Summary
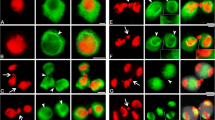
The microtubule and F-actin cytoskeleton of nondifferentiated germlings ofUromyces phaseoli was studied using immunofluorescence methodologies. The microtubules were oriented mostly parallel to the longitudinal axis of the hypha. Microtubule depolymerizing agents, such as cold, demecolcine, griseofulvin and nocodazole, were effective in destroying the microtubule network, but not the F-actin system. Repolymerization of microtubules, following release from these agents, occurred first in the hyphal apices and not near the nuclei or spindle pole bodies. It was concluded that the microtubule nucleating region in such fungal cells is located in the apical regions. Enhanced microtubule arrays were visualized following incubation of the cells in taxol, an agent known to favor microtubule polymerization.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Brooks, R. F., Richmond, R. N., 1983: Microtubule-organizing centres during the cell cycle of 3T3 cells. J. Cell. Sci.61, 231–245.
De Brabander, M., Guens, G., Nuydens, R., Willebrords, R., de Mey, J., 1981: Microtubule assembly in living cells after release from nocodazole block: The effects of metabolic inhibitors, taxol and pH. Cell Biol. Intern. Reports5, 913–920.
Euteneuer, U., Jackson, W. T., McIntosh, J. R., 1982: Polarity of spindle microtubules inHaemanthus endosperm. J. Cell Biol.94, 644–653.
Giloh, H., Sedat, J. W., 1982: Fluorescence microscopy: Reduced photobleaching of rhodamine and fluorescein protein conjugates by n-propyl gallate. Science217, 1252–1255.
Heath, I. B., 1975: The effect of antimicrotubule agents on the growth and ultrastructure of the fungusSaprolegnia ferax and their ineffectiveness in disrupting hyphal microtubules. Protoplasma85, 147–176.
Heath, I. B., 1981: Nucleus-associated organelles in fungi. Intern. Rev. Cytology69, 191–221.
—,Heath, M. C., 1976: Ultrastructure of mitosis in the cowpea rust fungusUromyces phaseoli var.vignae. J. Cell Biol.70, 592–607.
— —, 1978: Microtubules and organelle motility in the rust fungusUromyces phaseoli var.vignae. Cytobiologie16, 393–411.
Hepler, P. K., Palevitz, B. A., 1974: Microtubules and microfilaments. Ann. Rev. Plant Physiol.25, 309–362.
Hoch, H. C., Howard, R. J., 1980: Ultrastructure of freeze-substituted hyphae of the Basidiomycete,Laetisaria arvalis. Protoplasma103, 281–297.
—,Staples, R. C., 1983 a: Ultrastructural organization of the non-differentiated uredospore germling ofUromyces phaseoli. Mycologia75, 795–824.
— —, 1983 b: Visualization of actinin situ by rhodamine-conjugated phalloin in the fungusUromyces phaseoli. Europ. J. Cell. Biol.3, 52–58.
— —, 1984: Cyclic AMP initiates nuclear division and infection structure formation in germlings ofUromyces phaseoli. Exp. Mycol.8, 37–46.
Howard, R. J., 1981: Ultrastructural analysis of hyphal tip cell growth in fungi: Spitzenkörper, cytoskeleton and endomembranes after freeze-substitution. J. Cell Sci.48, 89–103.
Kilmartin, J., Fogg, J., 1982: Partial purification of yeast spindle pole bodies. In: Microtubules in microorganisms (Cappuccinelli, P., Morris, N. R., eds.), pp. 157–170. New York: Marcel Dekker, Inc.
Kilmartin, J. V., Wright, B., Milstein, C., 1982: Rat monoclonal antitubulin antibodies derived by using a new nonsecreting rat cell line. J. Cell Biol.93, 576–582.
Lockwood, A. H., 1980: Cyclic nucleotide dependent protein kinases as molecular regulators of cell morphology. In: Microtubules and microtubule inhibitors (de Brabander, J., de Mey, J., eds.), pp. 71–84. Amsterdam: Elsevier/North-Holland Biomedical Press.
Newhouse, J. R., Hoch, H. C., MacDonald, W. L., 1982: The ultrastructure ofEndothia parasitica: Comparison of a virulent with a hypovirulent isolate. Can. J. Bot.61, 389–399.
O'Donnell, K. L., McLaughlin, D. J., 1981: Ultrastructure of meiosis in the hollyhock rust fungus,Puccinia malvacearum. I. Prophase I — Prometaphase I. Protoplasma108, 225–244.
Osborn, J., Weber, K., 1976: Cytoplasmic microtubules in tissue culture cells appear to grown from an organizing structure towards the plasma membrane. Proc. Nat. Acad. Sci. U.S.A.73, 867–871.
Osborn, M., Webster, R. E., Weber, K., 1978: Individual microtubules viewed by immunofluorescence and electron microscopy in the same PtK2 cell. J. Cell Biol.77, R27-R34.
Pickett-Heaps, J. D., 1969: The evolution of the mitotic apparatus: An attempt at comparative cytology in dividing plant cells. Cytobios1, 257–280.
Puck, T. T., 1977: Cyclic AMP, the microtubule-microfilament system, and cancer. Proc. Nat. Acad. Sci. U.S.A.74, 4491–4495.
Roisen, F. J., Braden, W. G., Friedman, J., 1975: Neurite developmentin vitro. III. The effects of several derivatives of cyclic AMP, colchicine, and colcemid. Ann. N.Y. Acad. Sci.253, 545–561.
Schatten, G., Schatten, H., Bestor, T. H., Balczon, R., 1982: Taxol inhibits the nuclear movements during fertilization and induces asters in unfertilized sea urchin eggs. J. Cell Biol.94, 455–465.
Schiff, P. B., Fant, J., Horwitz, S. B., 1979: Promotion of microtubule assemblyin vitro by taxol. Nature77, 665–667.
—,Horwitz, S. B., 1980: Taxol stabilizes microtubules in mouse fibroblast cells. Proc. Nat. Acad. Sci. U.S.A.77, 1561–1565.
Staples, R. C., Hoch, H. C., 1982: A possible role for microtubules in the induction of nuclear division in bean rust uredospore germlings. Exp. Mycol.6, 293–302.
Toda, T., Yamamoto, M., Yanagida, M., 1981: Sequential alterations in the nuclear chromatin region during mitosis of the fission yeastSchizosaccharomyces pombe: Video fluorescence microscopy of synchrononously growing wild-type and cold-sensitivecde mutants by using a DNA-binding fluorescent probe. J. Cell Sci.52, 271–287.
Weber, K., Osborn, M., 1981: Microtubule and intermediate filament networks in cells viewed by immunofluorescence microscopy. In: Cytoskeletal elements and plasma membrane organization (Poste, G., Nicolson, G. L., eds.), pp. 1–53. Amsterdam: North-Holland Publ. Co.
—,Rathke, P. C., Osborn, M., 1978: Cytoplasmic microtubular images in glutaraldehyde fixed tissue cells viewed by electron microscopy and by immunofluorescence microscopy. Proc. Nat. Acad. Sci. U.S.A.75, 1820–1824.
Wieland, Th., Hollosi, M., Nassal, M., 1983: δ-Aminophalloin, ein 7-analoges Phalloidin, und biochemisch nützliches, auch fluoreszierendes Derivat. Liebigs Ann. Chem.1983, 1533–1540.
Witt, P. L., Ris, H., Borisy, G. G., 1980: Origin of kinetochore microtubules in Chinese hamster ovary cells. Chromosoma81, 483–505.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hoch, H.C., Staples, R.C. The microtubule cytoskeleton in hyphae ofUromyces phaseoli germlings: Its relationship to the region of nucleation and to the F-actin cytoskeleton. Protoplasma 124, 112–122 (1985). https://doi.org/10.1007/BF01279730
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01279730