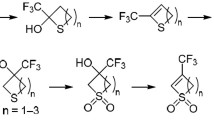
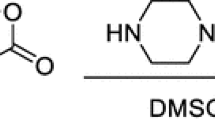
Abstract
The reactions of trifluoropyruvates with nitromethane and arylmagnesium bromides leading to trifluoromethyl-containing α-hydroxy acid esters were studied and various derivatives of the acids were obtained.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
P. Wang, L.-W. Feng, L. Wang, J.-F. Li, S. Liao, Y. Tang, J. Am. Chem. Soc., 2015, 137, 4626.
N. V. Sokolova, D. V. Vorobyeva, S. N. Osipov, T. P. Vasilyeva, V. G. Nenajdenko, Synthesis, 2012, 44, 130.
I. Deb, M. Dadwal, S. Mobin, I. Namboothiri, Org. Lett., 2006, 8, 1201.
I. Deb, P. Shanbhag, S. Mobin, I. Namboothiri, Eur. J. Org. Chem., 2009, 24, 4091.
A. V. Fokin, V. A. Komarov, A. I. Rapkin, K. V. Frosina, T. M. Potarina, K. I. Paseviba, O. V. Verenikin, Bull. Acad. Sci. USSR, Div. Chem. Sci., 1978, 27, 1692.
J.-L. Zhao, L. Liu, Y. Sui, Y.-L. Liu, D. Wang, Y.-J. Chen, Org. Lett., 2006, 8, 6127.
C. Zhang, D.-M. Zhuang, J. Li, S.-Y. Chen, X.-L. Du, J.-Y. Wang, J.-Y. Li, B. Jiang, J.-H. Yao, Org. Biomol.Chem., 2013, 11, 5621.
V. I. Dyachenko, A. F. Kolomiyets, A. V. Fokin, Russ. J. Org. Chem., 1992, 1345.
M. Montesinos-Magraner, C. Vila, G. Blay, I. Fernandez, M. C. Munoz, J. R. Pedro, Adv. Synth. Catal., 2015, 357, 3047.
G. Blay, I. Fernandez, A. Marco-Aleixandre, B. Monje, J. Pedro, R. Ruitz, Tetrahedron, 2002, 58, 8565.
E. A. Markova, A. F. Kolomiets, A. V. Fokin, Bull. Russ. Acad. Sci. Div. Chem. Sci., 1992, 41, 1102.
L. Bellamy, The Infra-red Spectra of Complex Molecules, Methuen and Co., Ltd, London, 1954.
G. Ricci, R. Ruzziconi, J.Org. Chem., 2005, 70, 611.
V. I. Saloutin, L. V. Saloutina. A. Ya. Zapevalov, O. N. Chupakhin, Russ. Chem. Bull., 2016, 65, 2163.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 8, pp. 1426–1432, August, 2018.
Rights and permissions
About this article
Cite this article
Vasilyeva, T.P., Vorobyeva, D.V. Synthesis of trifluoromethylated α-hydroxy acids and their derivatives based on alkyl trifluoropyruvates. Russ Chem Bull 67, 1426–1432 (2018). https://doi.org/10.1007/s11172-018-2235-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-018-2235-z