Abstract
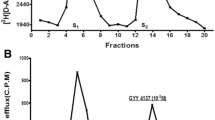
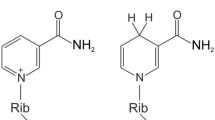
N-acetyl-l-histidine (NAH) is a major constituent of poikilotherm brain, eye, heart, and muscle, but for which there is no known function. NAH is characterized by high tissue concentrations, a high tissue/extracellular fluid (ECF) gradient, and by a continuous selective and regulated efflux into ECF. In the eye, there is a complete compartmentalization of the synthetic and hydrolytic enzymes, with synthesis of NAH from AcCoA andl-histidine (His) occurring in the lens, and its hydrolysis to acetate and His restricted to surrounding ocular fluids. Using14C-isotopes, the cycling of NAH between lens and ocular fluids in a simple support medium consisting of NaCl (0.9%), Ca2+ (4 mEq/L) andd-glucose (5 mM) at pH 7.4 has previously been observed. In the present study, using the isolated lens of the goldfish eye, each of the components of that support medium has been individually varied in order to determine its effect on NAH release down its intercompartmental gradient. As a result of these and related studies, it is suggested that NAH may function as a metabolically recyclable gradient-driven molecular water pump. It is proposed that water influx or generation of metabolic water serves as the trigger mechanism to open a Ca-dependent gate for the release of NAH down its gradient, along with its associated water. Preliminary analyses suggest that in addition to its potential for multiple daily cycles, a strongly ionized hydrophilic molecule, such as NAH, may include a large power function as a result of its attraction to water, and it has been calculated that an aqua complex of each NAH molecule may have 33 dipole-dipole-associated water molecules as it passes into ECF. It is this unique combination of a capacity for multiple cycles per day, coupled with a large power function, that may allow for such an intracellular osmolyte to be present in relatively low concentration in comparison to total cellular osmolality, and yet to perform a large and important task with little expenditure of energy. With each NAH molecule recycled up to 10 times/d, and a power factor of 33, there could be 330 mmol of water transported/mmol of NAH each day. With typical NAH concentrations in brains of poikilothermic vertebrates of 5–10 mmol/kg, there is the potential for up to 3.3 mol (60 mL) of water to be removed each day/kg of brain, a value that represents about 8% of total brain water content. Dewatering of the released osmolyte would occur in two additional steps, consisting of its hydrolysis and the subsequent active uptake of its metabolites. It is also suggested that NAH is the archetype of several metabolically and structurally related cellular osmolytes found in both poikilotherms and homeotherms, for which there is similarly no known function, and these may form a family of cycling hydrophilic osmolytes that serve as molecular water pumps in a variety of tissues. These include the basic His containing derivatives: NAH, carnosine, anserine, ophidine, and homocarnosine, and the acidic aspartate derivatives:N-acetyl-l-aspartate (NAA) andN-acetyl-l-aspartylglutamate (NAAG). In each of these cases, the high intracellular/extracellular osmolyte gradient appears to be maintained by combining a hydrophilic protein amino acid with a nonprotein moiety to block its use in other intracellular metabolic pathways, and by blocking catabolism of the derivative by maintaining its hydrolytic enzyme in an extracytosolic membrane or extracellular compartment. Unlike other known water-regulating mechanisms, the proposed cellular system is unique in that as a water pump, it can function as a water regulator independently of extracellular solute composition or osmolality. Finally, based on the hypothesis developed, the NAH system would represent the first cellular water pump to be identified.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baslow M. H. (1967)N-acetyl-l-histidine metabolism in the fish eye: Evidence for ocular fluid-lensl-histidine recycling.Exp. Eye Res. 6, 336–342.
Baslow M. H. (1997) A review of phylogenetic and metabolic relationships between the acylamino acids,N-acetyl-l-aspartic acid andN-acetyl-l-histidine in the vertebrate nervous system.J. Neurochem. 68(4), 1335–1344.
Baslow M. H. and Resnik T. R. (1997) Canavan disease: Analysis of the nature of the metabolic lesions responsible for development of the observed clinical symptoms.J. Mol. Neurosci. 9(2), 109–126.
Baslow M. H. and Yamada S. (1997a) Identification ofN-Acetylaspartate in the lens of the vertebrate eye: a new model for the investigation of the function ofN-acetylated amino acids in vertebrates.Exp. Eye Res. 64(2), 283–286.
Baslow M. H. and Yamada S. (1997b) Distribution ofN-acetyl-l-histidine andN-acetyl-l-aspartate in the vertebrate eye.J. Neurochem. 69(Suppl.), S175D.
Buniatian H. C. H., Hovhannissian V. S., and Aprikian G. V. (1965) The participation ofN-acetyl-l-aspartic acid in brain metabolism.J. Neurochem. 12, 695–703.
Dupont Y. (1983) Is Ca2+-ATPase a water pump?FEBS Lett. 161(1), 14–20.
Fischbarg J., Kuang K., Vera J. C., Arant S., Silverstein S. C., Loike J., et al. (1990) Glucose transporters serve as water channels.Proc. Natl. Acad. Sci. USA 87, 3244–3247.
Hess W. R. (1997) Localization of an open reading frame with homology to human aspartoacylase upstream from psbA in the prokaryoteProchlorococcus marinus CCMP 1375.DNA Sequence 7(5), 301–306.
Heuser J., Zhu Q., and Clarke M. (1993) Proton pumps populate the contractile vacuoles ofDictyostelium amoebae.J. Cell. Biol. 121(6), 1311–1327.
Hummer G., Garcia A. E., and Soumpasis D. M. (1996) A statistical mechanical description of biomolecular hydration.Faraday Discuss. 103, 175–189.
Jackson M. C., Scollard D. M., Mack R. J., and Lenney J. F. (1994) Localization of a novel pathway for the liberation of GABA in the human CNS.Brain Res. Bull. 33, 379–385.
Knott R. M. and Forrester J. V. (1995) Role of glucose regulatory mechanisms in diabetic retinopathy.Br. J. Ophthalmol. 79, 1046–1049.
Koch A. L. (1994) Development and diversification of the last universal ancestor.J. Theoret. Biol. 168(3), 269–280.
Kumagai A. K., Glasgow B. J., and Pardrigdge W. M. (1994) GLUT1 glucose transporter expression in the diabetic and nondiabetic human eye.Invest. Ophthalmol. Vis. Sci. 35, 2887–2894.
Lang F., Busch G. L., Ritter M., Volkl H., Waldegger S., Gulbins E., et al. (1998) Functional significance of cell volume regulatory mechanisms.Physiol. Rev. 78, 247–306.
Lea E. J. A. (1996) Main intrinsic polypeptide and water movement—recent developments and future prospects.Ophthalmic Res. 28(Suppl. 1), 69–72.
Lenney J. F., Peppers S. C., Kucera C. M., and Sjaastad O. (1983) Homocarnosinosis: Lack of serum carnosinase is the defect probably responsible for elevated brain and CSF homocarnosine.Clin. Chim. Acta 132, 157–165.
Meinild A.-K., Klaerke D. A., Loo D. D. F., Wright E. M., and Zeuthen T. (1998) The human Na+-glucose cotransporter is a molecular water pump.J. Physiol. 508(1), 15–21.
Pasantes-Morales H., Chácon E., Murray R. A., and Morán J. (1994) Properties of osmolyte fluxes activated during regulatory volume decrease in cultured cerebellar granule neurons.J. Neurosci. Res. 37, 720–727.
Perry T. L., Kish S. J., Sjaastad O., Gjessing L. R., Nesbakken R., Schrader H., et al. (1979) Homocarnosinosis: Increased content of homocarnosine and deficiency of homocarnosinase in brain.J. Neurochem. 32, 1637–1640.
Rae J. L. and Levis R. A. (1984) Patch clamp recordings from the epitheliium of the lens obtained using glasses selected for low noise and improved sealing properties.Biophys. J. 45, 144–146.
Rae J. L., Bartling C., Rae J. and Mathias R. T. (1996) Dye transfer between cells of the lens.Memb. Biol. 150, 89–103.
Sancho M., Partenskii M. B., Dorman V., and Jordan P. C. (1995) Extended dipolar chain model for ion channels: electrostriction effects and the translocation energy barrier.Biophys. J. 68, 427–433.
Strange K. (1992) Regulation of solute and water balance and cell volume in the central nervous system.J. Am. Soc. Nephrol. 3, 12–27.
Taylor D. L., Davies S. E. C., Obrenovitch T. P., Doheny M. H., Patsalos P. N., Clark J. B., et al. (1995) Investigation into the role ofN-acetylaspartate in cerebral osmoregulation.J. Neurochem. 65, 275–281.
Tsai G. and Coyle J. T. (1995)N-acetylaspartate in neuropsychiatric disorders.Prog. Neurobiol. 46, 531–540.
Unwin N. (1989) The structure of ion channels in membranes of excitable cells.Neuron 3, 665–676.
Urenjak J., Williams S. R., Gadian D. G., and Noble M. (1992) Specific expression ofN-acetylaspartate in neurons, oligodendrocyte-type-2 astrocyte progenitors, and immature oligodendrocytes in vitro.J. Neurochem. 59, 55–61.
Watanabe T., Mio Y., Hoshino F. B., Nagamatsu S., Hirosawa K., and Nakahara K. (1994) GLUT2 expression in the rat retina: localization at the apical ends of Müller cells.Br. Res. 655, 128–134.
Willi S. M., Zhang Y., Hill J. B., Phelan M. C., Michaelis R. C., and Holden K. R. (1997) A deletion in the long arm of chromosome 18 in a child with serum carnosinase deficiency.Pediatr. Res. 41, 210–213.
Zhang W. Z. and Augusteyn R. C. (1995) Glucose supply and enzyme activities in the lens.Exp. Eye Res. 61, 633–655.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Baslow, M.H. Function of theN-acetyl-l-histidine system in the vertebrate eye. J Mol Neurosci 10, 193–208 (1998). https://doi.org/10.1007/BF02761774
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02761774