Abstract
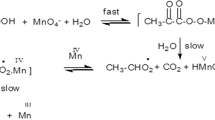
Adonitol is oxidized to ribose by Mn(III) pyrophosphate under the conditions [adonitol] ≫ [Mn(III)]. The oxidation rate is first order with respect to both oxidatn and substrate and decreases inversly with [pyrophosphate]free. Thermodynamic parameters are reported and a suitable mechanism involving free radicals is proposed.
Abstract
Адонитол окисляется до рибозы пирофосфатом марганца(III) в условиях [адонитол]≫ [Mn(III)]. Скорость окисления подчиняется закону первого порядка как по окислителю, так и по субстрату и уменьшается обратно-пропорционально [пирофосфат]своб. Приводятся термодинамические параметры и вероятный свободно-радикальный механизм реакции.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. G. Ram Reddy, B. Sethuram, T. Navneet Rao: Curr. Sci. (India),42, 677 (1973).
A. Meenakshi, M. Santappa: J. Catal.,19, 300 (1970).
A. G. Fadnis, S. K. Kulshrestha: J. Indian Chem. Soc., 763 (1981).
R. P. Bhatnagar, A. G. Fadnis: Montash. Chem.,109, 319 (1978).
I. M. Kolthoff, J. I. Watters: J. Amer. Chem. Soc.,70, 2455 (1948).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fadnis, A.G., Kulshrestha, S.K. Kinetics of oxidation of adonitol by manganese(III) pyrophosphate. React Kinet Catal Lett 19, 267–269 (1982). https://doi.org/10.1007/BF02074043
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02074043