Abstract
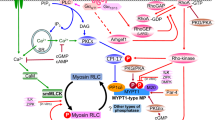
Phosphorylation of the regulatory light chain of myosin by the Ca2+/calmodulin-dependent myosin light chain kinase plays an important role in smooth muscle contraction, nonmuscle cell shape changes, platelet contraction, secretion, and other cellular processes. Smooth muscle myosin light chain kinase is also phosphorylated, and recent results from experiments designed to satisfy the criteria of Krebs and Beavo for establishing the physiological significance of enzyme phosphorylation have provided insights into the cellular regulation and function of this phosphorylation in smooth muscle. The multifunctional Ca2+/calmodulin-dependent protein kinase II phosphorylates myosin light chain kinase at a regulatory site near the calmodulin-binding domain. This phosphorylation increases the concentration of Ca2+/calmodulin required for activation and hence increases the Ca2+ concentrations required for myosin light chain kinase activity in cells. However, the concentration of cytosolic Ca2+ required to effect myosin light chain kinase phosphorylation is greater than that required for myosin light chain phosphorylation. Phosphorylation of myosin light chain kinase is only one of a number of mechanisms used by the cell to down regulate the Ca2+ signal in smooth muscle. Since both smooth and nonmuscle cells express the same form of myosin light chain kinase, this phosphorylation may play a regulatory role in cellular processes that are dependent on myosin light chain phosphorylation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Hai C-M, Murphy RA: Ca2+, crossbridge phosphorylation, and contraction. Annu Rev Physiol 51: 285–298, 1989
Stull JT, Gallagher PJ, Herring BP, Kamm KE: Vascular smooth muscle contractile elements: cellular regulation. Hypertension 17: 723–732, 1991
Sellers JR, Adelstein RS: Regulation of contractile activity. In: PD Boyerand, EG Krebs (eds) The Enzymes. Academic Press, Orlando, 1987, pp 381–418
Korn ED, Hammer JA III: Myosins of nonmuscle cells. Annu Rev Biophys Biophys Chem 17: 23–45, 1988
Ikebe M, Hartshorne DJ, Elzinga M: Phosphorylation of the 20,000-dalton light chain of smooth muscle myosin by the calcium-activated, phospholipid-dependent protein kinase. Phosphorylation sites and effects of phosphorylation. J Biol Chem 262: 9569–9573, 1987
Bengur AR, Robinson EA, Appella E, Sellers JR: Sequence of the sites phosphorylated by protein kinase C in the smooth muscle myosin light chain. J Biol Chem 262: 7613–7617, 1987
Colburn JC, Michnoff CH, Hsu L, Slaughter CA, Kamm KE, Stull JT: Sites phosphorylated in myosin light chain in contracting smooth muscle. J Biol Chem 263: 19166–19173 1988
Kamm KE, Hsu L-C, Kubota Y, Stull JT: Phosphorylation of smooth muscle myosin heavy and light chains: Effects of phorbol dibutyrate and agonists. J Biol Chem 264: 21223–21229, 1989
Singer HA: Protein kinase C activation and myosin light chain phosphorylation in32P-labeled arterial smooth muscle. Am J Physiol 259: C631-C639, 1990
Corson MA, Sellers JR, Adelstein RS, Schoenberg M: Substance P contracts bovine tracheal smooth muscle via activation of myosin light chain kinase. Am J Physiol 259: C258-C265, 1990
Kawamoto S, Bengur AR, Seller JR, Adelstein RS:In situ phosphorylation of human platelet myosin heavy and light chains by protein kinase C. J Biol Chem 264: 2258–2265, 1989
Ludowyke RI, Peleg I, Beaven MA, Adelstein RS: Antigen-induced secretion of histamine and the phosphorylation of myosin by protein kinase C in rat basophilic leukemia cells. J Biol Chem 264: 12492–12501, 1989
Naka M, Nishikawa M, Adelstein RS, Hidaka H: Phorbol esterinduced activation of human platelets is associated with protein kinase C phosphorylation of myosin light chains. Nature 306: 490–492, 1983
Conti MA, Adelstein RS: The relationship between calmodulin binding and phosphorylation of smooth muscle myosin kinase by the catalytic subunit of 3′:5′ cAMP-dependent protein kinase. J Biol Chem 256: 3178–3181, 1981
Miller JR, Silver PJ, Stull JT: The role of myosin light chain kinase phosphorylation in β-adrenergic relaxation of tracheal smooth muscle. Mol Pharmacol 24: 235–242, 1983
de Lanerolle P, Nishikawa M, Yost DA, Adelstein RS: Increased phosphorylation of myosin light chain kinase after an increase in cyclic AMP in intact smooth muscle. Science 223: 1415–1417, 1984
Krebs EG, Beavo JA: Phosphorylation-dephosphorylation of enzymes. Annu Rev Biochem 48: 923–959, 1979
Lukas TJ, Burgess WH, Prendergast FG, Lau W, Watterson DM: Calmodulin binding domains: characterization of a phosphorylation and clamodulin binding site from myosin light chain kinase. Biochemistry 25: 1458–1464, 1986
Payne ME, Elzinga M, Adelstein RS: Smooth muscle myosin light chain kinase. Amino acid sequence at the site phosphorylated by adenosine cyclic 3′,5′-phosphate-dependent protein kinase whether or not calmodulin is bound. J Biol Chem 261: 16346–16350, 1986
Nishikawa M, de Lanerolle P, Lincoln TM, Adelstein RS: Phosphorylation of mammalian myosin light chain kinases by the catalytic subunit of cyclic AMP-dependent protein kinase and by cyclic GMP-dependent protein kinase. J Biol Chem 259: 8429–8436, 1984
Nishikawa M, Hidaka H, Adelstein RS: Phosphorylation of smooth muscle heavy meromyosin by calcium-activated, phospholipid-dependent protein kinase. The effect on actin-activated MgATPase activity. J Biol Chem 258: 14069–14072, 1983
Hashimoto Y, Soderling TR: Phosphorylation of smooth muscle myosin light chain kinase by Ca2+/calmodulin-dependent protein kinase-II. Comparative study of the phosphorylation sites. Arch Biochem Biophys 278: 41–45, 1990
Ikebe M, Reardon S: Phosphorylation of smooth myosin light chain kinase by smooth muscle Ca2+/calmodulin-dependent multifunctional protein kinase. J Biol Chem 265: 8975–8978, 1990
Nomura M, Stull JT, Kamm KE, Mumby MC: Site-specific dephosphorylation of smooth muscle myosin light chain kinase by protein phosphatases 1 and 2A. Biochemistry 31: 11915–11920, 1992
Pato MD, Kerc E: Comparison of the properties of the protein phosphatases from avian and mammalian smooth muscles: purification and characterization of rabbit uterine smooth muscle phosphatases Arch Biochem Biophys 276: 116–124, 1990
Stull JT, Hsu L-C, Tansey MG, Kamm KE: Myosin light chain kinase phosphorylation in tracheal smooth muscle. J Biol Chem 265: 16683–16690, 1990
Tansey MG, Word RA, Hidaka H, Singer HA, Schworer CM, Kamm KE, Stull JT: Phosphorylation of myosin light kinase by the multifictional calmodulin-dependent protein kinase II in smooth muscle cells. J Biol Chem 267: 12511–12516, 1992
Tansey MG, Stull JT: Phosphorylation of myosin light chain kinase by the multi-functional calmodulin-dependent protein kinase affects the calcium sensitivity of myosin light chain phosphorylation. Biophys J 61: A316, 1992 (Abstract)
Tang D-C, Stull JT, Kubota Y, Kamm KE: Regulation of the Ca2+ dependence of smooth muscle contraction. J Biol Chem 267: 11839–11845, 1992
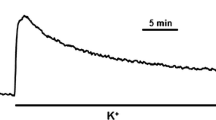
Taylor DA, Bowman BF, Stull JT: Cytoplasmic Ca2+, is a primary determinant for myosin phosphorylation in smooth muscle cells. J Biol Chem 264: 6206–6213, 1989
Word RA, Tang DC, Kamm KE: Increases in [Ca2+] myosin light chain (LC) phosphorylation, and force during the initiation of smooth muscle contraction. Biophys J 64: A258, 1993 (Abstract)
Carafoli E: Calcium pump of the plasma membrane. Physiol Rev 71: 129–153, 1991
Ryu SH, Lee SY, Lee KY, Rhee SG: Catalytic properties of inositol trisphosphate kinase: activation by Ca2+ and calmodulin. FASEB J 1: 388–393, 1987
Forstermann U, Pollock JS, Schmidt HH, Heller M, Murad F Calmodulin-dependent endothelium-derived relaxing factor/nitric oxide synthase activity is present in the particulate and cytosolic fractions of bovine aortic endothelial cells. Proc Natl Acad Sci USA 88: 1788–1792, 1991
Gallagher PJ, Herring BP, Griffin SA, Stull JT: Molecular characterization of a mammalian smooth muscle myosin light chain kinase. J Biol Chem 266: 23936–23944, 1991
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stull, J.T., Tansey, M.G., Tang, DC. et al. Phosphorylation of myosin light chain kinase: a cellular mechanism for Ca2+ desensitization. Mol Cell Biochem 127, 229–237 (1993). https://doi.org/10.1007/BF01076774
Issue Date:
DOI: https://doi.org/10.1007/BF01076774