Abstract
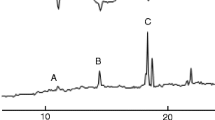
The chirality of the pheromone of the rice weevil,Sitophilus oryzae (L.), and the maize weevil,S. zeamais (Motschulsky), 5-hydroxy-4-methyl-3-heptanone, was determined using an acetyl lactate derivatization procedure. Maize weevils were shown to produce >98% 4S,5R. Determination was more difficult with rice weevils due to a smaller quantity of insect extract, but they were shown to produce at least 92% 4S, 5R.
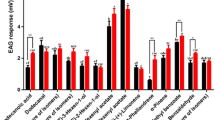
The attractancy of the four synthetic stereoisomers of 5-hydroxy-4-methyl-3-heptanone was tested using rice and maize weevils. As expected, both species were most strongly attracted to the 4S, 5R enantiomer. Maize weevils also showed low but significant responses (P < 0.05) to both 4R, 5R and 4S,5S. Rice weevils showed a highly significant (P < 0.01) response to 4R, 5S, although it was only about one third the response to 4S, 5R. Thus, (4S,5R)-5-hydroxy-4-methyl-3-heptanone is clearly the major component of the pheromone of bothS. zeamais andS. oryzae.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Duncan, D.B. 1955. Multiple range and multipleF tests.Biometrics 11:1–41.
Halstead, D.G.H. 1963. External sex differences in stored-products Coleoptera.Bull. Entomol. Res. 54:119–134.
Mori, K. 1984. The significance of chirality: Methods for determining absolute configuration and optical purity of pheromones and related compounds, pp. 323–370,in H.E. Hummel and T.A. Miller (eds.). Techniques in Pheromone Research. Springer-Verlag, New York.
Mori, K. andEbata, T. 1986. Synthesis of all of the four possible stereoisomers of 5-hydroxy-4-methyl-3-heptanone (sitophilure), the aggregation pheromone of the rice weevil and the maize weevil.Tetrahedron 42:4421–4426.
Phillips, J.K. andBurkholder, W.E. 1981. Evidence for a male-produced aggregation pheromone in the rice weevil.J. Econ. Entomol. 74:539–542.
Phillips, J.K., Walgenbach, C. A., Klein, J.A., Burkholder, W.E., Schmuff, N.R., andFales, H.M. 1985. (R*,S*)-5-hydroxy-4-methyl-3-heptanone: A male-produced aggregation pheromone ofSitophilus oryzae (L.) andS. zeamais Motsch.J. Chem. Ecol. 11:1263–1274.
Schmuff, N.R., Phillips, J.K., Burkholder, W.E., Fales, H.M., Chen, C.W., Roller, P.P., andMa, M. 1984. The chemical identification of the rice weevil and maize weevil aggregation pheromone.Tetrahedron Lett. 25:1533–1534.
Silverstein, R.M. 1979. Enantiomeric composition and bioactivity of chiral semiochemicals in insects, pp. 133–146,in F.J. Ritter (ed.). Chemical Ecology: Odor Communication in Animals. Elsevier/North Holland Biomedical Press, Amsterdam.
Slessor, K.N., King, G.G.S., Miller, D.R., Winston, M.L., andCutforth, T.L. 1985. Determination of chirality of alcohol or latent alcohol semiochemicals in individual insects.J. Chem. Ecol. 11:1659–1667.
Walgenbach, C.A., andBurkholder, W.E. 1986. Factors affecting the response of the maize weevil,Sitophilus zeamais (Coleoptera: Curculionidae), to its aggregation pheromone.Environ. Entomol. 15:733–738.
Walgenbach, C.A., Phillips, J.K., Faustini, D.L. andBurkholder, W.E. 1983. Male-produced aggregation pheromone of the maize weevil,Sitophilus zeamais, and interspecific attraction between threeSitophilus species.J. Chem. Ecol. 9:831–841.
Author information
Authors and Affiliations
Additional information
This article reports the results of research only. Mention of a proprietary product does not constitute an endorsement or a recommendation for its use by USDA.
Rights and permissions
About this article
Cite this article
Walgenbach, C.A., Phillips, J.K., Burkholder, W.E. et al. Determination of chirality in 5-hydroxy-4-methyl-3-heptanone, the aggregation pheromone ofSitophilus oryzae (L.) andS. zeamais Motschulsky. J Chem Ecol 13, 2159–2169 (1987). https://doi.org/10.1007/BF01012564
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01012564