Abstract
Bioenergetic science started in the eighteenth century with the pioneer works by Joseph Priestley and Antoine de Lavoisier on photosynthesis and respiration, respectively. New developments were implemented by Pasteur in the 1860s with the description of fermentations associated with microorganisms, further documented by Buchner brothers who discovered that fermentations also occurred in cell extracts in the absence of living cells. In the beginning of the twentieth century, Harden and Young demonstrated that orthophosphate and other heat-resistant compounds (cozymase), later identified as NAD, ADP, and metal ions, were mandatory in the fermentation of glucose. The full glycolysis pathway has been detailed in the 1940s with the contributions of Embden, Meyeroff, Parnas, and Warburg, among others.
Studies on the citric acid cycle started in 1910 (Thunberg) and were elucidated by Krebs et al. in the 1940s.
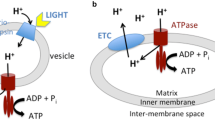
Mitochondrial bioenergetics gained emphasis in the late 1940s and 1950s with the works of Lehninger, Racker, Chance, Boyer, Ernster, and Slater, among others. The prevalent “chemical coupling hypothesis” of energy conservation in oxidative phosphorylation was challenged and replaced by the “chemiosmotic hypothesis” originally formulated in the 1960s by Mitchell and later substantiated and extended to energy conservation in bacteria and chloroplasts, besides mitochondria, with clear-cut identification of molecular proton pumps.
After identification of most reactive mechanisms, emphasis has been directed to structure resolution of molecular complex clusters, e. g., cytochrome c oxidase, complex III, complex II, ATP synthase, photosystem I, photosynthetic water-splitting center, and energy collecting antennae of several photosynthetic systems.
Modern trends concern to the reactivity of radical and other active species in association with bioenergetic activities. A promising trend concentrates on the cell redox status quantified in terms of redox potentials.
In spite of significant development and advances of bioenergetic knowledge, major issues remain mainly related with poor experimental designs not representative of the real native cell conditions. Therefore, a major effort has to be implemented regarding direct observations in situ.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Priestley J (1775) An account of further discoveries in air. Philosoph Trasact 65:384–394
Priestley J (1775) Experiments and observations on different kinds of air, 2nd edn. J. Johnson, London
Lavoisier AL (1789) Traité elementaire de chimie. Cuchet, Paris
Lavoisier AL (1864) In oeuvres de Lavoisier, Tome II. memoires de chimie et de physique. Imprimerie Imperiale, Paris
Lehninger AL (1975) Biochemistry. Worth Publishers, Inc., New York
Buchner E (1897) Alkoholische gärung ohne hefezellen. Berichte der Deutschen Chemischen Gesellshaft 30:117–124
Buchner E, Rapp R (1899) Alkoholische gärung ohne hefezellen. Berichte der Deutschen Chemischen Gesellshaft 32:2086–2094
Mahler HR, Cordes EH (1971) Biological chemistry, 2nd edn. Harper and Row, New York, p 495
Harden A, Young JW (1905) Proc Chem Soc 21:189–195
Stryer L (1995) Biochemistry, 5th edn. W. H. Freeman and Co., New York, pp 483–484
Krebs HA (1970) The history of the tricarboxylic acid cycle. Prespect Biol Med 14:154–170
Lehninger A (1965) The mitochondrion: molecular basis of structure and function. Benjamin, Menlo Park, CA
Mitchell P, Moyle J (1969) Estimation of membrane potential and pH difference across the crystal membranes of rat liver mitochondria. Eur J Biochem 7:471–478
Nicholls DG, Ferguson SJ (1992) Bioenergetics 2. Academic Press, London
Bott M, Thauer RK (1989) Proton translocation coupled to oxidation of carbon monoxide to CO2 and H2 in Methanosarcina barkeri. Eur J Biochem 179:469–472
Wikström MKF (1977) Proton pump coupled to cytochrome c oxidase in mitochondria. Nature 266:271–273
Solioz M, Carafoli E, Ludwig B (1982) The cytochrome c oxidase of Paracoccus denitrificans pumps protons in a reconstituted system. J Biol Chem 257:1579–1582
Yagi T, Matsuno-Yagi (2003) The proton-translocating NADH-quinone oxidoreductase in respiratory chain: the secret unlocked. Biochemistry 42:2266–2274
Hackenbrock CR (1981) Lateral diffusion and electron transfer in mitochondrial inner membrane. Trends Biochem Sci 6:151–154
Madigan MT, Martinko JM, Parker J (1997) Brock biology of microorganisms. Prentice Hall, London
Nelson DL, Cox MM (2000) Lehninger principles of biochemistry, 3rd edn. Worth Publishers, New York
Tsukihara T, Aoyama H, Yamashita E et al (1996) The whole structure of 13-subunit oxidized cytochrome c oxidase at 2.8 Å. Science 272:1136–1144
Iwata S, Osteimer C, Ludwig B, Michel H (1995) Structure at 2.8 Å resolution of cytochrome c oxidase from Paracoccus denitrificans. Nature 376:660–669
Zang Z, Huang L, Shulmeister VM et al (1998) Electron transfer by domain movement in cytochrome bc1. Natura 392:677–684
Yankovskaya V, Horsefield R, Törnroth S et al (2003) Architecture of succinate dehydrogenase and reactive oxygen species generation. Science 299:700–704
Abrahams JP, Leslie AGW, Luter R, Walker JE (1994) Structure at 2.8 Å resolution of F1-ATPase from bovine heart mitochondria. Nature 370:621–628
Chen C, Ko Y, Delannoy M, Ludtke J, Chiu W, Pedersen PL (2004) Mitochondrial ATP synthasome. J Biol Chem 23:31761–31768
Deisenhofer J, Epp O, Sinning I, Michel H (1995) Crystallographic refinement at 2.3 Å resolution and refined model of the photosynthetic reaction centre from Rhodopseudomonas viridis. J Mol Biol 246:429–457
Jordan P, Fromme P, Witt HT, Klukas O, Saenger W, Krauss N (2001) Three-dimensional structure of cyanobacterial photosystem I at 2.5 Å resolution. Nature 411:909–917
Ferreira KN, Iverson TM, Maghlaoui K, Barber J, Iwata S (2004) Architecture of the photosynthetic oxygen-evolving center. Science 303:1831–1838
McDermott G, Prince SM, Freer AA, Hawthornthwaite-Lawless AM, Papiz MZ, Cogdell RJ, Isaacs NW (1995) Crystal structure of an integral membrane light-harvesting complex from photosynthetic bacteria. Nature 374:517–521
Karrasch S, Bullough PA, Gosh R (1995) The 8.5 Å projection map of the light-harvesting complex I from Rhodospirillum rubrum reveals a ring composed of 16 subunits. EMBO J 14:631–638
Liu Z, Yan H, Wang K, Zhang J, Gui L, An X, Chang W (2004) Crystal structure of spinach major light-harvesting complex at 2.72 Å resolution. Nature 428:287–292
Jones DP (2006) Disruption of mitochondrial redox circuitry in oxidative stress. Chem Biol Interact 163:38–53
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Madeira, V.M.C. (2018). Overview of Mitochondrial Bioenergetics. In: Palmeira, C., Moreno, A. (eds) Mitochondrial Bioenergetics. Methods in Molecular Biology, vol 1782. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7831-1_1
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7831-1_1
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7830-4
Online ISBN: 978-1-4939-7831-1
eBook Packages: Springer Protocols