Summary
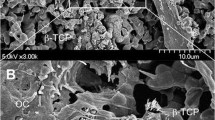
The nature of the multinucleated cells involved in the resorption processes occurring inside macroporous calcium-phosphate biomaterials grafted into rabbit bone was studied using light microscopy, histomorphometric analysis, enzymatic detection of tartrate-resistant acid phosphatase (TRAP) activity, scanning, and electron microscopy. Samples were taken at days 7, 14, and 21 after implantation. As early as day 7, osteogenesis and resorption were observed at the surface of the biomaterials, inside the macropores. Resorption of both newly formed bone and calcium-phosphate biomaterials was associated with two types of multinucleated cells. Giant multinucleated cells were found only at the surface of the biomaterials; they showed a large number of nuclei, were TRAP negative, developed no ruffled border, and contained numerous vacuoles with large accumulation of mineral crystals from the biomaterials. Osteoclasts exhibited TRAP positivity and well-defined ruffled border. They were observed at the surface of both newly formed bone and biomaterials, around the implant, and inside the macropores. In contact with the biomaterials, infoldings of their ruffled border were observed between the mineral crystals, deeply inside the microporosity. The microporosity of the biomaterials (i.e., the noncrystalline spaces inside the biomaterials) increased underneath this type of cell as compared with underneath giant cells or to the depth of the biomaterials. These observations demonstrate that macroporous calcium-phosphate biomaterials implanted in bone elicit osteogenesis and the recruitment of a double multinucleated cell population having resorbing activity: giant multinucleated cells that resorb biomaterials and osteoclasts that resorb newly formed bone and biomaterials.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jarcho M (1981) Calcium phosphate ceramics as hard tissue prosthetics. Clin Orthop 157:259–278
Kenney EB, Lekovic V, Sa Ferreira JC, Han T, Dimitrijevict Carranza FA Jr (1986) Bone formation within porous hydroxyapatite implants in human periodontal defects. J Periodontol 57:76–83
Holmes RE, Bucholz RW, Mooney V (1987) Porous hydroxyapatite as a bone graft substitute in diaphyseal defects: a histometric study. Orthop Res 5:114–121
Okumura M, Ohgushi H, Tamai S, Shors EC (1991) Primary bone formation in porous hydroxyapatite ceramic: a light and scanning electron microscopic study. Cells Mater 1:29–34
Ohgushi H, Goldberg VM, Caplan AI (1989) Heterotopic osteogenesis in porous ceramics induced by marrow cells. J Orthop Res 7:568–578
Takeshita N, Akagi T, Yamasaki M, Ozeki T, Nojima T, Hiramatsu Y, Nagai N (1992) Osteoclastic features of multinucleated giant cells responding to synthetic hydroxyapatite implanted in rat jaw bone. J Electron Microsc 41:141–146
Glowacki J, Cox KA (1986) Osteoclastic features of cells that resorb bone implants in rats. Calcif Tissue Int 39:97–103
Ogilvie A, Frank RM, Benque EP, Gineste M, Heughebaert M, Hemmerle J (1987) The biocompatibility of hydroxyapatite implanted in the human periodontium. J Periodont Res 22:270–283
Marks SC, Chambers TJ (1991) The giant cells recruited by subcutaneous implants of mineralized bone particles and slices in rabbits are not osteoclasts. J Bone Miner Res 6:395–400
Ash P, Loutit JF, Townsend KMS (1980) Osteoclasts derived from haematopoietic stem cells. Nature 283:669–670
Mundy GR, Roodman GD (1987) Osteoclast ontogeny and function. In: Peck WA (ed) Bone and mineral research, Vol 5. Elsevier, Amsterdam, New York, Oxford, pp 209–279
Marks SC, Popoff SN (1988) Bone cell biology: the regulation of development, structure and function in the skeleton. Am J Anat 183:1–44
Vaes G (1988) Cellular biology and biochemical mechanism of bone resorption: a review of recent developments on the formation, activation, and mode of action of osteoclasts. Clin Orthop 231:239–271
Sutton JS, Weiss L (1966) Transformation of monocytes in tissue culture into macrophages, epithelioid cells, and multinucleated giant cells: an electron microscope study. J Cell Biol 28:303–332
Papadimitriou JM, Spector WG (1971) The origin, properties and fate of epithelioid cells. J Pathol 105:187–203
Mariano M, Spector WG (1974) The formation and properties of macrophage polykaryon inflammatory giant cells. J Pathol 113:1–19
Holtrop ME, Cox CA, Glowacki J (1982) Cells of the mononuclear phagocytic system resorb implanted bone matrix: a histologic and ultrastructural study. Calcif Tissue Int 34:488–494
Chambers TJ, Revell PA, Fuller K, Athanasou NA (1984) Resorption of bone by isolated rabbit osteoclasts. J Cell Sci 66:383–399
Jones SJ, Boyde A (1977) Some morphological observations on osteoclasts. Cell Tissue Res 185:387–397
Baron R (1989) Molecular mechanisms of bone resorption by the osteoclast. Anat Rec 224:317–324
Baron R, Neff L, Brown W, Courtoy P, Louvard D, Farquhar MG (1988) Polarized secretion of lysosomal enzymes: codistribution of cation-independent mannose-6-phosphate receptors and lysosomal enzymes along the osteoclast exocytic pathway. J Cell Biol 1063:1863–1872
Blair HC, Teitelbaum SL, Ghiselli R, Gluck S (1989) Osteoclastic bone resorption by a polarized vacuolar proton pump. Science 245:855–857
Arnett TR, Dempster DW (1990) Protons and osteoclasts. J Bone Miner Res 5:1099–1103
Minkin C (1982) Bone acid phosphatase: tartrate-resistant acid phosphatase as a marker of osteoclastic function. Calcif Tissue Int 34:285–290
Andersson GN, Marks SC (1989) Tartrate-resistant acid ATPase as a cytochemical marker for osteoclasts. J Histochem Cytochem 37:115–117
Hattersley G, Chambers TJ (1989) Generation of osteoclastic function in mouse bone marrow cultures: multinuclearity and tartrate-resistant acid phosphatase are unreliable markers for osteoclast differentiation. Endocrinology 124:1689–1696
Modderman WE, Tuinenfburg-Bol Raap AC, Nijweide PJ (1991) Tartrate-resistant acid phosphatase is not an exclusive marker for mouse osteoclasts in cell culture. Bone 12:81–87
Hattersley G, Chambers TJ (1989) Calcitonin receptors as markers for osteoclastic differentiation: correlation between generation of bone-resorptioe cells and cells that express calcitonin receptors in mouse bone marrow cultures. Endocrinology 125:1606–1612
Oursler MJ, Bell LV, Clevinger B, Osdoby P (1985) Identification of osteoclast-specific monoclonal antibodies. J Cell Biol 100:1592–1600
Horton MA, Lewis D, McNulty K, Pringle AS, Chambers TJ (1985) Monoclonal antibodies to osteoclastomas (giant cell bone tumors): Definition of osteoclast-specific antigens. Cancer Res 45:5663–5669
Chilosi M, Giglioli E, Lestani M, Menestrina F, Fiore-Donati L (1988) Immunohistochemical characterization of osteoclasts and osteoclast-like cells with monoclonal antibody MB 1 on paraffin-embedded tissues. J Pathol 156:251–254
Kukita T, Roodman GD (1989) Development of a monoclonal antibody to osteoclasts formed in vitro which recognizes mononuclear osteoclast precursors in the marrow. Endocrinology 125:630–637
Popoff SN, Marks SC (1986) Ultrastructure of the giant cell infiltrate of subcutaneously implanted bone particles in rats and mice. Am J Anat 177:491–503
Kelly J, McNulty JA, Schneider GB (1988) Histochemical and morphological comparison of osteoclasts and multinucleated giant cells recruited to ectopic bone implants. J Cell Biol 37:3376
Kerby JA, Hattersley G, Collins DA, Chambers TJ (1992) Derivation of osteoclasts from hematopoietic colony-forming cells in culture. J Bone Miner Res 7:353–362
Webber D, Osdoby P, Hauschka P, Krukowski M (1990) Correlation of an osteoclast antigen and ruffled border on giant cells formed in response to resorbable substrates. J Bone Miner Res 5:401–410
Krukowski M, Kahn AJ (1982) Inductive specificity of mineralized bone matrix in ectopic osteoclast differentiation. Calcif Tissue Int 34:474–479
Bagi CM, Miller SC (1989) Osteoclast features of cells that resorb demineralized and mineral-containing bone implants in rats. Scanning Microsc 3:963–970
Goldring SR, Roelke M, Glowacki J (1988) Multinucleated cells elicited in response to implants of devitalized bone particles possess receptors for calcitonin. J Bone Miner Res 3:117–120
Glowacki J, Rey C, Glimcher MJ, Cox K, Lian J (1991) A role for osteocalcin in osteoclast differentiation. J Cell Biochem 45:292–302
Franzen A, Heinegard D (1985) Proteoglycans and bone proteins of rat bone. Purification and biosynthesis of major noncollagenous macromolecules. In: Butler WT (ed) The chemistry and biology of mineralized tissues. Ebsco Media, Birmingham, Alabama, pp 132–141
Horton MA (1988) Osteoclast-specific antigens. ISI Atlas of Science. Immunology 1:35–42
Reinholt FP, Kultenby K, Oldberg A, Heinegard D (1990) Osteopontin: a possible anchor of osteoclast to bone. Proc Natl Acad Sci USA 87:4473–4475
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Baslé, M.F., Chappard, D., Grizon, F. et al. Osteoclastic resorption of Ca-P biomaterials implanted in rabbit bone. Calcif Tissue Int 53, 348–356 (1993). https://doi.org/10.1007/BF01351842
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01351842