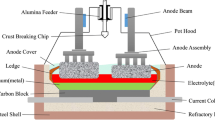
Abstract
The need for energy reduction in the electrolytic production of aluminum led to the concept of advanced Hall cells that can be operated at lower interelectrode gaps compared to existing cells. However, gas bubbles generated by the anodic reaction increase the resistivity of electrolyte and cancel out part of the reduction in interelectrode resistance expected from bringing the electrodes closer together. Therefore, the primary objective of this work was to determine a cell design in which flow can be managed to promote the removal of anode gas bubbles from the interelectrode gap. In particular, this article focuses on advanced Hall cells equipped with “flat” anodes, similar to those used in existing cells. The principal experimental tool has been a “water” model consisting of a large tank in which simulated anodes can be suspended in either the horizontal or near-horizontal configurations. Gas was generated by forcing compressed air through porous graphite, and the fine bubbles characteristic of inert anodes used in advanced Hall cells were produced by adding butanol to water. Velocities were measured using a laser-Doppler velocimeter (LDV). This study indicates that the existing cell configuration might not be the optimum configuration for advanced Hall cells. The results also show that operation of an advanced Hall cell with a fully submerged anode should give rise to higher electrolyte velocities and thus rapid removal of bubbles. The bubble effect should be further lowered in a near-horizontal configuration; however, the flow pattern could have an adverse effect on current efficiency and alumina distribution in the cell. It has also been shown that the bubble size, and, therefore, the physical properties of the electrolyte, can have a significant effect on the electrolyte flow pattern in the interelectrode gap.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
R.C. Dorward:J. Appl. Electrochem., 1982, vol. 13, p. 569.
R.C. Dorward and J.R. Payne:An Evaluation of a Titanium Diboride Composite Material as Cathode for Low Energy Alumina Reduction Cells, Topical Report DOE/CS/40215-2, Kaiser Aluminum and Chemical Corp., Oakland, CA, 1985.
R.C. Dorward and J.R. Payne:Development of Monolithic Titanium Diboride Cathodes for Retrofit Hall Cell Applications, Report DOE, DE-AC07-76CS40215, Kaiser Aluminum and Chemical Corp., Oakland, CA, 30 June 1976–30 June 1983.
K. Grjotheim, C. Krohn, R. Naeumann, and K. TØrklep:Metall. Trans., 1970, vol. 1, p. 3133–3141; 1971, vol. 2, pp. 199–204; 1973, vol. 4, pp. 1945–52.
E. Dernedde and E.L. Cambridge:AIME Light Met., 1975, vol. 1, p. 111.
A. Solheim, S.T. Johansen, S. Rolseth, and J. Thonstad:AIME Light Met., 1989, p. 245.
J.D. Weyand, S.P. Ray, F.W. Baker, D.H. De Young, and G.P. Tarcy:Inert Anodes for Aluminum Smelting, Final Report, DOE/ CS/40158-20, ALCOA, Feb. 1986.
J.R. Grace:Trans. Inst. Chem. Eng., 1973, vol. 51, p. 116.
S. Fortin, M. Gerhardt, and A.J. Gesing:AIME Light Met., 1984, p. 721.
J.W. Evans and R. Shekhar:Physical Modeling of Bubble Phenomena, Electrolyte Flow and Mass Transfer in Simulated Advanced Hall Cells, Report to DOE and Addendum, Subcontract No. C87-101226-002, University of California, Berkeley, CA, 1990 (DOE-ID-10281 and DOE-ID-10281-ADD).
R. Moreau and J.W. Evans:J. Electrochem. Soc., 1984, vol. 131, p. 2251.
S.D. Lympany and J.W. Evans:Metall. Trans. B, 1983, vol. 14B, pp. 63–70.
K. Grjotheim, R. Huglen, and H. Kvande: inUnderstanding the Hall-Héroult Process for Production of Aluminum, K. Grjotheim and H. Kvande, eds., Aluminium-Verlag, DŪseldorf, 1986, p. 19.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shekhar, R., Evans, J.W. Physical modeling studies of electrolyte flow due to gas evolution and some aspects of bubble behavior in advanced hall cells: Part I. Flow in cells with a flat anode. Metall Mater Trans B 25, 333–340 (1994). https://doi.org/10.1007/BF02663382
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02663382