Abstract
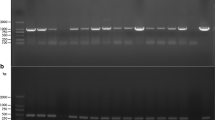
AsAgrobacterium tumefaciens, which has long been used to transform plants, is known to transfer T-DNA to budding yeast,Saccharomyces cerevisiae, a variety of fungi were subjected to theA. tumefaciens-mediated transformation to improve their transformation frequency and feasibility. TheA. tumefaciens-mediated transformation of chestnut blight fungus,Cryphonectria parasitica, is performed in this study as the first example of transformation of a hardwood fungal pathogen. The transfer of the binary vector pBIN9-Hg, containing the bacterial hygromycin B phosphotransferase gene under the control of theAspergillus nidulans trpC promoter and terminator, as a selectable marker, led to the selection of more than 1,000 stable, hygromycin B-resistant transformants per 1×106 conidia ofC. parasitica. The putative transformants appeared to be mitotically stable. The transformation efficiency appears to depend on the bacterial strain, age of the bacteria cell culture and ratio of fungal spores to bacterial cells. PCR and Southern blot analysis indicated that the marker gene was inserted at different chromosomal sites. Moreover, three transformants out of ten showed more than two hybridizing bands, suggesting more than two copies of the inserted marker gene are not uncommon.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bennett, J. W. (1998) Mycotechnology: The role of fungi in biotechnology.J. Biotechnol. 66: 101–107.
Ballance, D. J., F. P. Buxton, and G. Turner (1983) Transformation ofAspergillus nidulans by the orotidine-5′-phosphate decarboxylase gene ofNeurospora crassa, Bioche. Biophys. Res. Commun. 112: 284–289.
Yelton, M. M., J. E. Hamer, and W. E. Timberlake (1984) Transformation ofAspergillus nidulans by using a trpC plasmid.Proc. Natl. Acad. Sci. USA 81: 1470–1474.
Lemke, P. A. and M. Peng (1995) Genetics and biotechnology, pp. 109–139,In: U. Kuck (ed.),The Mycota. Vol. 2. Springer-Verlag, Berlin, Germany.
Chaure, P., S. J. Gurr, and P. Spanu (2000) Stable transformation ofErysiphe grminis an obligate biotrophic pathogen of barley.Nat. Biotechnol. 18: 205–207.
Abuodgeh, R. O., M. J. Orbach, M. A. Mandel, A. Das, and J. N. Galgiani (2000) Genetic transformation ofCoccidioides immitis facilitated byAgrobacterium tumefaciens.J. Infect. Dis. 81: 2106–2110.
Chen, X., M. Stone, C. Schlagnhaufer, and C. P. Romaine (2000) A fruiting body tissue method for efficientAgrobacterium-mediated transformation ofAgaricus bisporus.Appl. Environ. Microbiol. 66: 4510–4513.
Covert, S. F., P. Kapoor, M. Lee, A. Briley, and C. J. Nairn (2001)Agrobacterium tumefaciens-mediated transformation ofFusarium circinatum, Mycol. Res. 105: 259–264.
de Groot, M. J. A., P. Bundock, P. J. J. Hookaas, and A. G. M. Beijersbergen (1998)Agrobacterium tumefaciens-mediated transformation of filamentous fungi.Nat. Biotechnol. 16: 839–842.
Malonek, S. and F. Meinhardt (2001) Genetic transformation of the phYtopathogenic ascomYceteCalonectria morganii.Curr. Genet. 40: 152–155.
Van de Rhee, M. D., P. M. A. Graca, H. J. Huizing, and H. Mooibroek (1996) Highly efficient homologous integration via tandem exo-beta-1,3-glucanase genes in the common mushroom,Agaricus bisporus.Curr. Genet. 30: 166–173.
Anagnostakis, S. L. (1982) Biological control of chestnut blight.Science 215: 466–471.
Nuss, D. L. (1992) Biological control of chestnut blight: an example of virus-mediated attenuation of fungal pathogenesis.Microbiol. Rev. 56: 561–576.
Elliston, J. E. (1985) Characterization of dsRNA-free and dsRNA-containing strains ofEndothia parasitica in relation to hypovirulence.Phytopathology 75: 151–158.
Havir, E. A. and S. L. Anagnostakis (1985) Oxalate production by virulent but not by hypovirulent strains ofEndothia parasitica.Physiol. Plant Pathol. 23: 369–376.
Rigling, D., U. Heiniger, and H. R. Hohl (1989) Reduction of laccase activity in dsRNA-containing hypovirulent strains ofCryphonectria (Endothia) parasitica.Phytopathology 79: 219–223.
Kim, M. J., J. W. Choi, S. M. Park, B. J. Cha, M. S. Yang, and D. H. Kim (2002) Characterization of a fungal protein kinase fromCryphonectria parasitica and its transcriptional upregulation by hypovirus.Mol. Microbiol. 45: 933–941.
Rigling, D. and N. K. Van Alfen (1991) Regulation of laccase biosynthesis in the plant-pathogenic fungusCryphonectria parasitica by double-stranded RNA.J. Bacteriol. 173: 8000–8003.
Varley, D. A., G. K. Podila, and S. T. Hiremath (1992) Cutinase inCryphonectria parasitica, the chestnut blight fungus: suppression of cutinase gene expression in isogenic hypovirulent strains containing double-stranded RNAs.Mol. Cell. Biol. 12: 4539–4544.
Zhang, L., D. Villalon, Y. Sun, P. Kazmierczak, and N. K. Van Alfen (1994) Virus-associated down-regulation of the gene encoding cryparin, an abundant cell-surface protein from the chestnut blight fungus,Cryphonectria parasitica.Gene 139: 59–64.
Mullins, E. D., X. Chen, P. Romaine, R. Raina, D. M. Geiser, and S. Kang (2001)Agrobacterium tumefaciens-mediated transformation ofFusarium oxysporum: An efficient tool for insertional mutagenesis and gene transfer.Phytopathology 91: 173–180.
Sweigard, J. A., A. M. Carroll, L. Farrall, F. G. Chumley, and B. Valent (1998)Magnaporthe grisea pathogenicity genes obtained through insertional mutagenesis.Mol. Plant-Microbe Interact. 11: 404–412.
Hooykaas, P. J., C. Roobol, and R. A. Schilperoot (1979) Regulation of the transfer of Ti-plasmids ofAgrobacterium tumefaciens.J. Gen. Microbiol. 110: 99–109.
Bevan, M. (1984) BinaryAgrobacterium vectors for plant transformation.Nucleic Acids Res. 12: 8711–8721.
Kim, D. H., D. Rigling, L. Zhang, and N. K. Van Alfen (1995) A new extracellular laccase ofCryphonectria parasitica is revealed by deletion ofLac1.Mol. Plant-Microbe Interact. 8: 259–266.
Figurski, L. L. and D. R. Helinski (1979) Replication of origin-containing derivative of plasmid RK2 dependent on a plasmid function provided in trans.Proc. Natl. Acad. Sci. USA 76: 1648–1652.
Jung, W. H., C. H. Baek, J. K. Lee, and K. S. Kim (1999) Analysis of trans-acting elements for regulation of moc operons of p Ti15955 inAgrobacterium tumefaciens.J. Microbial. Biotechnol. 9: 637–645.
Bundock, P., A. den Dulk-Ras, A. Beijersbergen, and P. J. Hooykaas (1995) Trans-kingdom T-DNA transfer fromAgrobacterium tumefaciens toSaccharomyces cerevisiae.EMBO J. 14: 3206–3214.
Rho, H. S., S. Kang, and Y. H. Lee (2001)Agrobacterium tumefaciens-mediated transformation of the plant pathogenic fungus,Magnaporthe grisea.Mol. Cells 31: 407–411.
Kim, M. J., T. H. Kwon, Y. S. Jang, M. S. Yang, and D. H. Kim (2000) Expression of murine GM-CSF in recombinantAspergillus niger.J. Microbial. Biotechnol. 10: 287–292.
Lazo, G. R., P. A. Stein, and R. A. Ludwig (1991) A DNA transformation-competentArabidopsis genomic library inAgrobacterium.Bio/Technol. 9: 963–968.
Gallie, D. R., D. Zaitlin, K. L. Perry, and C. Kado (1984) Characterization of the replication and stability regions ofAgrobacterium tumefaciens plasmid pTAR.J. Bacteriol. 157: 739–745.
Krishnamohan, A., V. Balaji, and K. Veluthambi. (2001) Efficientvir gene induction inAgrobacterium tumefaciens requiresvirA, virG, andvir box from the same Ti plasmid.J. Bacteriol. 183: 4079–4089.
Koncz, C. and J. Schell (1986) The promoter of TL-DNA gene 5 controls the tissue-specific expression of chimeric genes carried by a novel type of Agrobacterium binary vector.Mol. Gen. Genet. 204: 383–396.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, SM., Kim, DH. Transformation of a filamentous fungusCryphonectria parasitica usingAgrobacterium tumefaciens . Biotechnol. Bioprocess Eng. 9, 217–222 (2004). https://doi.org/10.1007/BF02942296
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02942296