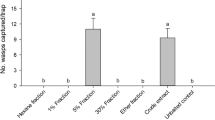
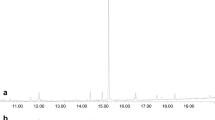
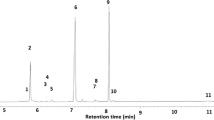
Abstract
Enantiomer separation of (6Z,9Z)-cis-3,4-epoxynonadecadiene and (3Z,9Z)-cis-6,7-epoxynonadecadiene could be achieved using chiral high-resolution gas chromatography and a cyclodextrin-bond column. (3Z,9Z)-(6R,7S)-Epoxynonadecadiene was identified from ovipositor extracts ofColotois pen-Naria, while inErannis defoliaria the 6S,7R-enantiomer was found. In field trapping tests pure synthetic enantiomers caught only conspecific males of these species. (3Z,6Z,9Z)-Nonadecatriene was found in both species, while the presence of (3Z,6Z,9Z)-heneicosatriene was indicated inC. Pennaria only. A 10∶10∶3 blend of (3Z,9Z)-(6R,7S)-epoxynonadecadiene, (3Z,6Z,9Z)-heneicosatriene, and (3Z,6Z,9Z)-nonadecatriene was found to be optimal for catchingC. Pennaria, whileE. Defoliaria males were optimally caught by a 1∶1 mixture of (3Z,9Z)-(6S,7R)-epoxynonadecadiene and (3Z,6Z,9Z)-nona-decatriene. (6Z,9Z)-(3S,4R)-Epoxynonadecadiene was identified from ovipositor extracts ofAgriopis (Erannis) aurantiaria. In field tests the pure enantiomer proved to be a highly specific sex attractant for both the late autumn/early winter flyingA. Aurantiaria and the late winter/early spring flyingA. Leucophearia. Males ofAgriopis marginaria, which fly in late winter/early spring, were attracted to (3Z,9Z)-(6S,7R)-epoxynonadecadiene. The addition of (3Z,6Z,9Z)-nonadecatriene to theS,R-enantiomer increased captures. Optimal catches were recorded with a 10∶3 epoxide-hydrocarbon blend. Enantiomer specificity in all species was confirmed in EAG measurements.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arn, H., Rauscher, S., andSchmid, A. 1979. Sex attractant formulations and traps for the grape mothEupoecilia ambiguella.Mitt. Schweiz. Entomol. Ges. 52:49–55.
Arn, H., Tóth, M., andPriesner, E. 1992. List of sex pheromones of Lepidoptera and related attractants, 2nd. ed., International Organisation for Biological Control; Montfavet. 179 pp.
Becker, D., Cyjon, R., Cossé, A., Moore, L., Kimmel, T., andWysoki, M. 1990. Identification and enantioselective synthesis of (Z,Z)-6,9-cis-3S,4R-epoxynonadecadiene, the major sex pheromone component ofBoarmia selenaria.Tetrahedron Lett. 31:4923–4926.
Brown, C.A., andAhuja, V.K. 1973. P-2 nickel catalyst with ethylenediamine, a novel system for highly stereospecific reduction of alkynes tocis-olefins.J. Chem. Soc. Chem. Commun. 1973:553–554.
Cossé, A.A., Cyjon, R., Moore, I., Wysocki, M., andBecker, D. 1992. Sex pheromone components of the giant looper,Boarmia selenaria Schiff. (Lepidoptera: Geometridae).J. Chem. Ecol. 18:165–182.
Escherich, K. 1931. Die Forstinsekten Mitteleuropas. III. Paul Parey Verlag, Berlin, 826 pp.
Hansson, B.S., Szöcs, G., Schmidt, F., Francke, W., Löfstedt, C., andTóth, M. 1990. Electrophysiological and chemical analysis of sex pheromone communication system of the mottled umber,Erannis defoliaria (Lepidoptera: Geometridae).J. Chem. Ecol. 16:1887–1897.
König, W.A., Gehrke, B., Icheln, D., Evers, P., Dönnecke, J., andWang, W. 1992. New, selectively substituted cyclodextrins as stationary phases for the analysis of chiral constituents of essential oils.J. High Resol. Chromatogr. 15:367–372.
Millar, J.G., Giblin, M., Barton, D., Morrison, A., andUnderhill, E.W. 1990a. Synthesis and field testing of enantiomers of 6Z,9Z-cis-3,4-epoxydienes as sex attractants for geometrid moths.J. Chem. Ecol. 16:2317–2339.
Millar, J.G., Giblin, M., Barton, D., andUnderhill, E.W. 1990b. (3Z,6Z,9Z)-nonadecatriene and enantiomers of (3Z,9Z)-cis-6,7-epoxynonadecadiene as sex attractants for two geometrid and one noctuid moth species.J. Chem. Ecol. 16:2153–2166.
Mori, K., andBrevet, J.-L. 1991. Pheromone synthesis (CXXXIII). Synthesis of both the enantiomers of (3Z,9Z)-cis-6,7-epoxy-3,9-nonadecadiene, a pheromone component ofErannis defoliara. Synthesis 1125–1129.
Mori, K., andEbata, T. 1986. Synthesis of optically active pheromones with an epoxy ring, (+)-disparlure and both the enantiomers of (3Z,6Z)-cis-9,10-epoxy-3,6-heneicosadiene.Tetrahedron 42:3471–3478.
Mori, K., andTakeuchi, T. 1989. Synthesis of the enantiomers of (3Z,6Z)-cis-9,10-epoxy-1,3,6-henicosatriene and (3Z,6Z)-cis-9,10-epoxy-1,3,6-icosatriene, the new pheromone components ofHyphantria cunea. Liebigs Ann. Chem. 453–457.
Pietruszka, J., Hochmuth, D.H., Gehrke, B., Icheln, D., Runge, T., andKönig, W. 1992. Gas Chromatographic enantioseparation of allenes.Tetrahedron Asymmetrie 3:661–670.
Rumbo, E.R. 1981. An inexpensive electroantennogram amplifier, storage and display unit. CSIRO Div. Ent. Rep. No. 22, 1–19.
Steel, R., andTorrie, J. (eds.). 1960. Principles and Procedures of Statistics. McGraw-Hill, New York. 481 pp.
Tóth, M., Buser, H.R., Guerin, P.M., Arn, H., Schmidt, F., Francke, W., andSzöcs, G. 1992.Abraxas glossulariata L. (Lepidoptera: Geometridae): Identification of (3Z,6Z,9Z)-3,6,9-heptadecatriene and (6Z,9Z)-6,9-cis-3,4-epoxyheptadecadiene in the female sex pheromone.J. Chem. Ecol. 18:13–25.
Vojnits, A. 1980. Geometridae I. Fauna Hungariae XVI. 8. Akadémiai kiadó, Budapest. 157 pp (in Hungarian).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Szöcs, G., Tóth, M., Francke, W. et al. Species discrimination in five species of winter-flying geometrids (Lepidoptera) based on chirality of semiochemicals and flight season. J Chem Ecol 19, 2721–2735 (1993). https://doi.org/10.1007/BF00980703
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00980703