Abstract
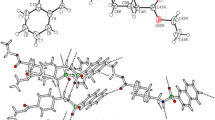
The X-ray structure of the biologically most active polymorph (Form II) of 5-methoxysulfadiazine (1) and that of the 1∶1 complex between this sulfonamide and acetylsalicylic acid (2) are reported. The polymorph1 is monoclinic, space groupP21/c witha=13.086(2) Å,b=5.583(1) Å,c=17.222(3) Å, β=99.98(1)°,Z=4 and occurs as centrosymmetric hydrogen bonded (N−H...N) dimers in the crystal. This arrangement differs from that observed in other polymorphs of the drug. Complex2 is triclinic,\(P\bar 1\),a=8.102(1)Å,b=12.033(1) Å,c=12.170(2) Å, α=111.67(1)°, β=93.77(1)°, γ=103.82(1)°,Z=2. Complexation involves linear intermolecular hydrogen bonds, N−H...O=C and N...O−H, between the amide group and pyrimidinyl N atom of the sulfonamide and the carboxylic group of the acetylsalicylic acid. On complexation to form2, neither molecular component undergoes any major structural change.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Caira, M.R..J. Crystallogr. Spectr. Res. 1991,21, 641.
Caira, M.R.,J. Crystallogr. Spectr. Res. 1992,22, 193.
Caira, M.R.; Mohamed, R..Acta Cryst. 1993,B49, 760.
Rambaud, J.; Alberola, S.; Grignon, H.; Masse, J.; Sabon, F.Trav. Soc. Pharm. Montpellier,1974,34, 293.
Bettinetti, G.P.; Giordano, F.; La Manna, A.; Giuseppetti, G.,Il Farmaco-Ed.Pr. 1974,29, 493.
Yokoyama, T.; Umeda, T.; Kuroda, K.. Watanabe, A.Chem. Pharm. Bull. 1978,26, 1044.
Burger, A.; Ramberger, R.; Schulte, K..Arch. Pharm. (Weinheim) 1980,313, 1020.
Moustafa, M.A.; Khalil, S.A.; Ebian, A.R.; Motawi, M.M.J. Pharm. Pharmac. 1972,24, 921.
Ebian, A.R.; Moustafa, M.F.; Khalil, S.A.; Motawi, M.M..J. Pharm. Pharmac. 1973,25, 13.
Giuseppetti, G.; Tadini, C.; Bettinetti, G.P.; Giordano, F..Cryst. Struct. Comm. 1977,6, 263.
North, A.C.T.; Philips, D.C.; Scott Mathews, F..Acta Crystallogr. 1968,A24, 351.
Frenz, B.A.: Associates Inc. (1982)Structure Determination Package. College Station; Texas.
Sheldrick. G.M. SHELXS86. InCrystallographic Computing 3. Sheldrick, G.M., Kruger, C., and Goddard. R. Eds.: p. 175: Oxford University Press, 1985.
Sheldrick, G.M.. SHELX, A system of computer programs for X-ray structure determination; England, University of Cambridge: 1976.
International Tables for X-Ray Crystallography Vol. IV.1974, (Kynoch Press, Birmingham; (Present distributor: Reidel, D., Dordrecht).
Nardelli, M..Comput. Chem. 1983,7, 95.
Motherwell, S.: PLUTO. A program for plotting molecular and crystal structures: University of Cambridge. England,1979.
Byrn, S.R.:Solid-State Chemistry of Drugs, Academic Press. London: 1982, p. 103.
Yang, S.S.; Guillory, J.K.:J. Pharm. Sci. 1972,61, 26.
Caira, M.R.; Mohamed, R.:Supramolec. Chem. 1993,2, 201.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Caira, M.R. Molecular complexes of sulfonamides. 3. Structure of 5-methoxysulfadiazine (form II) and its 1∶1 complex with acetylsalicylic acid. J Chem Crystallogr 24, 695–701 (1994). https://doi.org/10.1007/BF01668233
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01668233