Abstract
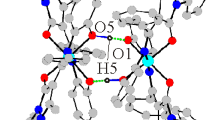
The cobalt(II)-Schiff base complexes [Co(nbsal)2] and [Co(tbsal)2] [nbsal=N-n-butylsalicylideneiminate and tbsal=N-tert-butysalicylideneiminate,o-OC6H4CH=NR,R+Bun and But, respectively] both have distorted tetrahedral structures, but the presence of thetert-butyl groups in [Co(tbsal)2] causes much greater angular distortion, of the coordination tetrahedron. Although [Co(nbsal)2] will react with nitric oxide and oxygen, [Co(tbsal)2] reacts with neither and this appears to be due to the shielding of the cobalt by thetert-vutyl groups. The reactive complex [Co(nbsal)2] crystallizes in the tetragonal system,a+14.244,c+5.395, Å,Z+2 and space group\(P\bar 4\). The structure was determined by the heavy-atom method, using MoKα diffractometer data, and refined by full matrix least-squares toR+0.035 for 777 reflections. The unreactive complex [Co(tbsal)2] crystallizes in the orthorhombic systemPbc21,a+10.977,b+20.037,c+9.866 Å,Z+4. The structure was determined as above toR+0.051 for 1458 reflections.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Groombridge, C J., Larkworthy, L. F., Marécaux, A., Mason, J., Povey, D.C., and Smith, G. W. (1992)J. Chem. Soc. Dalton Trans., 3125.
Killean, R. C. G. and Lawrence J. L. (1969)Acta, Crystallogr B 25, 1750.
Nishikawa, H., and Yamada, S. (1964)Bull. Chem. Soc. Japan,37, 1154.
Orioli, P. L., Di Vaira, M.,and Sacconi, L. (1965),Chem. Commun., 103.
Walker, N. and Stuart, D. (1983)Acta Crystallogr. A39, 158.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bahron, H., Larkworthy, L.F., Marécaux, A. et al. Structures of bis(N-n-butylsalicylideneiminato)cobalt(II) and bis(N-tert-butylsalicylideneiminato)cobalt(II) complexes and reactivity towards oxygen and nitric oxide. J Chem Crystallogr 24, 145–150 (1994). https://doi.org/10.1007/BF01833671
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01833671