Abstract
Objective
The present study aimed to comprehensively investigate the changes in oxylipins during murine sepsis induced by lipopolysaccharide (LPS) or cecal ligation and puncture (CLP).
Methods
Twenty-four hours after induction of sepsis in male C57BL/6 mice by LPS or CLP, plasma and liver, lung, kidney and heart tissues were sampled. Oxylipin levels in plasma and tissue were quantified by means of LC–MS. Moreover, clinical chemistry parameters were determined in plasma and interleukin levels (MCP-1 and IL-6) were determined in kidney and liver.
Results
Elevation of liver function plasma parameters at 24 h revealed that both models were successful in the induction of sepsis. LPS induced sepsis resulted in a dramatic increase of plasma PGE2 (2,100 % change in comparison to control) and other cyclooxygenase metabolites, whereas this effect was less pronounced in CLP induced sepsis (97 % increase of PGE2). Plasma epoxy-fatty acids (FAs) and hydroxy-FAs and most of the dihydroxy-FAs were elevated in both models of sepsis. Changes of tissue oxylipin concentrations were organ dependent. Only few changes were detected in the lung and liver tissue, epoxy-FAs were elevated in the kidney. In the heart tissue a trend towards lower levels of hydroxy-FAs and epoxy-FAs was observed.
Conclusion
Both murine models of sepsis are characterized by changes of oxylipins formed in all branches of the arachidonic acid (AA) cascade. The more pronounced effects in the LPS model make this model more suitable for the investigation of the AA cascade and its pharmacological modulation in sepsis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sepsis is a severe medical condition with high incidence and mortality [1–3]. Sepsis arises when a host is not able to contain an infection and is characterized by a complex deregulation of inflammation resulting in multiple organ failure [4]. Aside from cytokines the pro-inflammatory prostaglandins (PGs), especially PGE2, play a role in sepsis [5]. Plasma PG levels are increased in several animal models of sepsis [6–9] and patients [10, 11]. PGs modulate a variety of biological functions and are enzymatically formed from arachidonic acid (AA) or other polyunsaturated fatty acids (FAs) by cyclooxygenases (COXs) and several PG-synthases [12]. For example, PGE2 plays a role in the regulation of immune response, blood pressure, inflammation and pain [13]. PGI2 is a potent vasodilator and inhibitor of platelet aggregation and therefore important for cardiovascular homeostasis [13]. 6-Keto-PGF1α is its inactive, non-enzymatically formed hydrolysis product and used as marker for the in vivo production of PGI2 [13]. 13,14-Dihydro-15-keto-PGF2α is a plasma metabolite of PGF2α, which is involved in several physiological processes, such as luteolysis, ovarian function, parturition as well as in acute and chronic inflammation and cardiovascular diseases [14, 15]. Apart from the COX branch, polyunsaturated FAs are converted in the AA cascade enzymatically by lipoxygenases (LOXs) and cytochrome-P450-monooxygenases (CYPs) or non-enzymatically via autoxidation. Conversion by enzymes of the CYP2C or CYP2J families results in the formation of epoxy-FAs with vasodilatory, anti-inflammatory, and analgetic properties and which are subsequently hydrolyzed by soluble epoxide hydrolase (sEH) to the less biologically active dihydroxy-FAs [16, 17]. ω-Hydroxylation of AA by members of the CYP4A or CYP4F families form 20-HETE, acting as a vasoconstrictor [18]. The primary products of LOXs are hydroperoxy-FAs, which can be subsequently reduced to the corresponding hydroxy-FAs, such as 5-, 12- or 15-HETE or the eicosapentaenoic acid (EPA) derived 5-, 12- or 15-HEPE [19]. These hydroxy-FAs can be used as markers for 5-, 12- or 15-LOX activity. Under physiological conditions, 5-HpETE can be further processed by 5-LOX to LTA4 which serves as precursor for several highly biological active lipid mediators, such as the chemoattractant LTB4 or the anti-inflammatory LXA4 [19]. Moreover, resolvins and related compounds, which are believed to be involved in the resolution of inflammation, are formed by the action of COX and LOX and primary products such as 18-HEPE and 17-HDHA are used as markers for this pathway [20]. Oxygenated FAs can also be formed via autoxidation. Whereas enzymatic oxidation is stereoselective, autoxidation results in the formation of racemic mixtures. Primary autoxidation products are hydroperoxides, which can be reduced to hydroxy-FAs, such as 9- or 11-HETE [21]. Further reaction of the initially formed hydroperoxides results in the formation of F2-isoprostanoids, such as 5-iPF2α-IV, which are used as markers of oxidative stress in vivo [22].
Previous studies investigating the role of lipid mediators, generally referred as oxylipins, in sepsis focused only on single or few compounds [7–9, 23]. Although there is a crosstalk between different pathways [24, 25], comprehensive information about sepsis related changes in all pathways is scarce. Particularly, no data on differences in oxylipin pattern induced by different animal models of sepsis is available. Initiation of sepsis in animal models can be achieved via three strategies: (1) administration of exogenous toxins [such as lipopolysaccharide (LPS)], (2) administration of viable pathogens or (3) by an alteration of the intestinal barrier [4, 26]. LPS-induced sepsis and polymicrobial sepsis induced by cecal ligation and puncture (CLP) are the most commonly used animal models. LPS-induced sepsis can be simply and robustly archived by i.p. injection and is characterized by a rapid and massive elevation of systemic cytokines, such as TNF-α, IL-1β and IL-6 as well as PG levels [8, 26]. Notably, in clinical sepsis the increase of systemic cytokine levels is more prolonged and the concentrations are lower. Thus, LPS-induced sepsis gives insights in septic processes albeit there are limitations regarding the complex physiological response in septic patients [4, 26]. The CLP model is considered to be the gold standard in sepsis models. In CLP surgery the intestinal barrier is damaged, allowing bacterial translocation which leads to peritonitis and eventually multiorgan failure. The cytokine release after CLP surgery is prolonged and resembles the cytokine profile observed in septic patients [4, 26, 27].
This study aims to comprehensively investigate and compare the role of lipid mediators in these two models of sepsis. For this purpose, both oxylipin plasma as well as tissue levels were analyzed using a targeted metabolomics approach and correlated with clinical chemistry parameters indicating multiorgan failure due to acute sepsis.
Materials and methods
Chemicals
LPS from E. coli O111:B4 was purchased from Sigma (Schnelldorf, Germany, L 2630 Lot#043M4104 V). Oxylipin standards and internal standards were purchased from Cayman Chemicals (local distributor: Biomol, Hamburg). Epoxy octadecadienoic acids (EpODEs) and dihydroxy octadecadienoic acids (DiHODEs), 1-(1-methanesulfonyl-piperidin-4-yl)-3-(4-trifluoromethoxy-phenyl)-urea (TUPS) were a kind gift from the laboratory of Bruce Hammock (UC Davis, CA, USA). LC–MS grade acetonitrile, acetic acid (HAc) and methanol (MeOH) were obtained from Fisher Scientific (Nidderau, Germany). n-Hexane (HPLC grade) was from Carl Roth (Karlsruhe, Germany). All other chemicals were from Sigma (Schnelldorf, Germany).
In vivo studies
C57BL/6 male mice (H2b, 11–13 weeks of age) were obtained from Charles River (Sulzfeld, Germany). Animals were cared for in accordance with the institution’s guidelines for experimental animals and with the guidelines of the American Physiological Society. The animal protection committee of the local authorities (Lower Saxony state department for food safety and animal welfare LAVES) approved all experiments (approval: 33.9-42502-04-12/0846). Mice were housed under conventional conditions in individually ventilated cages produced by Tecniplast Inc. (Italy) with a 12 h light/dark cycle and had free access to food (Altromin 1324 standard mouse diet) and domestic quality drinking water ad libitum.
LPS model
C57BL/6 mice were i.p. challenged with 10 mg/kg BW LPS (n = 6) or vehicle (n = 6). After 24 h mice were anesthetized with isoflurane for EDTA-blood sampling from the retro orbital venous plexus using EDTA coated capillaries. Plasma was generated from blood immediately by centrifugation (10 min, 4,000 g) and stored at −80 °C till analysis. After perfusion with ice cold PBS lung, liver, kidney and heart tissue was collected. Tissue was immediately shock frozen in liquid nitrogen and stored at −80 °C till analysis.
Cecal ligation puncture (CLP)
CLP surgery was performed under isoflurane anesthesia as described previously [28]. Briefly, after incision of the left upper quadrant of the peritoneal cavity the cecum was exposed and a tight ligature was placed around the cecum distal to the insertion of the small bowel. One puncture wound was made with a 24-gauge needle into the cecum and small amounts of cecal content were expressed through the wound, and 500 µL sterile normal saline solution was flushed into the abdomen. The cecum was placed back into the peritoneal cavity and the laparotomy site was closed in two layers. Finally, animals were returned to their cages with free access to food and water.
Blood sampling and clinical chemistry
Several days prior to sepsis induction and 24 h afterwards EDTA blood was drawn from the retro orbital venous plexus. After 10 min centrifugation at 4,000 g plasma was obtained and stored at −20 °C to be used for clinical chemistry. Plasma urea, creatinine, aspartate transaminase (AST), alanine transaminase (ALT) and lactate dehydrogenase (LDH) levels were determined by using the fully automated Olympus AU 400 analyzer (Beckman Coulter Inc.). Statistical differences between groups were determined by one-way ANOVA followed by Tukey post test.
Oxylipin analysis
Oxylipin analysis in plasma and tissue was carried out by solid phase extraction (SPE) followed by LC–MS as described with slight changes [29]. In brief, internal standards and 10 µL of an antioxidant/inhibitor solution [0.2 mg/mL EDTA, 0.2 mg/mL butylated hydroxytoluene, 100 µM indomethacin, 100 µM TUPS in MeOH/water (50/50 v/v)], 480 µL H2O (pH 6) and 120 µL MeOH were added to 200 µL of plasma. Tissue samples were homogenized in 250 µL methyl formate in 1.5 mL tubes with a 3 mm metal bead using a ball mill (5 min, 25 Hz, Retsch, Haan, Germany) following addition of internal standards and antioxidant/inhibitor solution. After centrifugation, the supernatant was diluted with H2O (pH 6) to a total volume of 6 mL. Extraction was carried out on Chromabond C18 ec cartridges (500 mg, Macherey–Nagel, Düren, Germany) preconditioned with 2 column volumes of methyl formate, 1 column volume of MeOH and 3 column volumes of H2O (pH 6). Directly before plasma and tissue samples were loaded onto the SPE cartridge, samples were acidified with 80 µL HAc resulting in a pH of 3. After loading the sample, the cartridge was washed with 10 mL H2O (pH 6) and 6 mL n-hexane. The cartridge was dried for 20 min at −200 mbar. The analytes were then eluted with 8 mL methyl formate in glass tubes containing 6 µL of 30 % glycerol in MeOH. Utilizing a Speedvac (Christ, Osterode, Germany), the extract was evaporated to dryness until only the glycerol plug was left. The residue was dissolved in 50 µL methanol. After centrifugation, 5 µL of the supernatant was injected to the LC–MS system. Mass spectrometric detection after electrospray ionization using an AB Sciex 6500 QTRAP instrument (AB Sciex, Darmstadt, Germany) was performed as described [29, 30] ). A list of all oxylipins covered by the method can be found in the SI (Table S1). Multiquant (Sciex) was used for peak integration and determination of oxylipin concentration.
Determination of transcription of pro-inflammatory sepsis associated cytokines IL-6 and MCP-1 in kidney and liver tissue
Total mRNA was extracted using the RNeasy mini kit system (Qiagen, Hilden, Germany) and transcribed using Superscript II Reverse transcriptase (Invitrogen). Quantitative (q) PCR was performed on a Lightcycler 420 II (Roche Diagnostics, Penzberg, Germany) using FastStart Sybr-Green chemistry. Gene-specific primers for IL-6 (Quantitec QT00098875, Qiagen) and MCP-1 (Quantitec QT00167832, Qiagen) were used for the genes of interest and HPRT served as housekeeping gene for normalization (Quantitec QT00166768, Qiagen). Quantification was carried out using QGene software.
Data analysis
Data analysis was performed using GraphPad Prism 5.0 (GraphPad Software, San Diego, USA). For the clinical chemistry parameters statistical differences between groups and both time points were determined by two-way ANOVA followed by Bonferroni post test. Regarding oxylipin concentrations and cytokine levels statistical differences between groups were determined by one-way ANOVA followed by Tukey post test.
Results
The clinical chemistry parameters revealed that in both models sepsis with impairment of kidney and liver function was successfully induced. In comparison to the vehicle group with a bodyweight of 23.2 ± 0.5 g, the LPS group had a significantly reduced bodyweight of 21.8 ± 0.2 g 24 h after LPS treatment (p < 0.05). 24 h after CLP the weight was also lower, however the difference between sham (24.1 ± 0.5 g) and the CLP (22.8 ± 0.5 g) group did not reach statistical significance. Plasma creatinine, urea, AST, ALT and LDH levels at baseline and 24 h after treatment/surgery are shown in Fig. 1. Plasma creatinine levels of the LPS group significantly increased from 28.5 ± 1.3 to 54.1 ± 7.1 µmol/L 24 h after LPS treatment (p < 0.001). Additionally, an about fivefold increase of plasma urea concentration was observed (p < 0.001). Plasma activity of AST was significantly increased from 60 ± 7.2 to 216 ± 19.8 U/L (p < 0.05), while ALT activity showed no statistically significant changes 24 h after LPS treatment. Plasma LDH levels almost doubled in the LPS treated animals indicating substantial cell damage (p < 0.05). CLP resulted in increased plasma urea levels (baseline: 8.1 ± 0.36 mmol/L, 24 h: 16.8 ± 5.20 mmol/L, p < 0.05) but the effect was less pronounced as after LPS treatment (Fig. 1). Plasma creatinine levels were not affected by CLP. ALT and AST activities were elevated 24 h after CLP surgery: ALT activity was increased more than tenfold (p < 0.001), whereas AST activity changed from 77 ± 6.5 to 494 ± 89.1 U/L (p < 0.001, Fig. 1). As observed in a similar manner for LPS treatment, plasma LDH level raised from 744 ± 59.7 to 1151 ± 257 U/L 24 h after CLP surgery (p < 0.05).
Clinical chemistry of liver and renal function parameters prior to the experiment (baseline, light grey bars) and 24 h after induction of sepsis by LPS or CLP (24 h, dark grey bars). Shown are mean ± SEM. Statistical differences were determined by two-way ANOVA followed by Bonferroni post test (*p < 0.05, **p < 0.01, ***p < 0.001)
Pro-inflammatory tissue cytokines
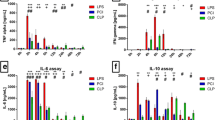
The most relevant pro-inflammatory cytokine in sepsis is IL-6 which was significantly increased in both sepsis models in kidney as well as in liver tissue (Fig. S1). The IL-6 elevation in the LPS model was significantly higher compared to the CLP model. The chemoattractant and pro-inflammatory cytokine MCP-1 was also elevated in the liver of both sepsis models compared to the controls. However, the difference between the LPS and CLP model was less pronounced and significantly higher elevation of MCP-1 was measured due to LPS injection (Fig. S1).
Prostaglandins
Plasma concentrations of all detected oxylipins (concentration > LOQ) are shown in Table S2. In the LPS treated group PGE2 and its analog derived from dihomo-γ-linoleic acid (DGLA 20:3, n-6), PGD2 and its analog from EPA, the PGI2 metabolite 6-keto-PGF2α, PGF2α and its analog from DGLA as well as its metabolite 13,14-dihydro-PGF2α, the adrenic acid (22:4 n-6) derived 1a,1b-dihomoPGF2α and 5-iPF2α were elevated in comparison to the vehicle. The increase was statistically significant (p < 0.05) for PGE2, PGE1, 6-keto-PGF1α, 5-iPF2α, dihomo-PGF2α, 13,14-dihydro-15-keto-PGF2α, PGF1α and 13,14-dihydro-15-keto-PGE1 (Table S2; Fig. 2). For example, the PGE2 plasma level was found to be 3.9 ± 0.92 nM in the LPS group in comparison to 0.18 ± 0.048 nM in the vehicle group (p < 0.001), corresponding to an increase of 2,100 %. The effect of LPS treatment on PG tissue levels was less pronounced. In the liver most of the PG concentrations were unchanged between the LPS and vehicle group, only PGE2 and 5-iPGF2α were detected at statistically significant different concentrations (Fig. 2; Table S3). In the kidney increased levels of PGD1, dihomo-PGF2α and PGF1a were observed in the LPS group (Table S4), while the concentrations of the other detected PGs were unchanged. 6-Keto PGF1α, 13,14-dihdydro-15-keto-PGE1 (p < 0.001) and 13,14-dihydro-15-keto-PGF2α (p < 0.05) were found to be elevated in the heart (Table S5). In lung tissue no changes in the detected PGs were observed (Table S6).
PGE2 and PGE1 levels in plasma and different tissues 24 h after induction of sepsis by LPS i.p. injection or CLP surgery. Shown are mean ± SEM. Statistical differences were evaluated by one-way ANOVA followed by Tukeys post test and shown for LPS versus vehicle and CLP versus sham (*p < 0.05, **p < 0.01, ***p < 0.001)
Twenty-four hours after inducing sepsis by CLP slightly elevated levels of PGE2 (CLP: 0.44 ± 0.04 nM; sham: 0.22 ± 0.02 nM), 6-keto-PGF1α (CLP: 3.1 ± 1.2 nM; sham: 0.81 ± 0.02 nM) and 13,14-dihdydro-15-keto-PGE1 [CLP: 0.28 ± 0.066 nM; sham: < LOQ (0.036 nM)] were observed in the treatment group (Table S2). Except for PGD2, the concentrations of the other detected PGs were not different between CLP and sham control. Interestingly, PGD2 levels were decreased in the CLP group (0.75 ± 0.11 nM, p < 0.01) compared to the sham group (4.2 ± 1.1 nM). PGD2 levels in the heart tissue showed the same trend: in the CLP group lower PGD2 concentrations (8.5 ± 1.0 fmol/mg) were detected compared to the sham group (20 ± 4.7 fmol/mg, p < 0.05, Table S5). For all other PGs no obvious changes in the tissue levels were observed (Table S3–S6).
Hydroxy-FA
LPS treatment showed different effects on the hydroxy-FAs. Plasma levels of 8-, 12- and 20-HETE, 15-HETrE and 10-, 14- and 17-HDHA were increased 24 h after LPS treatment in comparison to the vehicle group (p < 0.05, Table S2). A consistent trend towards higher concentrations in the LPS treated group was found for 8-HEPE, 12-HETE and 15-HEPE and 13-HDHA (Table S2; Fig. 3a, b). 5-HEPE and 9-HOTrE were detected at slightly lower levels after treatment, whereas the other detected hydroxy-FAs, such as 5- and 9-HETE, 18-HEPE and 4-, 7-, 8- and 11-HDHA were found to be unaffected (Table S2). In the liver tissue the levels of 20-HETE, 10- and 20-HDHA were increased by 53–230 % in the LPS group (p < 0.05, Table S3). 12-HETE, 12-HEPE and 14-HDHA showed also trends towards higher levels in the LPS group. Interestingly the concentrations of 5- and 15-HEPE were decreased by about 50 % in the LPS treated animals (p < 0.05, Table S3). The concentrations of other hydroxy-FAs, such as 9- and 13-HOTrE, 5-, 8-, 9-, 11- and 15-HETE, 18- and 20-HEPE and 4-, 7-, 8- and 11-HDHA were unchanged (Table S3). Similar observations were found in the kidney: 5-HEPE concentration was about 20 % lower in the LPS group (p < 0.001), whereas concentrations of other hydroxy-FAs, e.g. 20-HETE, 9- and 13-HODE and 7-, 10- and 11-HDHA were elevated (p < 0.05, Table S4). 12-HETE, 12-HEPE and 14-HDHA were found at 140–310 % higher concentrations in the LPS group (Table S4), most other hydroxy-FAs, such as 9- and 13-HOTrE, 5-, 8-, 9-, 11- and 15-HETE, 18- and 20-HEPE and 4- and 8-HDHA, were unaffected. Only a minor modulation of the hydroxy-FAs concentrations in the lung tissue was observed in the sepsis models. Following LPS treatment, only 5-HEPE, 20-HETE and 7-HDHA were elevated in the LPS group (p < 0.05, Table S6) and for 5-HETE a trend towards higher levels was observed.
Plasma concentrations of selected metabolites of the LOX and CYP branch of the AA cascade 24 h after induction of sepsis by i.p. LPS injection or CLP surgery. Shown are mean ± SEM. Statistical differences were determined by one-way ANOVA followed by Tukeys post test and shown for LPS versus vehicle and CLP versus sham (*p < 0.05, **p < 0.01, ***p < 0.001)
Interestingly, the change in the hydroxy-FAs pattern in the heart revealed a different picture (Table S5): most of the hydroxy-FAs, e.g. 13-HODE, 5-HETE and 4- and 11-HDHA showed a trend towards about 30 % lower levels in the LPS group, reaching statistical significance (p < 0.05) for 5- and 12-HEPE, 12-HETE and 10-, 14- and 17-HDHA.
Similar to LPS treatment, CLP induced sepsis resulted in different changes in plasma hydroxy-FA concentrations. 9-HOTrE and 8-, 12- and 20-HETE were significantly elevated (83–300 %, p < 0.05). A similar trend of increased levels was observed for 8- and 12-HETE, 8-, 12- and 15-HEPE, 15-HETrE and 10-, 13-, 14- and 17-HDHA (200–1,200 %, Table S2; Fig. 3). The concentration of 9-HOTrE was decreased by 71 % in the CLP group (p < 0.01). Levels of 5-, 9- and 11-HETE, 18-HEPE and 9- and 13-HODE were not affected by CLP induced sepsis (Table S2).
Consistent with the LPS model the concentration of 5-HEPE was decreased (p < 0.01) in the liver tissues of the CLP treated animals. Additionally, 9-HOTrE was also found at 47 % lower levels while concentrations of 12-HETE, 12-HEPE and 10- and 12-HDHA were elevated by 99–580 % during CLP induced sepsis (p < 0.05). The concentrations of the other hydroxy-FAs detected in the liver, e.g. 9- and 13-HODE, 5-, 8-, 9-, 11-, 15-HETE, 8-, 18- and 20-HEPE and 4-, 7-, and 8-HDHA were unchanged (Table S3). Similar to liver tissue, kidney 5-HEPE level is decreased in CLP induced sepsis (18 %, p < 0.01). Except for 16-HDHA (increase by 48 %, p < 0.05) and 14-HDHA (increase by 200 %) which concentrations were elevated, other hydroxy-FAs detected were unaffected, e.g. 8-, 9-, 11-, 12, -15-HETE and 4-, 7-, 8- and 11-HDHA (Table S4). In accordance to the LPS model, a trend towards lower hydroxy-FA levels in the heart was observed in CLP induced sepsis. The decrease was significant for 9- and 13-HOTrE, 9- and 13-HODE, 5-HEPE, 5- and 9-HETE and 11-HDHA (p < 0.05, Table S5). CLP surgery had no effect on the hydroxy-FA concentrations in the lung (Table S6).
Epoxy-FA
The plasma epoxy-FA levels during LPS induced sepsis were either increased or unchanged. 9(10)- and 12(13)-EpODE, 9(10)- and 12(13)-EpOME and 19(20)-EpDPE were found at 62–170 % higher concentrations 24 h after LPS treatment (p < 0.05, Table S2). The levels of other detected epoxy-FAs, such as 15(16)-EpODE, 5(6)-, 8(9)-, 11(12)-, 14(15)-EpETrE, 8(9)-, 14(15)-EpETE were unchanged (Fig. 3c, d; Table S2). In the kidney the effect of LPS on epoxy-FA levels was more pronounced: 14 out of 18 detected epoxy-FA concentrations were changed after LPS treatment (p < 0.05). Interestingly, the AA-derived epoxides 8(9)-, 11(12)- and 14(15)-EpETrE were decreased by 13–25 %, whereas most epoxy-FAs, e.g. 9(10)-, 12(13)- and 15(16)-EpODE, 9(10)- and 12(13)-EpOME, 10(11)-EpDPE or 11(12)- and 14(15)-EpETE were found at 11–87 % higher concentrations (p < 0.05, Table S4). Only 17(18)-EpETE, 5(6)-EpETrE, 8(9)-EpETE and 9(10)-epoxystearic acid kidney levels were not affected by LPS treatment. Heart tissue epoxy-FA concentrations after LPS treatment were decreased by about 35 % for the DHA derived epoxides 10(11)-, 13(14)- and 16(17)-EpDPE (p < 0.05). The effects of LPS treatment on lung epoxy-FA levels were marginal, only 5(6)-EpETrE was found in an about twofold higher concentration (p < 0.05). In the liver no changes of epoxy-FA concentrations were detected.
The CLP surgery showed only effects on a single plasma epoxy-FA level: 16(17)-EpDPE was increased in the treatment group (55 %, p < 0.001). The other epoxy-FAs were unchanged and no obvious trend could be observed (Fig. 3c, d; Table S2). Similar to LPS induced sepsis, there was a trend towards decreased epoxy-FA levels in the heart 24 h after CLP. These differences were found to be statistically significant for the ALA derived epoxy metabolites 9(10)-, 12(13)- and 15(16)-EpODE (decrease about 60 %, p < 0.05, Table S5). Regarding the epoxy-FA concentrations in liver, lung and kidney no changes by the CLP surgery were detected (Table S3, 4, 6).
Dihydroxy-FA
LPS treatment resulted in increased plasma dihydroxy-FA levels. Out of 20 detected dihydroxy-FAs 12 were significantly elevated by 57–170 % (p < 0.05), e.g. 8,9-; 11,12-; and 14,15-DiHETrE, 10,11-; 13,14- and 16,17-DiHDPE and 14,15- and 17,18-DiHETE (Fig. 3; Table S2). In kidney all dihydroxy metabolites derived from LA and DHA were monitored at 49–101 % higher concentrations in the LPS treated group (p < 0.05, Table S4). Dihydroxy-FAs derived from AA, ALA and DHA showed no significant change with a slight trend towards higher levels in LPS induced sepsis. Dihydroxy-FA levels in the lung were elevated in the LPS group, reaching statistically significance for 9,10- and 12,13-DiHODE, 9,10-DiHOME, 4,5-; 7,8- and 10,11-DiHDPE and 5,6-, and 8,9-DiHETrE (Table S5). In contrast to the other oxylipins (see above) detected in the heart tissue, dihydroxy-FA levels of 4,5-, 10,11-, 13,14- and 19,20-DiHDPE and 8,9- and 14,15-DiHETrE were increased by 22–78 % in the LPS treated group (p < 0.05, Table S5). The dihydroxy-FA metabolites in the liver were not affected by LPS treatment (Table S3).
Dihydroxy-FA levels in plasma showed a trend towards higher concentrations in CLP induced sepsis. 8 out of 20 detected plasma dihydroxy-FA levels were significantly elevated by 72–170 % after CLP surgery (9,10 and 12,13-DiHOME, 11,12- and 14,15-DiHETrE; 10,11-; 13,14-; 16,17- and 19,20-DiHDPE, p < 0.05, Fig. 3e, f; Table S2). In the kidney 14,15-DiHETE, 9,10-DiHOME and 4,5-; 10,11-; 13,14-; 16,17- and 19,20-DiHDPE were increased by 43–71 % after CLP surgery (p < 0.05, Table S4). Interestingly, for 15,16-DiHODE a 46 % lower concentration after CLP was observed (p < 0.05). The other dihydroxy-FA levels in kidney, such as 9,10-DiHODE, 12,13-DiHOME, 8,9-; 11,12- and 14,15-DiHETrE and 7,8-DiHDPE were unaffected. In the heart tissue inconsistent effects of CLP induced sepsis on dihydroxy-FA levels were observed: 15,16-DiHODE and 9,10-dihydroxystearic acid concentration were decreased by 69 and 32 %, respectively (p < 0.01). Levels of 17,18-DiHETE, 10,11- and 19,20-DiHDPE and 14,15-DiHETrE were elevated by 27–75 % (p < 0.05, Table S5). In the liver 12,13- and 15,16-DiHODE, 17,18-DiHETE and 12,13-DiHOME were significantly decreased by 30–62 % after CLP surgery (p < 0.05). Except for the 15-LOX metabolite 8,15-DiHETE, which was decreased by 56 % (p < 0.05) in the treatment group, no changes were observed for the CYP/sEH formed dihydroxy-FAs detected in the lung tissue (Table S6).
Discussion
Sepsis is a severe medical condition characterized by the release of pro-inflammatory mediators resulting in centralization with hypotension, malperfusion of the peripheral organs leading to multi organ failure. Aside from interleukins, induction of COX-2 leads to an increase of PGE2 in sepsis [5]. This study aims to comprehensively investigate if and how other lipid mediators derived from AA or other n-3 FAs and PGs are involved in the inflammatory processes of sepsis by comparing two commonly used sepsis models, the LPS and CLP model. Due to the high lethality in sepsis models only a single early time point at 24 h after sepsis induction was studied. In the majority of other sepsis studies this time point has also been chosen.
Both models successfully induced sepsis as characterized by clinical chemistry. The increased plasma creatinine and urea levels in the LPS group indicate an acute kidney injury (AKI), a common condition in human sepsis [31]. Liver injury was moderate in the LPS group, with only a slight increase in AST levels. CLP surgery resulted in a dramatic increase of AST and ALT, indicating a more pronounced liver damage. No change in plasma creatinine and only ~50 % increase in plasma urea compared to the LPS model was observed in the CLP induced sepsis model. Elevation of urea without plasma-creatinine elevation points towards increased catabolism [32] but subclinical AKI is already present as pro-inflammatory cytokines increased markedly in the kidney. Comparing both models the LPS induced sepsis resulted in clinical overt AKI with plasma-creatinine elevation, whereas after CLP surgery the liver failure was more pronounced. The inconsistent induction of AKI by the CLP surgery has been described previously [33] and depends on the severity of the model which can be modulated by the length of the ligated cecum. We have chosen a moderate CLP model in which lethality starts beyond the 24 h time point of this study [28, 34]. Due to the different induction procedures different time courses of organ failure are very likely. By CLP surgery two small puncture wholes into the ligated cecum are performed resulting in feceal contamination of the peritoneum with subsequent polymicrobial sepsis and bacteremia and constant LPS release. In comparison, single time LPS administration induces several clinical features of sepsis including elevation of pro-inflammatory cytokines such as IL-6 and MCP-1 but without bacteremia [35]. Renal failure in LPS injection has been explained by heavy disturbance of the renal blood flow due to NO mediated mechanisms and caspase activation [36]. The tissue cytokine elevation of IL-6 and MCP-1 in kidney and liver clearly shows the sepsis induced organ involvement in both models.
The induction of sepsis is characterized by increased COX-2 expression, e.g. in activated macrophages, and as a consequence by increased levels of PGs [5]. Utilizing targeted metabolomics we simultaneously monitored the increase of a large number of PGs, such as PGE2, the prostacyclin metabolite 6-keto-PGF1α and the PGF2α metabolite 13,14-dihydro-15-keto-PGF2α. Moreover, metabolites derived from other FAs, e.g. 13,14-dihydro-15-keto-PGE1 were observed for the first time in LPS-induced sepsis. The massive increase of PGE2 and 6-keto-PGF1α after LPS treatment is consistent with the results of previous studies [8]. CLP surgery led to a comparable moderate increase in PG levels, indicating a significant difference between both models. Similar observations have been made regarding cytokine production, which were about 100-fold higher in the LPS model in comparison to the CLP model [4, 37]. Moreover, it has been reported that LPS injection is followed by fast and transient increase of systemic cytokine levels, whereas after CLP surgery the increase is more continuous and sustained [4, 26]. Since both models were only analyzed after 24 h it can only be assumed that the increase of PGs may follow similar kinetics. In other studies a rapid increased of PGE2 levels in serum or peritoneal lavage fluid was observed after 5, 12 or 18 h after surgery [9, 38, 39]. Overall, it is difficult to compare different CLP studies because different numbers of cecal punctures and needle puncture size influence the release of intestinal bacteria which affects the onset and progression of sepsis [4].
Regarding lipid mediators formed in the LOX and CYP pathway of the AA cascade a large number of oxylipins is elevated in the LPS and CLP induced sepsis. Only a slight to moderate increase of the non-enzymatically formed autoxidation markers 9- and 11-HETE was observed indicating a specific effect rather than an unspecific formation in response to elevated activity of PLA2 or other lipases, as observed in inflammatory in vivo models [30, 40]. Remarkably, there is a trend towards increased 12-LOX products (12-HETE, 12-HEPE) in plasma, liver and kidney, indicating an elevated 12-LOX and 12-lipoxygenating ALOX-15 activity during sepsis. The massive plasma increase of the neutrophil chemoattractant 12-HETE [41, 42] suggests an involvement of this lipid mediator in the development of sepsis. A pharmacological decrease of this pro-inflammatory mediator may help to reduce neutrophil recruitment, as already shown in mouse models of acute lung injury [43], and therefore might allow attenuate multi organ failure during sepsis. However, it has to be considered that a reduced neutrophil recruitment is controversial in early stage of sepsis [44]. Interestingly, 20-HETE level is massively increased after LPS treatment (by 100 % in plasma, 230 % in liver, 68 % in kidney and 330 % in lung) and CLP surgery by 83 % in plasma, 170 % in liver, Table S2). This is somewhat surprising since 20-HETE is a potent vasoconstrictor [45, 46], and one would expect based on the reduced blood pressure during acute septic shock a low level of this oxylipin as reported earlier [47].
It is remarkable that sepsis leads to a general elevation of dihydroxy-FA plasma levels, which is consistent with earlier studies [8, 23, 48]. The CLP model elicits the same effect, albeit less pronounced. Regarding epoxy-FAs as precursors of dihydroxy-FAs, only few were elevated during sepsis. This suggests that sepsis increases CYP mediated formation of highly biological active epoxy-FAs [49] which are rapidly hydrolyzed to less active dihydroxy-FA by sEH. This assumption is substantiated by the observation that the most abundant epoxides, e.g. 9(10)- and 12(13)-EpOME, are increased in sepsis. Though EpOMEs are moderately good substrates for the sEH (K M ~ 3 µM, k cat 0.7 s [17]) the formation of high concentrations might exceed the capacity of sEH to abolish the increase in these epoxy-FAs.
Although clinical chemistry revealed a severe kidney injury in the LPS group, no effect on AA derived PGs was observed. Only levels of PGD1 and PGF1α derived from DGLA and dihomo PGF2α derived from adrenic acid—known to be produced in renal medulla—were elevated [50]. Interestingly, most of the changes in oxylipin levels in the kidney were detected in the CYP branch, especially for the epoxy-FAs resulting in increased epoxy to dihydroxy ratios of LA and ALA. The vasodilatory properties of the epoxy-FA [49] may contribute to the development of hypotension during sepsis. However, it should be noted that several others studies suggest that increasing/stabilizing epoxy-FA by administration of sEHi improves clinical symptoms of sepsis in the LPS-induced sepsis [8]. A limitation of the study is that no time course experiments have been performed and only the 24 h time point has been studied. In future experiments longitudinal investigations would be interesting as well.
Consistent with clinical chemistry which revealed no AKI in the CLP group, no effect on kidney PGs, hydroxy- and epoxy-FA levels was found, showing again differences in LPS- and CLP-induced sepsis. The liver injury in the CLP group was not accompanied by increased PG levels and only a small number of hydroxy-FAs were elevated. Because LPS treatment resulted in changes of few oxylipins as well, it is concluded that oxylipins are not a suitable marker for liver damage in LPS and CLP sepsis models.
Overall, the present study shows that both in vivo models of sepsis are characterized by massive changes in plasma oxylipin derived from all enzymatic branches of the AA cascade. Comparing both models 24 h after induction of sepsis the LPS model caused a more pronounced increase in oxylipins. Thus, this model seems to be better suited to investigate effects on the AA cascade than CLP.
References
Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med. 2013;369:840–51.
Annane D, Aegerter P, Jars-Guincestre MC, Guidet B. Current epidemiology of septic shock. Am J Respir Crit Care Med. 2003;168:165–72.
Martin GS. Sepsis, severe sepsis and septic shock: changes in incidence, pathogens and outcomes. Exp Rev Anti Infect Ther. 2012;10:701–6.
Buras JA, Holzmann B, Sitkovsky M. Animal models of sepsis: setting the stage. Nat Rev Drug Discov. 2005;4:854–65.
Cohen J. The immunopathogenesis of sepsis. Nature. 2002;420:885–91.
Ertel W, Morrison MH, Wang P, Ba ZF, Ayala A, Chaudry IH. The complex pattern of cytokines in sepsis. Association between prostaglandins, cachectin, and interleukins. Ann Surg. 1991;214:141–8.
Liu J-Y, Tsai H-J, Hwang SH, Jones PD, Morisseau C, Hammock BD. Pharmacokinetic optimization of four soluble epoxide hydrolase inhibitors for use in a murine model of inflammation. Br J Pharmacol. 2009;156:284–96.
Schmelzer KR, Kubala L, Newman JW, Kim I-H, Eiserich JP, Hammock BD. Soluble epoxide hydrolase is a therapeutic target for acute inflammation. Proc Natl Acad Sci. 2005;102:9772–7.
Uozumi N, Kita Y, Shimizu T. Modulation of lipid and protein mediators of inflammation by cytosolic phospholipase a2alpha during experimental sepsis. J Immunol. 2008;181:3558–66.
Halushka PV, Reines HD, Barrow SE, Blair IA, Dollery CT, Rambo W, et al. Elevated plasma 6-keto-prostaglandin F1[alpha] in patients in septic shock. Crit Care Med. 1985;13:451–3.
Reines HD, Cook JA, Halushka PV, Wise WC, Rambo W. Plasma thromboxane concentrations are raised in patients dying with septic shock. Lancet. 1982;320:174–5.
Smyth EM, Grosser T, Wang M, Yu Y, FitzGerald GA. Prostanoids in health and disease. J Lipid Res. 2009;50:S423–8.
Ricciotti E, Fitzgerald GA. Prostaglandins and Inflammation. Arterioscler Thromb Vasc Biol. 2011;31:986–1000.
Basu S. Novel cyclooxygenase-catalyzed bioactive prostaglandin F2α from physiology to new principles in inflammation. Med Res Rev. 2007;27:435–68.
Samuelsson B, Goldyne M, Granstrom E, Hamberg M, Hammarstrom S, Malmsten C. Prostaglandins and thromboxanes. Annu Rev Biochem. 1978;47:997–1029.
Imig JD. Epoxides and soluble epoxide hydrolase in cardiovascular physiology. Physiol Rev. 2012;92:101–30.
Morisseau C, Inceoglu B, Schmelzer K, Tsai HJ, Jinks SL, Hegedus CM, et al. Naturally occurring monoepoxides of eicosapentaenoic acid and docosahexaenoic acid are bioactive antihyperalgesic lipids. J Lipid Res. 2010;51:3481–90.
Kroetz DL, Xu F. Regulation and inhibition of arachidonic acid omega-hydroxylases and 20-HETE formation. Annu Rev Pharmacol Toxicol. 2005;45:413–38.
Haeggström JZ, Funk CD. Lipoxygenase and leukotriene pathways: biochemistry, biology, and roles in disease. Chem Rev. 2011;111:5866–98.
Serhan CN, Petasis NA. Resolvins and protectins in inflammation resolution. Chem Rev. 2011;111:5922–43.
Yin H, Brooks JD, Gao L, Porter NA, Morrow JD. Identification of novel autoxidation products of the omega-3 fatty acid eicosapentaenoic acid in vitro and in vivo. J Biol Chem. 2007;282:29890–901.
Milne GL, Musiek ES, Morrow JD. F2-isoprostanes as markers of oxidative stress in vivo: an overview. Biomarkers. 2005;10:10–23.
Liu J-Y, Lin Y-P, Qiu H, Morisseau C, Rose TE, Hwang SH, et al. Substituted phenyl groups improve the pharmacokinetic profile and anti-inflammatory effect of urea-based soluble epoxide hydrolase inhibitors in murine models. Eur J Pharm Sci. 2013;48:619–27.
Meirer K, Steinhilber D, Proschak E. Inhibitors of the arachidonic acid cascade: interfering with multiple pathways. Basic Clin Pharmacol Toxicol. 2013;114:83–91.
Peri KG, Varma DR, Chemtob S. Stimulation of prostaglandin G/H synthase-2 expression by arachidonic acid monooxygenase product, 14,15-epoxyeicosatrienoic acid. FEBS Lett. 1997;416:269–72.
Dejager L, Pinheiro I, Dejonckheere E, Libert C. Cecal ligation and puncture: the gold standard model for polymicrobial sepsis? Trends Microbiol. 2011;19:198–208.
Schabbauer G. Polymicrobial sepsis models: CLP versus CASP. Drug Discov Today. 2012;9:e17–21.
Kümpers P, Gueler F, David S, Slyke PV, Dumont DJ, Park JK, et al. The synthetic tie2 agonist peptide vasculotide protects against vascular leakage and reduces mortality in murine abdominal sepsis. Crit Care. 2011;15:R261.
Ostermann AI, Willenberg I, Schebb NH. Comparison of sample preparation methods for the quantitative analysis of eicosanoids and other oxylipins in plasma by means of LC–MS/MS. Anal Bioanal Chem. 2015;407:1403–14.
Willenberg I, Ostermann AI, Giovannini S, Kershaw O, von Keutz A, Steinberg P, et al. Effect of acute and chronic DSS induced colitis on plasma eicosanoid and oxylipin levels in the rat. Prostaglandins Other Lipid Mediat 2015; 120:155–160.
Zarjou A, Agarwal A. Sepsis and acute kidney injury. J Am Soc Nephrol. 2011;22:999–1006.
Hosten AO. BUN and creatinine. In: Walker HK, Hall WD, Hurst JW, editors. Clinical methods: the history, physical, and laboratory examinations. Boston: Butterworths; 1990.
Doi K, Leelahavanichkul A, Yuen PS, Star RA. Animal models of sepsis and sepsis-induced kidney injury. J Clin Investig. 2009;119:2868–78.
Rittirsch D, Huber-Lang MS, Flierl MA, Ward PA. Immunodesign of experimental sepsis by cecal ligation and puncture. Nat Protoc. 2009;4:31–6.
Remick DG, Newcomb DE, Bolgos GL, Call DR. Comparison of the mortality and inflammatory response of two models of sepsis: lipopolysaccharide vs. cecal ligation and puncture. Shock. 2000;13:110–6.
Tiwari MM, Brock RW, Megyesi JK, Kaushal GP, Mayeux PR. Disruption of renal peritubular blood flow in lipopolysaccharide-induced renal failure: role of nitric oxide and caspases. Am J Physiol Renal Physiol. 2005;289:F1324–32.
Deitch EA. Animal models of sepsis and shock: a review and lessons learned. Shock. 1998;9:1–11.
Bitto A, Minutoli L, David A, Irrera N, Rinaldi M, Venuti F, et al. Flavocoxid, a dual inhibitor of COX-2 and 5-LOX of natural origin, attenuates the inflammatory response and protects mice from sepsis. Crit Care. 2012;16:R32.
Spite M, Norling LV, Summers L, Yang R, Cooper D, Petasis NA, et al. Resolvin D2 is a potent regulator of leukocytes and controls microbial sepsis. Nature. 2009;461:1287–91.
Hamaguchi K, Kuwata H, Yoshihara K, Masuda S, Shimbara S, Oh-ishi S, et al. Induction of distinct sets of secretory phospholipase A2 in rodents during inflammation. Biochim Biophys Acta. 2003;1635:37–47.
Cunningham FM, Wollard PM. 12(R)-hydroxy-5,8,10,14-eicosatetraenoic acid is a chemoattractant for human polymorphonuclear leucocytes in vitro. Prostaglandins. 1987;34:71–8.
Wiggings RE, Jafri MS, Proia AD. 12(S)-hydroxy-5,8,10,14-eicosatetraenoic acid is a more potent neutrophil chemoattractant than the 12(R) epimer in the rat cornea. Prostaglandins. 1990;40:131–41.
Rossaint J, Nadler J, Ley K, Zarbock A. Eliminating or blocking 12/15-lipoxygenase reduces neutrophil recruitment in mouse models of acute lung injury. Crit Care. 2012;16:1–15.
Craciun FL, Schuller ER, Remick DG. Early enhanced local neutrophil recruitment in peritonitis-induced sepsis improves bacterial clearance and survival. J Immunol. 2010;185:6930–8.
Roman RJ. P-450 metabolites of arachidonic acid in the control of cardiovascular function. Physiol Rev. 2002;82:131–85.
Zou AP, Fleming JT, Falck JR, Jacobs ER, Gebremedhin D, Harder DR, et al. 20-HETE is an endogenous inhibitor of the large-conductance Ca(2 +)-activated K + channel in renal arterioles. Am J Physiol. 1996;270:R228–37.
Theken KN, Deng Y, Kannon MA, Miller TM, Poloyac SM, Lee CR. Activation of the acute inflammatory response alters cytochrome P450 expression and eicosanoid metabolism. Drug Metab Dispos Biol Fate Chem. 2011;39:22–9.
Kubala L, Schmelzer KR, Klinke A, Kolarova H, Baldus S, Hammock BD, et al. Modulation of arachidonic and linoleic acid metabolites in myeloperoxidase-deficient mice during acute inflammation. Free Radic Biol Med. 2010;48:1311–20.
Morisseau C, Hammock BD. Impact of soluble epoxide hydrolase and epoxyeicosanoids on human health. Annu Rev Pharmacol Toxicol. 2013;53:37–58.
Sprecher H, VanRollins M, Sun F, Wyche A, Needleman P. Dihomo-prostaglandins and -thromboxane. A prostaglandin family from adrenic acid that may be preferentially synthesized in the kidney. J Biol Chem. 1982;257:3912–8.
Acknowledgments
Our work is supported by Fonds der Chemischen Industrie, the European Union (Marie Curie Career Integration Grant CIG 293536) and the German Research Foundation (Grant SCHE 1801). We thank Martina Ackermann and Herle Chlebusch for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: John Di Battista.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Willenberg, I., Rund, K., Rong, S. et al. Characterization of changes in plasma and tissue oxylipin levels in LPS and CLP induced murine sepsis. Inflamm. Res. 65, 133–142 (2016). https://doi.org/10.1007/s00011-015-0897-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-015-0897-7