Summary
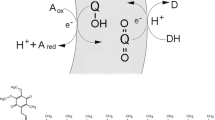
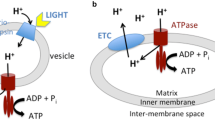
It is proposed that the energy-transducing system of the first cellular organism and its precursor was fueled by the oxidation of hydrogen sulfide and ferric sulfide to iron pyrites and two [H+] on the outside surface of a vesicle (the cell membrane), with the concomitant reduction of CO or CO2 on the interior. The resulting proton gradient across the cell membrane provides a proton-motive force, so that a variety of kinds of work can be done. It is envisioned as providing a selective advantage for cells capable of harvesting this potential. The proposed reactants for these reactions are consistent with the predicted composition of the Earth's early environment. Modern-day homologs of the ancestral components of the energy-transducing system are thought to be membrane-associated ferredoxins for the extracellular redox reaction, carbon monoxide dehydrogenase for the carbon fixation reaction, and ATPase for the harvesting of the proton gradient. With a source of consumable energy, the cell could drive chemical reactions and transport events in such a way as to be exploited by Darwinian evolution.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Berg JM, Holm RH (1982) Structure and reactions of ironsulfur protein clusters and their synthetic analogs. In: Spiro TG (ed) Iron-sulfur proteins. John Wiley, New York, pp 1–66
Cairns-Smith AG (1982) Genetic takeover and the mineral origin of life. Cambridge University Press, New York
Chang S, DesMarais D, Mack R, Miller SL, Strathearn GE (1983) Prebiotic organic syntheses and the origin of life. In: Schopf JW (ed) Earth's earliest biosphere. Princeton University Press, Princeton NJ, pp 53–92
DesMarais D, Chang S (1983) Processing procedure for abiotic samples and calculation of model atmospheric compositions. In: Schopf JW (ed) Earth's earliest biosphere. Princeton University Press, Princeton NJ, pp 416–427
Fuchs G (1989) Alternative pathways of autotrophic CO2 fixation. In: Schlegel HG, Bowen B (eds) Autotrophic bacteria. Science Tech, Madison WI, pp 365–382
George DG, Hunt LT, Yeh LL, Barker WC (1985) New perspectives on bacterial ferredoxin evolution. J Mol Evol 22: 20–31
Gogarten DG, Kibak H, Pittrich P, Taig L, Bowman EJ, Bowman BJ, Manolson MF, Poole RJ, Date T, Oshima T, Konishi J, Denda K, Yoshida M (1989) Evolution of the vacuolar H+-ATPase: implications for the origin of eukaryotes. Proc Natl Acad Sci USA 86:6661–6665
Hooper AB, Dispirito AA (1985) In bacteria which grow on simple reductants, generation of a proton gradient involves extracytoplasmic oxidation of substrate. Microbiol Rev 49: 140–157
Kasting JF (1989) Carbon oxidation state in the early atmosphere: CO2 or CO? Origins Life Evol Biosphere 19:225–226
Koch AL (1985) Primeval cells: possible energy-generating and cell-division mechanisms. J Mol Evol 21:270–277
Mitchell P (1966) Chemiosmotic coupling in oxidative and photosynthetic coupling. Glynn Research Ltd, Bodmin UK
Odum JM, Peck HK Jr (1981) Hydrogen cycling as a general mechanism for energy coupling in the sulfate-reducing bacteria,Desulfovibrio sp. FEMS Microbiol Lett 12:47–50
Ohnishi A, Salerno JC (1982) Iron-sulfur clusters in the mitochondrial electron transport chain. In: Spiro TG (ed) Ironsulfur proteins. John Wiley, New York, pp 285–327
Wächtershäuser G (1988a) Pyrite formation, the first energy source for life: a hypothesis. Syst Appl, Microbiol 10:207–210
Wächtershäuser G (1988b) Before enzymes and templates: theory of surface metabolism. Microbiol Rev 52:452–484
Weast RC (1986) CRC handbook of chemistry and physics. CRC Press, Boca Raton FL, pp D50-D93
Woese CR (1987) Bacterial evolution. Microbiol Rev 51:221–271
Wood PM (1978) A chemiosmotic model for sulfate respiration. FEBS Lett 95:12–18
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Koch, A.L., Schmidt, T.M. The first cellular bioenergetic process: Primitive generation of a proton-motive force. J Mol Evol 33, 297–304 (1991). https://doi.org/10.1007/BF02102860
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02102860