Abstract
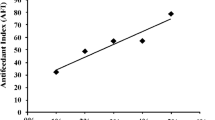
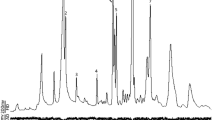
NeonatePlutella xylostella moved more rapidly, spent more time walking, and engaged in searching behaviors more often on leaves of NY 8329, a resistant cabbage with glossy leaves, than on Round-Up, a susceptible variety with normal wax bloom. The neonates also spent significantly more time palpating and more time biting and spinning silk on the susceptible cabbage (although the latter two differences were not significant). Very similar differences in neonate behavior occurred on leaf surface wax extracts (hexane and dichloromethane) of the two cabbage genotypes. Leaf surface waxes are thus strongly implicated in eliciting reduced acceptance of the glossy cabbage by neonateP. xylostella. The chemical compositions of the leaf wax extracts were markedly different. Several compounds, including the triterpenolsα- andβ-amyrin, were found only in the glossy waxes. The percentages of some major wax constituents differed between wax extracts of the two cabbage types. These differences in wax composition may condition the plant resistance in glossy types.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Albert, P.J., andParisella, S. 1983. Chemical bases of host-plant selection by eastern spruce budworm (Choristoneura fumiferana Clem.) (Lepidoptera: Tortricidae). Proceedings, Forest Defoliator-Host Interactions: A Comparison Between Gypsy Moth and Spruce Budworm, USDA Forest Service, General Technical Report NE-85 9-14.
Baker, E.A. 1974. The influence of environment on leaf wax development inBrassica oleracea var. gemmifera.New Phytol. 73:955–966.
Bernays, E.A.,Blaney, W.M.,Chapman, R.F., andCook, A.G. 1976. The ability ofLocusta migratoria L. to perceive plant surface waxes, pp. 35–40,in T. Jermy and A. Szentesi (eds.). The Host-Plant in Relation to Insect Behavior and Reproduction. Symposia Biologica Hungarica, Budapest.
Bernays, E.A., Woodhead, S., andHaines, L. 1985. Climbing by newly hatched larvae of the spotted stalk borerChilo partellus to the top of sorghum plants.Entomol. Exp. Appl. 39:73–79.
Bianey, W.M., andChapman, R.F. 1970. The function of the maxillary palps of Acrididae.Entomol. Exp. Appl. 13:363–376.
Chapman, R.F., andBernays, E.A. 1989. Insect behavior at the leaf surface and learning as aspects of host plant selection.Experientia 45:215–222.
Dickson, M.H., andEckenrode, C.J. 1980. Breeding for resistance in cabbage and cauliflower to cabbage looper, imported cabbageworm and diamondback moth.J. Am. Soc. Hortic. Sci. 105:782–785.
Dickson, M.H., Eckenrode, C.J., andBlamble, A.E. 1984. NYIR 9602, NYIR 9605, and NYIR 8329. lepidopterous pest resistant cabbages.HortSci. 19:311–312.
Dickson, M.H., Shelton, A.M., Eigenbrode, S.D., Vamosy, M.L., andMora, M. 1990. Selection for resistance to diamondback moth (Plutella xylostella) in cabbage.HortSci. 25:1643–1646.
Eigenbrode, S.D., andShelton, A.M. 1990. Behavior of neonate diamondback moth larvae (Lepidoptera: Plutellidae) on glossy-leaved resistant genotypes ofBrassica oleracea.Environ. Entomol. 19:566–1571.
Eigenbrode, S.D., Barnard, J., andShelton, A.M. 1989. A system for quantifying behavior of neonate caterpillars and other small, slow-moving animals.Can. Entomol. 121:1125–1126.
Eigenbrode, S.D., Shelton, A.M., andDickson, M.H. 1990. Two types of resistance to the diamondback moth (Lepidoptera: Plutellidae) in cabbage.Environ. Entomol. 19:1086–1090.
Eigenbrode, S.D.,Stoner, K.A.,Shelton, A.M., andKain, W.C. 1991. Role of leaf waxes in resistance to diamondback moth larvae in glossy-leavedBrassica oleracea.J. Econ. Entomol. In press.
Espelie, K.E., andBernays, E.A. 1989. Diet-related differences in the cuticular lipids ofManduca sexta larvae.J. Chem. Ecol. 15:2003–2017.
Greenway, A.C., Griffiths, D.C., andLloyd, S.L. 1978. Response ofMyzus persicae to components of aphid extracts and to carboxylic acids.Entomol. Exp. Appl. 24:369–374.
Heupel, R.C. 1985. Varietal similarities and differences in the polycyclic isopentenoid composition of sorghum.Phytochemistry 24:2929–2937.
Holloway, P.J., Jeffree, C.E., andBaker, E.A. 1976. Structural determination of secondary alcohols from plant epicuticular waxes.Phytochemistry 15:1768–1770.
Holloway, P.J., Brown, G.A., Baker, E.A., andMacey, M.J.K. 1977. Chemical composition and ultrastructure of the epicuticular wax in three lines ofBrassica napus (L.).Chem. Phys. Lipids 19:114–127.
Jeffree, C.E. 1986. The cuticle, epicuticular waxes and trichomes of plants, with reference to their structure, functions and evolution, pp. 23–64,in B.E. Juniper and T.R.E. Southwood (eds.). Insects and the Plant Surface. Edward Arnold, London.
Jeffree, C.E., Baker, E.A., andHolloway, P.J. 1975. Ultrastructure and recrystallization of plant epicuticular waxes.New Phytol. 75:539–549.
Klingauf, F., Nöcker-Wenzel, K., andKlein, W. 1971. Einfluss einiger Wachskomponenten vonVicia faba L. auf das Wirtswahlverhalten vonAcyrthosiphon pisum (Harris) (Homoptera: Aphididae).Z. Pflanzenkrank. Pflanzensch. 78:641–648.
Klingauf, F., Nöcker-Wenzel, K., andRöttger, U. 1978. Die Rolle peripherer Phlanzen-wachse fr den Befall durch phytophage Insekten.Z. Pflanzenkrank. Pflanzensch. 85:228–237.
Lin, J., Eckenrode, C.J., andDickson, M.H. 1983. Variation inBrassica oleracea resistance to diamondback moth (Lepidoptera: Plutellidae).J. Econ. Entomol. 76: 1423–1427.
Lin, J., Dickson, M.H., andEckenrode, C.J. 1984. Resistance ofBrassica lines to the diamondback moth (Lepidoptera: Yponomeutidae) in the field, and inheritance of resistance.J. Econ. Entomol. 77:1293–1296.
Maloney, P.J., Albert, P.J., andTulloch, A.P. 1988. Influence of epicuticular waxes from white spruce and balsam fir on feeding behavior of the eastern spruce budworm.J. Insect Behav. 1:197–208.
McKibben, G.H., Thompson, M.J., Parrott, W.L., Thompson, A.C., andLusby, W.R. 1985. Identification of feeding stimulants for boll weevilsAnthonomus grandis from cotton buds and anthers.J. Chem. Ecol. 11:1229–1238.
Mori, M. 1982. n-Hexacosanol and n-octacosonol; feeding stimulants for larvae of the silkworm,Bombyx mori.J. Insect Physiol. 28:969–973.
Nayar, J.K., andFraenkel, G. 1962. The chemical basis of hostplant selection in the silkworm,Bombyx mori (L.).J. Insect Physiol. 8:505–525.
Netting, A.G., Macey, M.J.K., andBarber, H.N. 1972. Chemical genetics of a subglaucous mutant ofBrassica oleracea.Phytochemistry 11:579–585.
Salinas, P.J. 1984. Studies on the behavior of the larvae ofPlutella xylostella (Linnaeus)(Lepidoptera: Plutellidae), a world pest of cruciferous crops: Normal and spacing behavior.Turrialba 34:77–84.
Shankaranarayana, K.H., Ayyar, K.S., andKrishna Rao, G.S. 1980, Insect growth inhibitor from the bark ofSantalum album.Phytochemistry 19:1239–1240.
Shelton, A.M.,Wyman, J.A.,Cushing, N.L.,Apfelbeck, K.,Dennehy, T.J.,Mahr, S.E.R., andEigenbrode, S.D. 1991. Resistance of diamondback moth to insecticides in North America.J. Econ. Entomol. In press.
Talekar, N.S., andGriggs, T.D. (eds.). 1986. Diamondback moth management. Proceedings of the First International Workshop, Tainan, Taiwan. AVRDC, Shanhua.
Varela, L., andBernays, E.A. 1988. Behavior of newly hatched potato tuber moth larvae,Phthorimaea operuclella Zell. (Lepidoptera: Gelechiidae), in relation to their host plants.J. Insect Behav. 1:261–275.
Woodhead, S. 1983. Surface chemistry ofSorghum bicolor and its importance in feeding byLocusta migratoria.Physiol. Entomol. 8:345–352.
Woodhead, S. 1987. The influence of surface chemicals of sorghum on the behavior of the stem-borerChilo partellus (Swinhoe), p. 425,in V. Labeyrie, G. Fabres, and D. Lachaise (eds.). Insects-Plants: Proceedings, 6th International Symposium on Insect-Plant Relationships. Junk, Dordrecht.
Woodhead, S., andPadgham, D.E. 1988. The effect of plant surface characteristics on resistance of rice to the brown planthopper,Nilaparvata lugens.Entomol. Exp. Appl. 47:15–22.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eigenbrode, S.D., Espelie, K.E. & Shelton, A.M. Behavior of neonate diamondback moth larvae [Plutella xylostella (L.)] on leaves and on extracted leaf waxes of resistant and susceptible cabbages. J Chem Ecol 17, 1691–1704 (1991). https://doi.org/10.1007/BF00984697
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00984697