Abstract
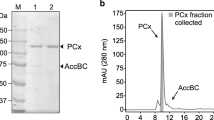
Carboxymethylcellulases (CMCases) fromAspergillus niger andCellulomonas biazotea were purified by a combination of ammonium sulfate precipitation, anion-exchange and gel-filtration chromatography with a 12- and 9-fold increase in the purification factor. The native and subunit molar mass of CMCase fromA. niger were 40 and 25–57 kDa, respectively, while those fromC. biazotea were 23 and 20–30 kDa, respectively. Low concentrations of Mn2+ activated the enzymes from both organisms (mixed activation) with apparent activation constants of 0.80 and 0.45 mmol/L of CMCases fromA. niger andC. biazotea, respectively, while at higher CMC concentrations Mn2+ inhibited the enzymes (mixed and partial uncompetitive inhibition). The reason for this complex behavior is that more than one Mn2+ bind to the same enzyme form with the apparent average inhibition constants of 2.7 and 1.3 mmol/L for CMCases fromA. niger andC. biazotea, respectively.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Akiba S., Kimura Y., Yamamoto K., Kumagai H.: Purification and characterization of a protease-resistant cellulase fromAspergillus niger.J. Ferment. Bioeng. 79, 125–130 (1995).
Bae H.D., Mcallister T.A., Yanke J., Cheng K.J., Muir A.D.: Effects of condensed tannins on endoglucanase activity and filter paper digestion byFibrobacter succinogenes S85.Appl. Environ. Microbiol. 59, 2132–2138 (1993).
Bradford M.M.: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding.Anal. Biochem. 72, 248–254 (1976).
Calza R.E., Irwin D.C., Wilson D.B.: Purification and characterization of two β-1,4-endoglucanases fromThermomonospora fusca.Biochemistry 24, 7797–7804 (1985).
Coughlan M.P.: Staining techniques for the detection of the individual components of cellulolytic enzyme system.Methods Enzymol. 160, 135–144 (1988).
Dixon M., Webb E.C.: Enzyme inhibition and activation, pp. 332–467 inEnzymes. Academic Press, New York 1979.
Fierobe H.P., Bagnaratardif C., Gaudin C., Guerlesquin F., Sauve P. Belaich A., Belaich J.P.: Purification and characterization of endoglucanase-C fromClostridium cellulolyticum—catalytic comparison with endoglucanase-A.Eur. J. Biochem. 217, 557–565 (1993).
Gokhale D.V., Puntambekar U.S., Deobagkar D.N., Peberdy J.F.: Production of cellulolytic enzyme by mutants ofAspergillus niger NCIM1207.Enzyme Microb. Technol. 10, 442–445 (1988).
Kim C.: Characterization and substrate specificity of an endo-β-1,4-d-glucanase 1 (avicelase 1) from an extracellular multienzyme complex ofBacillus circulans.Appl. Environ. Microbiol. 61, 959–965 (1995).
Kundu R.K., Dube S., Dube D.K.: Extracellular cellulolytic enzyme system ofAspergillus japonicus. 3. Isolation, purification and characterization of multiple forms of endoglucanase.Enzyme Microb. Technol. 10, 100–109 (1988).
Lindner W.A.: Carboxymethyl cellulase fromSclerotium rolfsii.Methods Enzymol. 160, 376–382 (1988).
Lupo D., Stutzenberger F.: Changes in endoglucanase patterns during growth ofThermomonospora curvata on cellulose.Appl. Environ. Microbiol. 54, 588–589 (1988).
Merril C.R.: Gel staining techniques.Methods Enzymol. 182, 477–488 (1990).
Nakamura K., Kitamura K.: Purification and some properties of a cellulase active on crystalline cellulose fromCellulomonas uda.J. Ferment. Technol. 61, 379–382 (1983).
Ng T.K., Zeikus J.G.: Endoglucanase fromClostridium thermocellum.Methods Enzymol. 160, 351–355 (1988).
Rajoka M.I., Malik K.A.: Comparison of different strains ofCellulomonas for production of cellulolytic and xylanolytic enzymes from biomass produced on saline lands.Biotechnol. Lett. 8, 753–756 (1986).
Rangarajan M., Hartley B.S.: Mechanism ofd-fructose isomerization byArthrobacter d-xylose isomerase.Biochem. J. 283, 223–233 (1992).
Rossomando E.F.: Ion-exchange chromatography.Methods Enzymol. 182, 309–317 (1990).
Sami A.J., Akhtar M.W.: Purification and characterization of two low-molecular weight endoglucanases ofCellulomonas flavigena.Enzyme Microb. Technol. 15, 586–592 (1993).
Sami A.J., Akhtar M.W., Malik N.N., Naz B.a.: Production of free and substrate-bound cellulases ofCellulomonas flavigena.Enzyme Microb. Technol. 10, 626–631 (1988).
See Y.P., Jackowski G.: Estimating molecular weights of polypeptides by SDS-gel electrophoresis, pp. 1–21 inProtein Structure: A Practical Approach (T.E. Creighton, Ed.). IRL Press, Oxford (UK) 1989.
Sexana S., Bahadur J., Verma A.: Effect of cobalt and nickle on growth and carboxymethylcellulase activity ofCellulomonas spp.Biometals 5, 209–212 (1992).
Siddiqui K.S., Loviny-Anderton T., Rangarajan M., Hartely B.S.:Arthrobacter d-xylose isomerase: chemical modification of carboxyl groups and protein engineering of pH optimum.Biochem J. 296, 685–691 (1993).
Siddiqui K.S., Rashid H., Shemsi A.M., Rajoka M.I.: A simple and nondestructive method for the separation of polysaccharides from β-glucosidase produced extracellularly byAspergillus niger.Enzyme Microb. Technol. 16, 912–917 (1994).
Stellwagen E.: Gel filtration.Methods Enzymol. 182, 317–328 (1990).
Wood T.M., Bhat K.M.: Methods for measuring cellulase activities.Methods Enzymol. 160, 87–112 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Siddiqui, K.S., Azhar, M.J., Rashid, M.H. et al. Purification and the effect of manganese ions on the activity of carboxymethylcellulases fromAspergillus niger andCellulomonas biazotea . Folia Microbiol 42, 303–311 (1997). https://doi.org/10.1007/BF02816940
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02816940