Summary
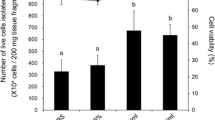
Rainbow trout (Salmo gairdneri) hepatocytes were isolated using a two-step perfusion through the portal vein. A typical perfusion yielded 2.92×106 liver cells with a mean viability of 96.3%. Hepatocytes comprised 93.4% of the total cell isolate. Survival of hepatocytes in suspension culture was dependent on fetal bovine serum concentration and temperature of incubation. Serum concentrations of 5, 10, and 20% produced the highest survival during primary culture. Hepatocyte survival was in inverse proportion to the incubation temperature. Trout hepatocyte DNA synthesis and mitosis decreased during the culture period. Cytochromep 450 activity decreased rapidly during the first 2 d of culture and then remained low but measurable during the remaining 8 d of culture. Culture temperature also influenced thep 450 activity with lower temperatures producing greater activity. Morphologic changes occurred in the cells during culture. Isolated hepatocytes self-aggregated, forming strands and clumps that increased in size with time in culture. Junctional complexes between cells were evident within the aggregates. Nuclear atypia, increases in size and number of autophagic vacuoles, and the appearance of bundles of intermediate filaments also were observed with increased time in culture.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bailey, G. S.; Taylor, M. J.; Selivonchichy, D. P. Alfatoxin B1 metabolism and DNA binding in isolated hepatocytes from rainbow trout (Salmo gairdneri). Carcinogenesis 3: 511–518; 1982.
Burton, K. A. A study of the conditions and mechanisms of the diphenylamine reaction for the colorimetric estimation of deoxyribonucleic acid. Biochem. J. 62: 315–323; 1956.
Fry, J.R.; Bridges, J.W. Use of primary hepatocyte cultures in biochemical toxicology. In: Hodges, E.; Bend, J. R.; Philpot, R.M., eds. Review in biochemical toxicology. New York: Elsevier/North Holland; 1979: 201–247.
haschemeyer, A.E.V.; Matthews, R.W. Temperature dependency of protein synthesis in isolated hepatocytes of antarctic fish. Physiol. Zool. 56: 78–87; 1983.
Hayashi, S.; Osshiro, Z. Gluconeogenesis and glycolysis in isolated perfused liver of the eel. Bull. Jpn. Soc. Sci. Fish. 41: 201–208; 1975.
Hazel, J.R.; Prosser, C.L. Incorporation of 1-14C-Acetate into fatty acids and sterols by isolated hepatocytes of thermally acclimated rainbow trout (Salmo gairdneri). J. Comp. Physiol. 134: 321–329; 1979.
Ishikawa, T.; Shimamiwe, T.; Takayama, S. Histologic and electron microscopy observation on diethylnitrosamine-induced hepatomas in small aquarium fish (Oryzias latipes). J. Natl. Cancer Inst. 55: 909–916; 1975.
Klaunig, J.E.; Lipsky, M.M.; Trump, B.F.; Hinton, D.E. Biochemical and ultrastructural changes in teleost liver following subacute exposure to PCB. J. Environ. Pathol. Toxicol. 2: 953–963; 1979.
Klaunig, J.E.; Goldblatt, P.J.; Hinton, D.E.; Lipsky, M.M.; Chacko, J.; Trump, B.F. Mouse liver cell culture. I. Hepatocyte isolation. In Vitro 17: 913–925; 1981.
Klaunig, J.E.; Goldblatt, P.J.; Hinton, D.E.; Lipsky, M.M.; Knipe, S.M.; Trump, B.F. Morphologic and functional studies of mouse hepatocytes in primary culture. Anat. Rec. 204: 231–243; 1982.
Klaunig, J.E. Primary culture of mouse hepatocytes: Effects of insulin and dexamethasone. In: Harris, R.A.; Cornell, N.W., eds. Isolation, characterization and use of hepatocytes. Elsevier Science Publishing Co., New York; 1983: 93–98.
Klaunig, J.E. Establishment of fish hepatocyte cultures for use in in vitro carcinogenicity studies. Natl. Cancer Inst. Mongr. 65: 163–173; 1984.
Maslansky, C.; Williams, G.M. Primary cultures and levels of cytochrome P450 in hepatocytes from mouse, rat, hamster, and rabbit liver. In Vitro 18: 683–693; 1981.
Porthe-Nibelle, J.; Lahlow, B. Mechanisms of glucocorticoid uptake by isolated hepatocytes of the trout. Comp. Biochem. Physiol. 69B: 425–433; 1981.
Saez, L.; Golcoechea, O.; Anthauer, R.; Krauskopf, M. Behavior of RNA and protein synthesis during the acclimatization of the carp. Studies with isolated hepatocytes. Comp. Biochem. Physiol. 72B: 31–38; 1982.
Sato, S; Matsushima, T.; Tanaka, N.; Sugimura, T.; Takashima, F. Hepatic tumors in the guppy induced by aflatoxin B1, dimethylnitrosamine, and 2-acetylaminofluorene. J. Natl. Cancer Inst. 50: 767–778; 1973.
Sinnhuber, R.O.; Hendricks, J.D.; Wales, J.H.; Putnam, G.B. Neoplasms in rainbow trout, a sensitive animal model for environmental carcinogenesis. Ann. NY Acad. Sci. 298: 389–408; 1977.
Sirica, A.E.; Pitot, H.C. Drug metabolism and effects of carcinogens in cultured hepatic cells. Pharmacol. Rev. 31: 205–227; 1980.
Tomatis, L.; Partensky, C.; Montesano, R. The predictive value of mouse liver tumor induction in carcinogenicity testing—A literature survey. Int. J. Cancer 12: 1–20; 1973.
Walton, M.J.; Cowey, C.B. Gluconeogenesis by isolated hepatocytes from rainbow troutSalmo gairdneri. Comp. Biochem. Physiol. 62B: 75–79; 1979.
Williams, G.M.; Bermudez, E.; Scaramuzzino, D. Rat hepatocyte primary cell cultures. Improved dissociation and attachment technologies and the enhancement of survival by culture medium. In Vitro 13: 809–817; 1978.
Wolf, K.; Mann, J.A. Poikilotherm vertebrate cell lines and viruses. A current listing for fishes. In Vitro 16: 168–179; 1980.
Author information
Authors and Affiliations
Additional information
This work was supported in part by an American Cancer Society Grant (Ohio Division, Inc.) and an NIH Biomedical Research Support Grant 5507RR05700010.
Rights and permissions
About this article
Cite this article
Klaunig, J.E., Ruch, R.J. & Goldblatt, P.J. Trout hepatocyte culture: Isolation and primary culture. In Vitro Cell Dev Biol 21, 221–228 (1985). https://doi.org/10.1007/BF02620933
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02620933