Abstract
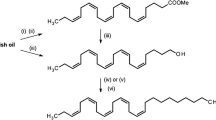
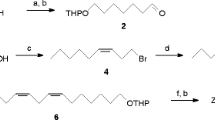
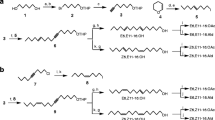
The synthesis and biological activity of some analogs of (Z)-13-hexadecen-11-ynyl acetate1, the major component of the sex pheromone of the processionary mothThaumetopoea pityocampa is described. The analogs have been formally derived by structural modification of the enyne and acetate functions of the parent compound1. In field tests, trifluoroacetate ester16 and the analog,11, with fluorine substitution at the olefin site, decreased the pheromone action, whereas epoxy derivative,10, from epoxidation of the olefin moiety in1, and propionate ester15 gave synergistic activity. The formate14 had a variable effect according to the composition of the lure. Formal reduction of the enyne to give the acetylene2 was found to retain activity. Alcohols12 and13, resulting from hydrolysis of the enyne1 and acetylene2, respectively, inhibited the action of their parent compounds.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Albans, K.R., Baker, R., andJones, O.T. 1984. Inhibition of response ofHeliothis virescens to its natural pheromone by antipheromones.Crop Prot. 3(4):501–506.
Brattesani, D.N., andHeathcock, H. 1973. A convenient procedure for the alkylation of acetylenes.Synth. Commun. 3:245–248.
Brown, C.A., andYamashita, A. 1975. The acetylenic zipper. An exceptionally facile “contrathermodynaraic” multipositional isomerization of alkynes with potassium 3-aminopropylamide.J. Am. Chem. Soc. 97:891–892.
Camps, F.,Canela, R.,Coll, J.,Guerrero, A., andRiba, M. 1981a. Synthesis of the two isomers of the potential sex pheromone ofThaumetopoea pityocampa (Lepidoptera, Notodontidae) and related model compounds.Chem. Lett. 703–706.
Camps, F.,Coll, J.,Guerrero, A., andRiba, M. 1981b. Procedimiento para la preparación por síntesis del acetato de (Z)-13-hexadecen-11-inilo con posible actividad de feromona sexual sobre la procesionaria del pino. Spain Patent No. 502990.
Camps, F., Coll, J., Guerrero, A., andRiba, M. 1983. Simple and stereoselective synthesis of the sex pheromone of the processionary mothThaumetopoea pityocampa (Denis and Schiff.).J. Chem. Ecol. 9:869–875.
Camps, F., Fabrias, G., andGuerrero, A. 1986. Synthesis of a fluorinated analog of the sex pheromone of the processionary mothThaumetopoea pityocampa (Denis and Schiff.).Tetrahedron 42:3623–3629.
Camps, F.,Gasol, V.,Guerrero, A., andLlebaria, A. 1987a. Procedimiento para la preparación por síntesis del acetato de (Z)-13-hexadecen-11-inilo y su aplicación como feromona sexual para el control de la procesionaria del pino.T. pityocampa (Denis y Schiff.). Spain Patent No. 8700834.
Camps, F.,Gasol, V.,Guerrero, A., andLlebaria, A. 1987b. Procedimiento para la preparación por síntesis de esteres acetilénicos y su aplicación como atrayentes sexuales al control de la procesionaria del pinoT. pityocampa (Denis y Schiff.). Spain Patent No. 8700835
Cuevas, P., Montoya, R., Belles, X., Camps, F., Coll, J., Guerrero, A., andRiba, M. 1983. Initial field trials with the synthetic sex pheromone of the processionary mothThaumetopoea pityocampa (Denis and Schiff.).J. Chem. Ecol. 9:85–93.
Guerrero, A., Camps, F., Coll, J., Riba, M., Einhorn, J., Descoins, C.H., andLallemand, J.Y. 1981. Identification of a potential sex pheromone of the processionary mothThaumetopoea pityocampa (Lepidoptera, Notodontidae).Tetrahedron Lett. 22(21):2013–2016.
Guerrero, A., Murgo, R., andMartorell, X. 1986. An improved electroantennogram apparatus with a new automatic sample injection system.Physiol. Entomol. 11:273–277.
Kamm, J.A., andMcDonough, L.M. 1980. Synergism of the sex pheromone of the cranberry girdler.Environ. Entomol. 9:795–797.
Liljefors, F., Thelin, B., andVan Der Pers, J.N.C. 1984. Structure-activity relationships between stimulus molecule and response of a pheromone receptor cell in turnip mothAgrotis segetum.J. Chem. Ecol. 10:1661–1675.
Michelot, D.,Guerrero, A., andRatovelomanana, V. 1982. Une synthèse courte et stéréoselective de l'acetoxyl-1-hexadécène-13(Z)-yne-11: Principal constituant de la secretion pheromonale produite par la femelle vierge de la processionaire du pinThaumetopoea pityocampa (Denis and Schiff.) (Lepidoptera, Notodontidae).J. Chem. Res. (S) 93.
Montoya, R. 1984. Descripción de un nuevo modelo de trampa para captura de machos de procesionaria del pino.Bol. Est. Cent. Ecol. 13:99–103.
Norris, W.P. 1959. Preparation of propene-d-1.J. Org. Chem. 24:1579.
Priesner, E. 1979. Specificity studies on pheromone receptors in noctuid and tortricid Lepidoptera, pp. 57–71,in F.J. Ritter (ed.). Chemical Ecology: Odour Communications in Animals. Elsevier/North Holland, Amsterdam.
Roelofs, W.L., andComeau, A. 1971. Sex pheromone perception: Synergists and inhibitors for the red-banded leaf roller attractant.J. Insect Physiol. 17:435–448.
Shani, A., Klug, J.T., andSkorka, J. 1983. Stereoselective synthesis of (Z)-13-hexadecen-11-yn-1-yl acetate, the major component of the pine processionary mothThaumetopoea pityocampa J. Chem. Ecol. 9:863–867.
Author information
Authors and Affiliations
Additional information
Lepidoptera, Thaumetopoeidae.
Rights and permissions
About this article
Cite this article
Camps, F., Fabriàs, G., Gasol, V. et al. Analogs of sex pheromone of processionary moth,Thaumetopoea pityocampa: Synthesis and biological activity. J Chem Ecol 14, 1331–1346 (1988). https://doi.org/10.1007/BF01020138
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01020138