Abstract
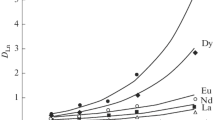
Distribution coefficients /Kd/ of lanthanide elements on layered hydrous titanium dioxide, H2Ti4O9.nH2O /where n=1.2–1.3/, have been determined as a function of the pH of the aqueous phase. The plots of 1g Kd vs. pH gave straight lines with slopes equal to +3 except for the data for heavier lanthanides, suggesting ideal ion-exchange equilibria between tervalent cations in the aqueous phase and hydrogen ions in the hydrous oxide. Mutual separations of La−Cs and La−Ba have been achieved on a column of this material on the basis of large differences in affinities between the metal ion pairs.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
R. Marchand, L. Brohan, M. Tournoux,Mater. Res. Bull., 15 /1980/ 1129.
H. Izawa, S. Kikkawa, M. Koizumi,J. Phys. Chem., 86 /1982/ 5023.
T. Sasaki, M. Watanabe, Y. Komatsu, Y. Fujiki,Inorg. Chem., 24 /1985/ 2265.
Y. Fujiki, Y. Komatsu, T. Sasaki,IONICS, 120 /1985/ 131.
Y. Komatsu, Y. Fujiki, T. Sasaki,Bunseki Kagaku, 31 /1983/ E225.
Y. Komatsu, Y. Fujiki, T. Sasaki,Bunseki Kagaku, 32 /1983/ E33.
T. Sasaki, Y. Komatsu, Y. Fujiki,Sep. Sci. Technol., 18 /1983/ 49.
T. Sasaki, Y. Komatsu, Y. Fujiki,Bull. Chem. Soc. Japan, 57 /1984/ 1331.
S. Ahrland, J. Albertsson,Acta Chem. Scand., 18 /1964/ 1861.
M. Tsuji, M. Abe,Solvent Extr. Ion Exch., 2 /1984/ 253.
D.D. Perrin, Dissociation Constants of Inorganic Acids and Bases in Aqueous Solution, IUPAC, Butterworth, London, 1969.
R.M. Smith, A.E. Martell, Critical Stability Constants, Plenum Press, New York, 4, 1976, 2.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sasaki, T., Komatsu, Y. & Fujiki, Y. Distribution coefficients of lanthanide elements and some separations on layered hydrous titanium dioxide. Journal of Radioanalytical and Nuclear Chemistry Letters 107, 111–119 (1986). https://doi.org/10.1007/BF02163446
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02163446