Abstract
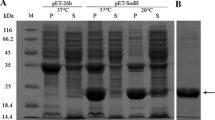
The gene for the Cu, Zn-superoxide dismutase (SOD) from the yeast Saccharomyces cerevisiae was cloned, characterized, and overexpressed in the methylotrophic Pichia pastoris. The sod gene sequence obtained is 465 bp and encodes 154 amino acid residues. The sod gene sequence was cloned into the pPIC9K vector, yielding pAB22. The linearized pAB22 DNA, digested with restriction enzyme SacI, was transformed into the genome of the GS115 strain of the yeast P. pastoris. The SOD was purified from the cultured yeast by ammonium sulfate precipitation and DEAE-cellulose column chromatography. This relatively simple purification method produced a single band on analysis by sodium dodecyl sulfate-polyacrylamide gel electrophoresis. The overexpressed SOD protein was shown to have immunologically biologic activity and to be enzymatically active. The yeast overexpressing Cu, Zn-SOD appeared to be more resistant to oxidative stress such as paraquat, menadione, and heat shock.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fridovich, I. (1979), Adv. Inorg. Biochem. 1, 69–70.
Gralla, E. B., Thiele, D. J., Silar, P., and Valentine, J. S. (1991), Proc. Natl. Acad. Sci. USA 88, 8558–8562.
Heinzen, R. A., Frazier, M. E., and Mallavia, L. P. (1992), Infect. Immun. 60(9), 3814–3823.
McCord, J. M. (1974), Science 185, 529–531.
Flohe, L., Giertz, H., and Beckman, R. (1985), Handbook of Inflammation, vol. 5, Elsevier Science, New York.
Oberley, L. W. and Buettner, G. R. (1979), Cancer Res. 39, 1141–1149.
Fridovich, I. (1983), Annu. Rev. Pharmacol. Toxicol. 23, 239–257.
Steinman, H. M. (1980), J. Biol. Chem. 255, 6758–6765.
Hartman, J. R., Geller, T., Yavin, Z., Bartfeld, D., Kanner, D., Aviv, H., and Gorecki, M. (1986), Proc. Natl. Acad. Sci. USA 83, 7142–7146.
Hallewell, R. A., Mills, R., Tekamp-Olson, P., et al. (1987), Biotechnology 5, 363–366.
Reddy, M. V. and Gangadharam, P. R. (1992), Infect Immun. 60, 2386–2390.
Sherman, F. (1986), in Methods in Yeast Genetics (Amberg, D. C. and Burke, D. J., eds.) Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, pp. 125–128.
McCord, J. M., Keele, B. B., and Fridovich, I. (1971), Proc. Natl. Acad. Sci. USA 68, 1024–1027.
Kumagai, Y., Shinyashiki, M., Sun, G. F., Shimojo, N., and Sagai, M. (1994), Experientia 50, 673–676.
Schagger, H. and Jagow, G. (1987), Anal. Biochem. 166, 368–379.
Harlow, E. and Lane, D. (1988) Antibodies: A Laboratory Manual, Spring Harbor Laboratory Press, Cold Spring Harbor, New York.
Deisseroth, A. and Dounce, A. L. (1978), Biochim. Biophys. Acta, 537, 86–99.
Jamieson, D. J., Stephen, D. W., and Terriere, E. C. (1996), FEMS Microbiol. Lett. 138, 83–88.
Liu, X. D. and Thiele, D. J. (1996), Genes Dev. 10, 592–603.
Moradas-Ferreinra, P., Costa, V., Piper, P., and Mager, W. (1996), Mol. Microbiol. 19, 651–658.
Kim, Y. H., Park, K. H., and Rho, H. M. (1996), J. Biol. Chem. 271, 24,539–24,543.
Nettleton, C. J., Bull, C., Baldwin, T. O., and Fee, J. A. (1984), Proc. Natl. Acad. Sci USA 81, 4970–4973.
Hassan, H. M. and Fridovich, I. (1977), J. Bacteriol. 129, 1574–1583.
Touati, D. and Carlioz, A. (1986), in Superoxide and Superoxide Dismutase in Chemistry, Biology and Medicine, Rotilio, G., ed. Elsevier, New York, pp. 287–292.
Gregory, E. M., Goscin, S. A., and Fridovich, I. (1974), J. Bacteriol. 117, 456–460.
Autor, A. P. (1982). J. Biol. Chem. 257, 2713–2718.
Orr, W. C. and Sohal, R. S. (1994), Science 263, 1128–1130.
Hwang, C. S., Rhie, G., Kim, S. T., Kim, Y. R., Huh, W. K., Baek, Y. U., and Kang, S. O. (1999), Biochem. Biophys. Acta 1427(2), 245–255.
Hernandez-Saavedra, N. Y. and Romero-Geraldo, R. (2001), Yeast, 14(6), 573–581.
Hernandez-Saavedra, N. Y., Egly, J. M., and Ochoa, J. L. (1998), Yeast 18(3), 1227–1238.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, JR., Yu, P. Expression of Cu, Zn-superoxide dismutase gene from Saccharomyces cerevisiae in Pichia pastoris and its resistance to oxidative stress. Appl Biochem Biotechnol 136, 127–139 (2007). https://doi.org/10.1007/BF02685943
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02685943