Abstract
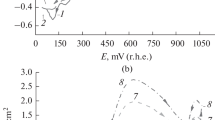
A study of the mechanism of electrooxidation of 1,3-propanediol on a gold electrode in basic medium has been made. The kinetic parameters measured, together with other experimental data, allow the formulation of a mechanism in which both 1,3-propanediol and OH− ions adsorb on the gold surface and the rate determining step is an interaction between both coverages with the result of the formation of an adsorbed radical, whose oxidation leads to the formation of 3-hydroxypropanoic acid which has been identified by nuclear magnetic resonance (NMR).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
H. Cnobloch, D. Groppel, H. Kohlmuller, D. Kuhl and G. Siemsen, in ‘Power Sources 7’ (edited by J. Thomson) (1979) Vol. 24, p. 389.
F. Kadirgan, B. Beden and C. Lamy,J. Electroanal. Chem. 136 (1982); 119;143, (1983) 135.
E. Santos and M. C. Giordano,Electrochim. Acta 30 (1985) 871.
E. Sokolova,Electrochim. Acta 24 (1979) 24.
H. Huser, J. M. Leger and C. Lamy,Electrochim. Acta 30 (1985) 1409.
S. N. Raicheva,Z. Phys. Chem. 264 (1983) 65.
S. N. Raicheva, E. I. Sokolova, S. Kalcheva and W. Vielstich, 29th Meeting of the ISE, Budapest, ‘Ext. Abstr’ (1978) p. 780.
P. I. Iotov, S. V. Kalcheva, E. I. Sokolova and S. N. Raicheva,Dok. Bulg. Akad. Nauk. 39 (1986).
R. Holze and M. Beltowska-Brzezinska,J. Electroanal. Chem. 201 (1986) 387.
F. Kadirgan, Thesis, Université de Poitiers, (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alonso, C., Gonzalez-Velasco, J. Study of the electrooxidation of 1,3-propanediol on a gold electrode in basic medium. J Appl Electrochem 18, 538–545 (1988). https://doi.org/10.1007/BF01022248
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01022248