Abstract
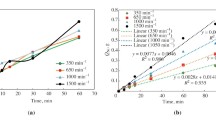
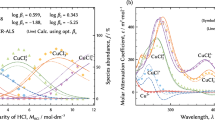
The rates of dissolution of synthetic cupric oxide in solutions containing perchloric, sulfuric, nitric or hydrochloric acid were studied using sintered disks. In each case, the dissolution rate increased with elapsed retention time until an essentially constant value was reached. This phenomenon can be attributed to an increase in the disk’s effective surface area. The dissolution rate is of the first order with respect to aH + for perchloric, nitric, and hydrochloric acids, while it is of a half order for sulfuric acid. High activation energies, ranging from 12.4 to 20.5 kcal/mol, and the independence of agitation speed on cupric oxide dissolution reaction rate suggest that chemical reactions are the major determinants of dissolution rates. The addition of electrolytes having anions common with the acids resulted in an acceleration of the dissolution rate due to increases in aH + values. However, the addition of electrolytes of noncommon anions revealed a quite different effect on dissolution rate. This suggests that the adsorption and/ or complexing of anions on the cupric oxide surface may have had a significant role in the determination of the dissolution rates. The type of acid used determined the identity of the adsorbed anion.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
I. H. Warren and E. Devuyst:Leaching of Oxide, D. I. J. Evans and R. S. Shoemaker, Eds., pp. 229–64, International Symposium on Hydrometallurgy, Chicago, AIME, NY, 1973.
D. J. G. Ives and J. J. George:Reference Electrodes, Academic Press, NY, 1961.
K. J. Vetter:Electrochemical Kinetics, p. 49, Academic Press, NY, 1967.
T. Yazaki: Master of Engineering, Thesis, Kyoto University, 1978.
V. S. Surana and I. H. Waren:frans. Inst. Min. Met., Sec. C, 1969, vol. 78, pp. 133–39.
N. P. Shevelev, I. G. Gorichev, and N. G. Kiyuchnikov:Zh. Fiz. Khim., 1974, vol. 48, pp. 2750–54.
J. M. West:Electrodeposition and Corrosion Processes, p. 114, D. Van Nostrand Co. Ltd., London, 1965.
Stability Constants, compiled by J. Bjerrumet al under the Auspice of the International Union of Pure and Applied Chemistry, The Chemical Society, London, 1958.
D. Dobos:Electrochemical Data, p. 186, Elsevier Scientific Publishing Co., Amsterdam, 1975.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Majima, H., Awakura, Y., Yazaki, T. et al. Acid dissolution of cupric oxide. Metall Trans B 11, 209–214 (1980). https://doi.org/10.1007/BF02668403
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02668403