Abstract
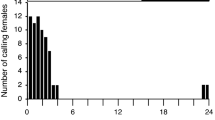
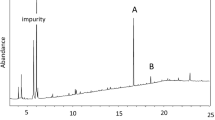
A field survey of 10,12-hexadecadienyl alcohols, acetates, and aldehydes showed attraction to a wide range of sphingid moths. Data are presented showing the attraction ofSmerinthus jamaicensis, Smerinthus cerisyi, Pachysphinx modesta, Hemaris diffinis, andProserpinus flavofasciata to these compounds. Mass spectral, EAG, and EAD data show evidence for the presence of these dienes in female extracts ofS. cerisyi, Hyles gallii, andSphinx drupiferarum.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arn, H., Städler, F., andRauscher, S. 1975. The electroantennographic detector—a selective and sensitive tool in the gas chromatographic analysis of insect pheromones.Z.Naturforsch.30c:722–725.
Butenandt, A., Beckman, R., Stamm, D., andHecker, F. 1959. Über den Sexual-Lockstoff des SeidenspinnersBombyx mori. Reindarstellung und Konstitution.Z. Naturforsch. Teil. B 14:283–284.
Chisholm, M.D., Steck, W.F., Arthur, A.P., andUnderhill, E.W. 1975. Evidence forcis 11-hexadecen-1-ol acetate as a major component of the sex pheromone of the bertha armyworm,Mamestra configurata (Lepidoptera: Noctuidae).Can. Entomol. 107:361–366.
Chisholm, M.D., Steck, W.F., Bailey, B.K., andUnderhill, E.W. 1981. Synthesis of sex pheromone components of the forest tent caterpillar,Malacosoma disstria (Hübner) and the western tent caterpillar,Malacosoma californicum (Packard).J. Chem. Ecol. 7:159–163.
Chisholm, M.D., Reed, D.W., Underhill, E.W., Palaniswamy, andP. Wong, 1985. Attraction of tortricid moths of subfamily Olethreutinae to field traps baited with dodecadienes.J. Chem. Ecol. 11:217–230.
Hall, D.R., Beevor, P.S., Lester, R., andNesbitt, B.F. 1980. (E,E)-10,12-Hexadecadienal: A component of the female sex phermone of the spiny bollworm,Earis insulana (Boisd.) (Lepidoptera: Noctuidae).Experientia 36:152–154.
Harris, P. 1984.Euphorbia esula-virgata complex, leafy spurge andE. cyparissias L., cypress spurge (Euphorbiaceae), pp. 159–169,in J.S. Kelleher and M.A. Hulme (eds). Biological Control Programmes against Insects and Weeds in Canada 1969–1980. Commonwealth Agricultural Bureau, Page Bros., Norwich, England.
Kasang, G., Kaissling, K.E., Vostrowsky, O., andBestmann, H.J. 1978a. Bombykal a second pheromone component of the silkworm mothBombyx mori L.,Angew.Chem. Int. Ed. Eng. 17:60–63.
Kasang, G., Schneider, D., andSchäfer, W. 1978b. The silkworm mothBombyx mori. Presence of the (E,E)-stereoisomer of bombykol in the female pheromone gland 1978.Naturwissenschaften 65:337–338.
Kuwahara, Y., andYen, L.T.M. 1979. Identification of bombykol inBombyx mandarina females (Lepidoptera: Bombycidae).Appl. Entomol. Zool. 14:114–117.
Reed, D.W., andChisholm, M.D. 1985. Attraction of moth species of the Tortricidae, Gelechiidae, Geometridae, Drepanidae, Pyralidae and Gracillariidae families to field traps baited with conjugated dienes.J. Chem. Ecol. 11:1645–1657.
Starratt, A.N., Dahm, K.H., Allen, N., Hildebrand, J.G., Payne, T.L., andRöller, H. 1979. Bombykal, a sex pheromone of the sphinx mothManduca sexta. Z. Naturforsch. 34c:9–12.
Steck, W., andBailey, B.K. 1978. Pheromone traps for moths: Evaluation of cone trap designs and design parameters.Environ. Entomol. 7:449–455.
Author information
Authors and Affiliations
Additional information
NRCC No. 25857.
In this paper the chemical nomenclature is abbreviated as follows: Z and E indicate thecis andtrans configurations of the double bond, the number followingZ orE indicates the position of that double bond, the last number indicates the carbon chain length, the terminal functional groups are: Ac=acetate; :OH=alcohol; :Aid=aldehyde.
Rights and permissions
About this article
Cite this article
Reed, D.W., Underhill, E.W. & Giblin, E.M. Attraction of sphingid moths (Lepidoptera: Sphingidae) to 10,12-hexadecadienyl aldehydes and acetates: Evidence of pheromone components. J Chem Ecol 13, 931–942 (1987). https://doi.org/10.1007/BF01020172
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01020172