABT-963[2-(34-difluoro-phenyl)-4-(3-hydroxy-3-methyl-butoxy)-5-(4-methanesulfonyl-phenyl)-2H-pyridazin-3-one] a highly potent and selective disubstituted pyridazinone cyclooxygenase-2 inhibitor. J Pharmacol Exp Ther 311:904–912
Heinisch G, Frank H (1990) Pharmacologically active pyridazine derivatives. Prog Med Chem 27:1–35
Hibi M, Shikada K, Iwama TY, Amamoto A, Sakashita M, Tanaka S (1989) Inhibitory effect of the newly synthesized pyridazinone derivative NZ-107 on bronchoconstriction induced by slow reacting substance of anaphylaxis in the guinea pig. Jpn J Pharmacol 51(3):411–419
Hieble JP, Boyce AJ, Caine M (1986) Comparison of the alpha-adrenoceptor characteristics in human and canine prostate. Fed Proc 45:2609–2614
Ishimori T, Gotanda K, Sasaki T, Shinbo A, Asano H, Miyazawa K, Miyasaka K (1994) Cardiac effects of the novel pyridazinone derivative 6-[4-[2-[3-(5-chloro-2-cyanophenoxy)-2-hydroxypropylamino]-2- methylpropylamino] phenyl]-45-dihydro-5-methyl-3(2H) pyridazinone monoethyl maleate and its metabolite in isolated heart preparations of guinea pigs and dogs. Arzneimittelforschung 44(5):583–588
Iwamoto H (1998) Effect of a cardiotonic agent MCI-154 on the contractile properties of skinned skeletal muscle fibers. Eur J Pharmacol 341(2–3):243–252
Kassab RR (2002) Simple synthesis and reactions of some new pyridazinono derivatives and their antimicrobial activity. Egypt J Chem 45:1055–1073
Korvald C, Nordhaug DO, Steensrud T, Aghajani E, Myrmel T (2002) Vasodilation and mechanoenergetic inefficiency dominates the effect of the “Ca(2+)-sensitizer” MCI-154 in intact pigs. Scand Cardiovasc J 36(3):131–135
Kubo SH (1997) Effects of pimobendan on exercise tolerance and quality of life in patients with heart failure. Cardiology 88(2):21–27
Kumar D, Carron R, La Calle CD, Jindal DP, Bansal R (1996) Synthesis and evaluation of 2-substituted-6-phenyl-45-dihydropyridazin-3(2H)-ones as potent inodilators. Acta Pharm 2008 58(4):393–402
Kurumbail RG, Stevens AM, Gierse JK, McDonald JJ, Stegeman RA, Pak JY, Gildehaus D, Miyashiro JM, Penning TD, Seibert K, Isakson PC, Stallings WC (1996) Structural basis for selective inhibition of cyclooxygenase-2 by anti-inflammatory agents. Nature 384:644–656
Laura K, Wing HA, Behanna LJ, Van ED, Martin W, Hantamalala RR (2006) De novo and molecular target-independent discovery of orally bioavailable lead compounds for neurological disorders. Curr Alzheimer Res 3:205–214
Leclerc G, Marciniak G, Decker N, Schwartz J (1986) Cardiotonic agents. 1. Synthesis and structure–activity relationships in a new class of 3-, 4- and 5-pyridyl-2(1H)-quinolone derivatives. J Med Chem 29:2427–2432
Lee SG, Kim JJ, Kim H-K, Kweon D-H, Kang YJ, Cho SD, Kim SK, Yoon YJ (2004) Recent progress in pyridazin-3(2H)-ones chemistry. Curr Org Chem 8:1463–1480
Li JJ, Norton MB, Reinhard EJ, Anderson GD, Gregory SA, Isakson PC, Koboldt CM, Masferrer JL, Perkins WE, Seibert K, Zhang Y, Zweifel BS, Reitz DB (1996) Novel terphenyls as selective cyclooxygenase-2 inhibitors and orally active anti-inflammatory agents. J Med Chem 39:1846–1856
Li CS, Brideau C, Chan CC, Savoie C, Claveau D, Charleson S, Gordon R, Greig G, Gauthier JY, Lau CK, Riendeau D, Thérien M, Wong E, Prasit P (2003) Pyridazinones as selective cyclooxygenase-2 inhibitors. Bioorg Med Chem Lett 3:597–609
Malinka W, Redzicka A, Lozach O (2004) New derivatives of pyrrolo[34-d]pyridazinone and their anticancer effects. Il Farmaco 59:457–462
Malinka WL, Redzicka A, Więsek MJ, Filipek B, Dybała M, Karczmarzyk Z, Urbańczyk-Lipkowska Z, Kalicki P (2011) Derivatives of pyrrolo[34-d]pyridazinone a new class of analgesic agents. Eur J Med Chem 46:4992–4999
Marquet JP, Bisagni E, Andre-Louisfert (1968) J Chim Ther 3:348–355
McCall JM, Tenbrink RE, Kamdar BV, Shaletzky LL, Perricone SC, Piper RC, Delehanty PJ (1986) 7-(Trifluoromethyl)-4-aminoquinoline hypotensives: novel peripheral sympatholytics. J Med Chem 29:133–139
Meade EA, Worting LL, Drach JC, Townsend LB (1997) Synthesis and antiproliferative and antiviral activity of carbohydrate-modified pyrrolo[23-d]pyridazin-7-one nucleosides. J Med Chem 40:794–801
Miguel FB, Monica CMLG, Elena PM, Berta L, de Beatriz PT, Ana R, Nuria A, Francisco L, Dolores MM, Olivier L, Laurent M (2005) Pyrazolo[34-c]pyridazines as novel and selective inhibitors of cyclin-dependent kinases. J Med Chem 48:6843–6854
Mikashima H, Nakao T, Goto K, Ochi H, Yasuda H, Tsumagari T (1984) Y-590 (a new pyridazinone derivative) a potent anti-thrombotic agent—II Inhibition of platelet phosphodiesterase. Thromb Res 35(5):589–594
Mojahidul I, Anees AS, Ramadoss R (2008) Synthesis antitubercular antifungal and antibacterial activities of 6-substituted phenyl-2-(3′-substituted phenyl pyridazin-6′-yl)-2345-tetrahydropyridazin-3-one. Acta Pol Pharm 65(3):353–362
Murineddu G, Cignarella G, Chelucci G, Lorgia G, Pinna GA (2002) Synthesis and cytotoxic activities of pyrrole[23-d]pyridazin-4-one derivatives. Chem Pharm Bull 50:754–765
Murry MD, Brater DC (1993) Indomethacin and aspirin abolish prostaglandin release from the spleen. Annu Rev Pharmacol Toxicol 33:435–465
Nagai H, Suda H, Iwama T, Daikoku M, Yanagihara Y, Koda A (1992) Effect of NZ-107 a newly synthesized pyridazinone derivative on antigen-induced contraction of human bronchial strips and histamine release from human lung fragments or leukocytes. Int Arch Allergy Immunol 98(1):57–63
Ochiai K, Ando N, Iwase K, Kishi T, Fukuchi K, Ohinata A, Zushi H, Yasue T, Adams DR, Kohno Y (2011) Phosphodiesterase inhibitors. Part 2: design synthesis and structure–activity relationships of dual PDE3/4-inhibitory pyrazolo[15-a]pyridines with anti-inflammatory and bronchodilatory activity. Bioorg Med Chem Lett 21:5451–5463
Ochiai K, Takita S, Eiraku T, Kojima A, Iwase K, Kishi T, Fukuchi K, Yasue T, Adams DR, Allcock RW, Jiang Z, Kohno Y (2012) Phosphodiesterase inhibitors. Part 3: design synthesis and structure–activity relationships of dual PDE3/4-inhibitory fused bicyclic heteroaromatic-dihydropyridazinones with anti-inflammatory and bronchodilatory activity. Bioorg Med Chem 20:1644–1658
Okcelik B, Unlu S, Banoglu E, Kupeli E, Yesilada E, Sahin MF (2003) Investigations of new pyridazinone derivatives for the synthesis of potent analgesic and anti-inflammatory compounds with cyclooxygenase inhibitory activity. Arch Pharm (Weinheim) 336:406–412
Osadchii OE (2007) Myocardial phosphodiesterases and regulation of cardiac contractility in health and cardiac disease. Cardiovasc Drugs Ther 21:171–194
Ovais S, Javed K, Yaseen S, Bashir R, Rathore P, Yaseen R, Hameed AD, Samim M (2013) Synthesis antiproliferative and anti-inflammatory activities of some novel6-aryl-2-(p-methanesulfonyl) phenyl)-4 5-dihydropyridazi-3 (2H)-ones. Eur J Med Chem 67:352–365
Packer M (1988) Do positive inotropic agents adversely affect the survival of patients with chronic congestive heart failure? J Am Coll Cardiol 12:562–566
Pahernik SA, Schmid J, Sauter T, Schildberg FW, Koebe HG (1995) Metabolism of pimobendan in long-term human hepatocyte culture: in vivo–in vitro comparison. Xenobiotica 8:811–823
Pastelin G, Mendez R, Kabela E, Farah A (1983) The search for a digitalis substitute II milrinone (Win 47203) Its action on the heart-lung preparation of the dog. Life Sci 33:1787–1796
Penning TD, Talley JJ, Bertenshaw SR, Carter JS, Collins PW, Doctor S, Graneto MJ, Lee LF, Malecha JW, Miyashiro JM, Rogers RS, Rogier DJ, Yu SS, Anderson GD, Burton EG, Cogburn JN, Gregory SA, Koboldt CM, Perkins WE, Seibert K, Veenhuizen AM, Zhang YY, Isakson PC (1997) Synthesis and biological evaluation of the 15-diarylpyrazole class of cyclooxygenase-2-inhibitors. J Med Chem 40:1347–1365
Pham HC, Lasserre B, Pham HCA, de Palhares MAL, Tronche P, Couquelet J, Rubat CA (1988) Selective inhibitor of thromboxane synthetase activity of rabbit heart tissue: a pyridazinic derivative prostaglandins. Leukot Essent Fatty Acids 33(2):143–157
Piaz VD, Giovannoni MP, Ciciani G, Barlocco D, Giardina G, Petrone G, Clarke GD (1996) 45-Functionalized 6-phenyl-3(2H)-pyridazinones: synthesis and evaluation of antinociceptive activity. Eur J Med Chem 31:65–70
Piaz VD, Giovannoni MP, Castellana C, Palacios JM, Beleta J, Domenech T, Segarra V (1998) Heterocyclic fused 3(2H)-pyridazinones as potent and selective PDE IV inhibitors: further structure activity relationships and molecular modeling studies. Eur J Med Chem 33:789–797
Piaz DV, Vergelli C, Giovannoni MP, Scheider MA, Petrone G, Zaratin P (2003) 4-Amino-3(2H)-pyridazinones bearing arylpiperazinylalkyl groups and related compounds: synthesis and antinociceptive activity. Eur J Med Chem 58:1063–1071
Pieretti S, Piaz DV, Matucci R, Giovannoni MP, Galli A (1999) Antinociceptive activity of a 3(2H)-pyridazinone derivative in mice. Life Sci 65(13):1381–1394
Pinna GA, Curzu MM, Barlocco D, Cignarella G, Cavalletti E, Germini M, Berger K (1988) Synthesis and pharmacological study of 5-aryl-6-methyl-4,5-dihydro-pyridazin-3(2H)ones and related 5-aryl-6-methyl-pyridazin-3(2H)ones. Farmaco Sci 43(6):539–549
Pita B, Sotelo E, Suarez M, Ravina E, Ochoa E, Verdecia Y, Novoa H, Blaton N, de Ranter C, Peeters O (2000) Pyridazine derivatives part 21: synthesis and structural study of novel 4-aryl-25-dioxo-8-phenylpyrido[23-d]pyridazines. Tetrahedron 56:2473–2479
Ranju B, Dinesh K, Rosalia C, Carmen DLC, Dharam PJ (2008) Synthesis and evaluation of 2-substituted-6-phenyl-45-dihydropyridazin-3(2H)-ones as potent inodilators. Acta Pharm 58:393–405
Raskin JB (1999) Gastrointestinal effects of nonsteroidal antiinflammatory therapy. Am J Med 106(5B):3S–12S
Rathish IG, Javed K, Ahmad S, Bano S, Alam MS (1997) Synthesis and identification of 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzene sulphonamide (SC-58635 Celecoxib). J Med Chem 40:1347–1359
Rathish IG, Javed K, Bano S, Ahmad S, Alam MS, Pillai KK (2009) Synthesis and blood glucose lowering effect of novel pyridazinone substituted benzenesulfonylurea derivatives. Eur J Med Chem 44(6):2673–2678
Rathish IG, Javed K, Ahmad S, Bano S, Alam MS, Akhter M, Pillai KK, Ovais S, Samim M (2012) Synthesis and evaluation of anticancer activity of some novel 6-aryl-2-(p-sulfamylphenyl)-pyridazin-3 (2H)-ones. Eur J Med Chem 49:304–311
Robertson DW, Leander JD, Lawson R, Beedle EE, Clark CR, Potts BD, Parli CJ (1987a) Discovery and anticonvulsant activity of the potent metabolic inhibitor 4-amino-N-(26-dimethylphenyl)-35-dimethylbenzamide. J Med Chem 30(10):1742
Robertson DW, Jones ND, Krushinki JH, Polock GD, Scwartzendruber JK, Hayes JS (1987b) Molecular structure of the dihydropyridazinone cardiotonic 13-dihydro-33-dimethyl-5-(1456-tetrahydro-6-oxo-pyridazinyl)-2H-indol-2-one a potent inhibitor of cyclic AMP phosphodiesterase. J Med Chem 30:623–633
Rohet F, Rubat C, Coudert P, Albuisson E, Couquelet J (1996) Synthesis and trazodone-like analgesic activity of 4-phenyl-6-aryl-2-[3-(4-arylpiperazin-1-yl)propyl]pyridazin-3-ones. Chem Pharm Bull 44(5):980–986
Rubat C, Coudert P, Tronche P, Bastide J, Bastide P, Privat AM (1989) Synthesis and pharmacological evaluation of N-substituted 4,6-diaryl-3-pyridazinones as analgesic, antiinflammatory and antipyretic agents.Chem Pharm Bull 37(10):2832–2835
Rubat C, Coudert P, Albuisson E, Bastide J, Couquelet J, Tronche P (1992) Synthesis of Mannich bases of arylidenepyridazinones as analgesic agents. J Pharm Sci 81:1084–1087
Rüegg JC, Pfitzer G, Eubler D, Zeugner C (1984) Effect on contractility of skinned fibres from mammalian heart and smooth muscle by a new benzimidazole derivative45-dihydro-6-[2-(4-methoxyphenyl)-1H-benzimidazol-5-yl]-5-methyl-3(2H)-pyridazinone. Arzneimittelforschung 34(12):1736–1738
Santagati NA, Duro F, Caruso A, Trombadore S, Amico-Roxas M (1985a) Synthesis and pharmacological study of a series of 3(2H)-pyridazinines as analgesic and anti-inflammatory agents. Farmaco 40:921–929
Santagati NA, Duro F, Caruso A, Trombadore S, Amico-Roxas M (1985) Synthesis and analgesic and anti-inflammatory activities 6-substituted-3(2H)-pyridazinone-2-acetyl-2-(p-substituted/nonsubstitute d benzal) hydrazone derivatives. IL Farmaco Ed Sci 40:921–929
Sato M (1979) Pharmacological studies of 4-ethoxy-2-methyl-5-morpholino-3(2H)-pyridazinone (M73101) (5) Action of M73101 on the central nervous system (author’s transl). Nippon Yakurigaku Zasshi 75(7):695–706
Sato M, Ishizaku Y, Yamaguchi A (1981) Pharmacological investigations of 4-ethoxy-2-methyl-5-morpholino-3(2H)-pyridazinone (M 73101) a new analgesic and antiinflammatory drug. Arzneim Forsch 31:1738–1745
Sayed GH, Sayed MA, Mahmoud MR, Shaaban SS (2002) Synthesis and reactions of new pyidazinone derivatives of expected antimicrobial activities. Egypt J Chem 45:767–776
Schneider P, Güttner J, Eckenfels A, Heinzel G, von Nicolai H, Trieb G, Lehmann H (1979) Comparative cardiac toxicity of the iv administered benzimidazole pyridazinon derivative Pimobendan and its enantiomers in female Beagle dogs. Exp Toxicol Pathol 49(3–4):217–224
Schudt C, Winder S, Müller B, Ukena D (1991) Zardaverine as a selective inhibitor of phosphodiesterase isozymes. Biochem Pharmacol 42(1):153–162
Seki T, Nakao T, Masuda T, Hasumi K, Gotanda K, Ishimori T, Honma S, Minami N, Shibata K, Yasuda K (1996) Studies on agents with vasodilator and beta-blocking activities IV. Chem Pharm Bull 44(11):2061–2069
Siddiqui AA, Wani SM (2004) Isolation of phytoconstltuentand hypotensive activity of Terminalia arjuna bark. Indian J Heterocycl Chem 13:115–123
Siddiqui AA, Syed RA, Mohammed SM, Syed AH, Mohammed R, Ravindra K (2008) Synthesis and in-vitro antifungal activity of 6-substituted-phenyl-2-{[(4′-substituted phenyl-5′-thioxo)-124-triazol-3-yl]-methyl}-2,3,4,5-tetrahydropyridazin-3-one derivatives. Acta Pol Pharm 65(2):223–228
Siddiqui AA, Mishra R, Shaharyar M (2010) Synthesis characterization and antihypertensive activity of pyridazinone derivatives. Eur J Med Chem 45:2283–2289
Sonmez M, Berber I, Akbas E (2006) Synthesis antibacterial and antifungal activity of some new pyridazinone metal complex. Eur J Med Chem 41:101–105
Sotelo E, Pita B, Ravina E (2000) Pyridazines. Part 22: highly efficient synthesis of pharmacologically useful 4-cyano-6-phenyl-5-substituted-3(2H)-pyridazinones. Tetrahedron Lett 41:2863–2866
Sotelo E, Fraiz N, Yanez M, Terrades V, Laguna R, Cano E, Ravina E (2002) Pyridazines. Part XXIX: synthesis and platelet aggregation inhibition activity of 5-substituted-6-phenyl-3(2H)-pyridazinones novel aspects of their biological action. Bioorg Med Chem 10:2873–2895
Sotelo E, Coelho A, Ravina E (2003) Pyridazines. Part 34: retro-ene-assisted palladium-catalyzed synthesis of 45-disubstituted-3(2H)-pyridazinones. Tetrahedron Lett 44:4459–4462
Strader CD, Sigal IS, Dixon RAF (1989a) Genetic approaches to the determination of structure–function relationships of G protein-coupled receptors. Trends Pharmacol Sci 26:30–39
Strader CD, Sigal IS, Dixon RAF (1989b) Structural basis of beta-adrenergic receptor function. FASEB J 3:1825–1832
Strappaghetti G, Barbaro R, Marucci G (2000) Synthesise and α1-antagonist activity of derivatives of 4-chloro-5-{4-[2-(2-methoxyphenoxy)-ehyl]-1-piperazinyl}-3(2H)-pyridazinone. Eur J Med Chem 35:773–779
Sureea N, Yi SW, Thieu W, Marohn M, Damoiseaux R, Chana A, Junga ME, Clubb RT (2009) Discovery and structure–activity relationship analysis of Staphylococcus aureus sortase A inhibitors. Bioorg Med Chem 17(20):7174–7185. doi:10.1016/jbmc200908067
Takaya M, Sato M, Terashima K, Tanizawa HA (1979) New nonsteroidal analgesic antiinflammatory agent synthesis and activity of 4-ethoxy-2-methyl-5-morpholino-3(2H) pyridazinone and related compounds. J Med Chem 22(1):53–58
Talley JJ, Brown DL, Carter JS, Graneto MJ, Koboldt CM, Masferrer JL, Perkins WE, Rogers RS, Shaffer AF, Zhang YY, Zweifel BS, Seibert K (2000) 4-[5-Methyl-3-phenylisoxazol-4-yl]-benzenesulfonamide valdeCOXib: a potent and selective inhibitor of COX-2. J Med Chem 43:775–777
Thyes M, Lehmann HD, Gries J, Koing H, Kretzschmar R, Kunze J, Lebkucher R, Lenke D (1983) 6-Aryl-45-dihydro-3(2H)-pyridazinones. A new class of compounds with platelet aggregation inhibiting and hypotensive activities. J Med Chem 26:800–807
Ton-That H, Marraffini LA, Schneewind O (2004) Protein sorting to the cell wall envelope of Gram-positive bacteria. Biochim Biophys Acta 269:1694–1708
Torphy TJ, Undem BJ, Cieslinski LB, Luttmann MA, Reeves ML, Hay DW (1998) Identification, characterization and functional role of phosphodiesterase isozymes in human airway smooth muscle. J Pharmacol Exp Ther 265:1213
Uhlmann R, Schwinger RH, Lues I, Erdmann E (1995) EMD 53998 acts as Ca(2+)-sensitizer and phosphodiesterase III-inhibitor in human myocardium. Basic Res Cardiol 90(5):365–371
van Meel JCA (1985) Cardiovascular effects of the positive inotropic agents pimobendan and sulmazole in vivo. Arzneim Forsch 35:284–295
Verdouw PD, Hartog JM, Duncker DJ, Roth W, Saxena PR (1986) Cardiovascular profile of pimobendan a benzimidazole-pyridazinone derivative with vasodilating and inotropic properties. Eur J Pharmacol 126(1–2):21–30
Viaud MC, Jamoneau P, Flouzat C, Bizod-Espiard JG, Pfeiffer B, Renard P, Caignard DH, Adam G, Guillaument G (1995) N-substituted oxazolo[5,4-b]pyridin-2(1H)-ones: a new class of non-opiate antinociceptive agents. J Med Chem 38(8):1278–1286
Walker SS, Xu YT, Waldman I, Mendrick MF, Brown C, Mann N, Chau P, Patel A, Bauman R, Norris N, Antonacci C, Gurnani B, Cacciapuoti M, McNicholas A, Wainhaus PM, Herr SJR, Kuang R, Aslanian RG, Ting PC, Black TA (2011) Discovery of a novel class of orally active antifungal β-13-d-glucan synthase inhibitors. Antimicrob Agents Chemother 55:5099–5106
Wang T, Dong Y, Wang LC, Chen Z (2007) Synthesis and bioactivity of 6-phenyl-45-dihydro-3(2H)-pyridazinonederivatives. Arzneimittelforschung 57(10):641–646
Wang T, Dong Y, Wang LC, Xiang BR, Chen Z, Qu LB (2008) Design synthesis and structure–activity relationship studies of 6-phenyl-45-dihydro-3(2H)-pyridazinone derivatives as cardiotonic agents. Arzneimittel forschung 58(11):569–573
Willemot C (1997) Simultaneous inhibition of linolenic acid synthesis in winter wheat roots and frost hardening by BASF 13-338 a derivative of pyridazinone. Plant Physiol 60(1):1–4
Xing W, Yan Fu, Shi Z, Dong Lu, Zhang H, Hu Y (2013) Discovery of novel 26-disubstituted pyridazinone derivatives as acetylcholinesterase inhibitors. Eur J Med Chem 63:95–103
Xu P, Wang SY, Chen Y, Liu WQ, Tao C (1991) Studies on synthesis anticonvulsant activity and the structure–activity relationships of 6-(substituted phenyl)-3 (2H) pyridazinones. Yao Xue Xue Bao 26(9):656–660
Yamamoto A, Iwama T, Takeda H, Nagai H (1995) Effects of NIP-502 on antigen-induced bronchial responses and allergic reactions in animal models. Jpn J Pharmacol 68(1):47–55
Zeng GQ, Wang XS, Sun DX, Lin AY, Ji Y, Chu JX, Rui YC (1993) Effects of 6-(alpha alpha-diphenylacetylpiperazinyl)phenyl-5-methyl-45-dihydro-3(2H)pyridazinone on rabbit platelet aggregation and TXB2 cAMP production. Yao Xue Xue Bao 28(4):25659
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dubey, S., Bhosle, P.A. Pyridazinone: an important element of pharmacophore possessing broad spectrum of activity. Med Chem Res 24, 3579–3598 (2015). https://doi.org/10.1007/s00044-015-1398-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-015-1398-5