Summary
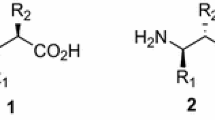
Methods for the synthesis of racemic and optically active title compounds are presented. Key step of these four-step procedures is the alkylation with 1-bromo-2-fluoroalkanes of glycine-ester-derived imines in anhydrous medium using lithium diisopropylamide as a base at low temperature or phase transfer catalyzed alkylation with 50% NaOH and triethylbenzylammoniumchloride as the phase transfer catalyst, respectively. Subsequent three-step deprotection gave the free acids in 13–33% overall yield. Deracemization ofγ-fluoro-α-aminobutyric acid methyl and ethyl esters withα-chymotrypsin was shown to give the (−)-enantiomers of the esters and (+)-γ-fluoro-α-aminobutyric acid in >98% ee, while from thetert-butylester the opposite stereochemical result was observed giving the (−)-acid with 88% ee. Optically activeγ-fluoro-α-amino acids were synthesized alternatively by phase transfer catalysis with N-benzyl-cinchonium chloride or using an auxiliary-directed asymmetric alkylation of the imine derived from (R)-(+)-camphor or (R)-(+)-2-hydroxypinan-3-one. These processes gave different enantiomers ofγ-fluoro-α-aminobutyric acid via a monomeric lithium enolate in the first or a dimeric lithium enolate in the second case, respectively. The enantiomeric excess can be improved by lithium/magnesium exchange.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alekseeva LV, Lundin BN, Burdé NL (1967) Synthesis and investigation of compounds with potential biological activity. Zh Obshch Khim 37: 1754–55 (J Gen Chem (USSR) (engl Transl) 37: 1671–1672)
Alvernhe G, Laurent A, Haufe G (1987) Triethylamine trishydrofluoride [(C2H5)3N·3HF]: a highly versatile source of fluoride ion for the halofluorination of alkenes. Synthesis 562–564
Bergman ED, Chun-Hsu L (1973) Organic fluorine compounds. Part 46.γ-Fluoroglutamic acid and fluorofolic acid. Synthesis: 44–46
Bory S, Dubois J, Gaudry M, Marquet A, Lacombe L, Weinstein S (1984) Resolution ofγ-methyl andγ-fluoroglutamic acids. Lack of stereoselectivity of Leucine Aminopeptidase with L-Leucyl-L-erythro-γ-substituted glutamates. J Chem Soc, Perkin Trans 1: 475–480
Buchanan RL, Dean FH, Pattison FLM (1962)γ-Fluoroglutamic acid. Can J Chem 40: 1571–1575
Butina D, Hudlický M (1980) The synthesis ofγ-fluoroisoleucine. J Fluorine Chem 16: 301–323
Cavalleri B, Bellasio E, Testa E (1966) Indagine su composti organici fluorurati a potenziale attività biologica. Derivati fluorurati dell'acidoα-amino eα,α′-diaminopimelico. Gazz Chim Ital 96: 253–263
Gershon H, Shanks L, Clarke DD (1978) Amino acid analogs, IV. 4-Fluoroisoleucine. J Pharm Sci 67: 715–717
Haufe G, Kröger S (1995) Synthesis ofγ-fluoro-α-amino acids. 4th International Congress on Amino Acids, Vienna, August 7–11th.
Haufe G, Alvernhe G, Laurent A, Ernet T, Goj O, Kröger S, Sattler A (1996) Bromofluorination of alkenes. Organic Syntheses (submitted)
Hudlický M (1960) The synthesis ofγ-fluoroglutamic acid. Tetrahedron Lett 14: 21–22
Hudlický M (1961) Organic compounds of fluorine, II. Fluorinated amino acids. Coll Czech Chem Commun 26: 1414–1421
Hudlický M, Merola JS (1990) New stereospecific syntheses and X-ray diffraction structures of (−)-d-erythro- and (+)-l-threo-4-fluoroglutamic acid. Tetrahedron Lett 31: 7403–7406
Hudlický M (1993) Stereospecific syntheses of all four stereoisomers of 4-fluroglutamic acid. J Fluorine Chem 60: 193–210
Kröger S (1996) Synthese fluorierter Aminosäuren. PhD Dissertation, Münster
Kukhar' VP, Soloshonok VA (1995) Fluorine-containing amino acids. Synthesis and properties. Wiley, Chilchester
Lettré H, Wölcke U (1967) Fluor-Derivate biogener aliphatischer Aminosäuren. Liebigs Ann Chem 708: 75–85
McIntosh JM, Leavitt RK, Mishra P, Cassidy KC, Drake JE, Chadha R (1988) Diastereoselective alkylation guided by electrophile-nucleophileπ-interactions. J Org Chem 53: 1947–1952
O'Donnel MJ, Boniece JM, Earp SE (1978) The synthesis of amino acids by phasetransfer reactions. Tetrahedron Lett 30: 2641–2644
O'Donnel MJ, Bennett WD, Wu S (1989) The stereoselective synthesis ofα-amino acids by phase-transfer catalysis. J Am Chem Soc 111: 2353–2355
Oguri T, Kawai N, Shioiri T, Yamada S-I (1978) Amino acids and peptides 29. A new efficient asymmetric synthesis ofα-amino acid derivatives with recycle of a chiral reagent-asymmetric alkylation of chiral Schiff base from glycin. Chem Pharm Bull (Jpn) 26: 803–808
Papageorgiou C, Borer X, French RR (1994) Calcineurin has a very tightbinding pocket for the chain of residue 4 to cyclosporin. Bioorg Med Chem Lett 4: 267–272
Raasch MS (1958) 5-Fluoronorvaline and 6-fluoronorleucine. J Org Chem 23: 1567–1568
Schöllkopf U (1993) Enantioselective synthesis of nonproteinogenic amino acids. Topics Current Chemistry 109: 65–84
Solladié-Cavallo A, Simon MC (1989) Enantioselective synthesis of optically pure natural S(+) or unnatural R(−) DABA. Tetrahedron Lett 30: 6011–6014
Solladié-Cavallo A, Simon-Wermeister MC, Schwarz J (1993) Diastereoselective monoalkylation of lithium and potassium enolates of a chiral imine of ethyl glycinate: the role of added salts. Organometallics 12: 3743–3747
Tolman V (1993) Chemistry of 4-fluorogluatmic acid. Part 1. A critical survey of its syntheses: an attempt to optimize reaction conditions for large-scale preparation. J Fluorine Chem 60: 179–183
Tolman V (1995) Syntheses of fluorine-containing amino acids by methods of classical amino acid chemistry. In: Kukhar' VP, Soloshonok VA (eds) Fluorine-containing amino acids. Synthesis and properties. Wiley, Chichester, pp 1–70
Tolman V, Vereš K (1966) Potential antimetabolites derived from 4-fluoroglutamic acid. Tetrahedron Lett 3909–3912
Tolman V, Vereš K (1967) Synthesis of certain monofluorinated aliphatic amino acids. Coll Czech Chem Commun 32: 4460–4469
Tolman V, Špronglová P (1983) Synthesis of 2-fluoropenoic acid derivatives. Coll Czech Chem Commun 48: 319–326
Unkeless JC, Goldman P (1970) Fluorinatedγ-aminobutyric acid. Enzymatic synthesis and biological activity of a potentially useful analogue. Mol Pharmacol 6: 46–53
Unkeless JC, Goldman P (1971) The diastereomers ofγ-fluorogluatmate: complementary structural analogues. Mol Pharmacol 7: 293–300
Yamada S-I, Oguri T, Shioiri T (1976) Asymmetric synthesis ofα-amino acid derivatives by alkylation of a chiral Schiff base. J Chem Soc, Chem Commun: 136–137
Yaozhong J, Guilan L, Changyou Z, Huri P, Lanjun W, Aiqiao M (1991) Asymmetric synthesis XIII: the stereocontrolled synthesis of (R)-α-amino acids via a double chiral induction. Synth Commun 21: 1087–1090
Yarovenko NN, Raksha MA (1959) Fluorination by means ofα-fluorinated amines. J Gen Chem USSR (engl Transl) 29: 2125–2128
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Haufe, G., Kröger, S. Syntheses ofγ-fluoro-α-amino acids. Amino Acids 11, 409–424 (1996). https://doi.org/10.1007/BF00807945
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00807945