Abstract.
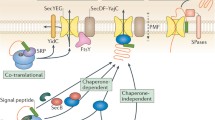
SecB is only one of a plethora of cytosolic chaperones in E. coli whose common property is that they bind nonnative proteins. It plays a crucial role during protein export via the general secretory pathway by modulating the partitioning of precursors between folding or aggregation and delivery to the membrane-bound translocation apparatus. In this latter role SecB demonstrates specific binding to a unique partner, SecA. SecB has the potential to participate in functions outside of export acting as a general nonspecific chaperone to provide buffering capacity of the nonnative state of proteins in the cytosolic pool. We discuss the interactions of SecB with its many binding partners in light of its recently determined structure, emphasizing both kinetic and thermodynamic parameters.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
RID="*"
ID="*"Corresponding author.
Rights and permissions
About this article
Cite this article
Randall, L., Hardy, S. SecB, one small chaperone in the complex milieu of the cell. CMLS, Cell. Mol. Life Sci. 59, 1617–1623 (2002). https://doi.org/10.1007/PL00012488
Issue Date:
DOI: https://doi.org/10.1007/PL00012488