Summary
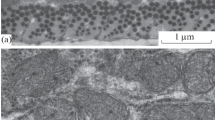
Various electron microscopical techniques have been applied to biopsy material obtained from patients suffering from mitral stenosis in order to characterize the subcellular organization of the hypertrophied papillary muscle. Small pieces of the same sample were processed for correlative transmission - (TEM) and scanning -(SEM) electron microscopical studies. TEM was carried out on conventionally fixed tissue with or withouten bloc staining with a Cu-Pb citrate solution, and on freeze fracture replicas, while cryofractured material was studied by SEM. Stereo electron micrographs of the Cu-Pb impregnated tissue and of the cryofractured material were especially useful for studying the spatial distribution and relationship between various cell organelles.
The myofilaments of the hypertrophied cells were arranged in a normal hexagonal pattern. Regions with irregular orientation of the myofibrils were occasionally seen. Accumulations of interfilamentous glycogen particles adjacent to the Z-bands were characteristic patterns of the contracted muscle cells. The extensive nexuses frequently observed in the subsarcolemmal regions may reflect functional alterations of the intercommunication between hypertrophied cells. The T-tubules were relatively few and irregularly distributed, and the complexity of the sarcotubular system (SR) revealed regional variations. Excellent visualization of the interior couplings between the SR and the T-tubules was achieved by studying thick sections of Cu-Pb impregnated tissue in the TEM.
The dense staining of the various intracellular membranes when compared with the almost unstained external membranes including the free cell surface, intercalated disc and T-system, strongly indicates differences in chemical and functional properties of the two membrane systems.En bloc staining resulted also in contrasted glycogen as well as components of the nucleolus and the heterochromatin. The biochemical basis for the selective staining remains obscure; it may be a result of binding of heavy metal ions to carboxyl groups of specific proteins, and/or it may represent deposits of lead phosphate.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anversa P, Olivetti G, Melissari M, Loud AV (1979) Morphometric study of myocardial hypertrophy induced by abdominal aortic stenosis. Lab Invest 40:341–349
Ayettey AS, Navaratnam V (1978) The T-tubule system in the specialized and general myocardium of the rat. J Anat 127:125–140
Breisch EA, White FC, Bloor CM (1984) Myocardial characteristics of pressure overload hypertrophy. A structural and functional study. Lab Invest 51:333–342
Brightman MW, Reese TS (1969) Junctions between intimately apposed cell membranes in the vertebrate brain. J Cell Biol 40:648–677
Dalen H (1987) An ultrastructural study of the myocardial cell mitochondria in the hypertrophied human papillary muscle Virchows Arch [Pathol Anat] (submitted)
Dalen H, Myklebust R, Sætersdal TS (1978) Cryofracture of paraffin-embedded heart muscle cells. J Microsc 112:139–151
Dalen H, Scheie P, Myklebust R, Sætersdal T (1983) An ultrastructural study of cryofractured myocardial cells with special attention to the relationship between mitochondria and sarcoplasmic reticulum. J Microsc 131:35–46
Dalen H, Sætersdal TS, Ødegården S (1987) Some ultrastructural features of the myocardial cells in the hypertrophied human papillary muscle. Virchows Arch [Pathol Anat] (in press)
Echlin P (1975) Sputter coating techniques for scanning electron microscopy. Scan Elect Microsc I:217–224
Edge MB, Walker SM (1970) Evidence for a structural relationship between sarcoplasmic reticulum and Z-lines in dog papillary muscle. Anat Rec 166:51–66
Ericsson JLE, Brunk UT, Arborgh B (1978) Fixation. In: Johannessen JV (ed) Electron Microscopy in Human Medicine, vol 1, McGraw-Hill International Book Company, New York, p 99
Fawcett DW, McNutt NC (1969) The ultrastructure of the cat myocardium. I. Ventricular papillary muscle. J Cell Biol 42:1–45
Ferrans VJ, Butany JW (1983) Ultrastructural pathology of the heart. In: Trump BF, Jones RT (eds) Diagnostic Electron Microscopy, vol. 4. John Wiley and Sons Inc, New York, p 319
Ferrans VJ, Thiedemann K-U (1983) Ultrastructure of the normal heart. In: Silver MD (ed) Cardiovascular Pathology, vol. 1. Churchill Livingstone New York, p. 31
Forbes MS, Hawkey LA, Sperelakis N (1984) The transverseaxial tubular system (TATS) of mouse myocardium: Its morphology in the developing and adult animal. Am J Anat 170:143–162
Forbes MS, Plantholt BA, Sperelakis N (1977) Cytochemical staining procedures selective for sarcotubular systems of muscle: Modifications and applications. J Ultrastruct Res 60:306–327
Forbes MS, Sperelakis N (1980) Structures located at the levels of the Z bands in mouse ventricular myocardial cells. Tissue & Cell 12:467–489
Forbes MS, Sperelakis N (1983) The membrane systems and cytoskeletal elements of mammalian myocardial cells. In: Dowben RM, Shay JW (eds) Cell and Muscle Mobility, vol. 3. Plenum Press, New York, p 89
Franzini-Armstrong C, Peachey LD (1981) Striated muscle - contractile and control mechanisms. J Cell Biol 91:166s–186s
Goodpasture C, Bloom SE (1975) Visualization of nucleolar organizer regions in mammalian chromosomes using silver staining. Chromosoma 53:37–50
Jones M, Ferrans VJ (1979) Myocardial ultrastructure in children and adults with congenital heart disease. In: Roberts WC (ed) Congenital Heart Disease in Adults. F.A. Davis Company. Philadelphia, p 501
Luft JH (1961) Improvements in epoxy resin embedding methods. J Biophys Biochem Cytol 9:409–414
Maron BJ, Ferrans VJ (1978) Ultrastructural features of hypertrophied human ventricular myocardium. Prog Cardiovasc Dis 21:207–238
McCallister LP, Mumaw VR, Munger BL (1974) Stereo ultrastructure of cardiac membrane systems in the rat heart. Scan Elect Microsc III 713–728
Moses HL, Rosenthal AS (1968) Pitfalls in the use of lead ion for histochemical localization of nucleoside phosphatases. J Histochem Cystochem 16:530–539
Myklebust R, Dalen H, Sætersdal TS (1975) A comparative study in the transmission electron microscope and scanning electron microscope of intracellular structures in sheep heart muscle cells. J Microsc 105:57–65
Myklebust R, Dalen H, Sæterdal TS (1980) A correlative transmission and scanning electron microscopic study of the pigeon myocardial cell. Cell Tissue Res 207:31–41
Olert J, Sawatzki G, Kling H, Gebauer J (1979) Cytological and histochemical studies on the mechanism of the selective silver staining of nucleolus organizer regions (NORs). Histochem 60:91–99
Page E, McCallister LP (1973) Quantitative electron microscopic description of heart muscle cells: Application to normal, hypertrophied and thyroxin-stimulated hearts. Am J Cardiol 31:172–181
Pébusque M-J, Seite R (1981) Electron microscopic studies of silver-stained proteins in nucleolar organizer regions: Location in nucleoli of rat sympathetic neurons during light and dark periods. J Cell Sci 51:85–94
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–212
Richter GW, Kellner A (1963) Hypertrophy of the human heart at the level of fine structure. An analysis and two postulates. J Cell Biol 18:195–206
Robinson TF, Winegrad S (1977) Variation of thin filament length in heart muscle. Nature 267:74–75
Robinson TF, Winegrad S (1979) The measurement and dynamic implications of thin filament lengths in heart muscle. J Physiol 286:607–619
Scales DJ (1983) III. Three-dimensional electron microscopy of mammalian cardiac sarcoplasmic reticulum at 80 kV. J Ultrastruct Res 83:1–9
Schwarzacher HG, Mikelsaar AV, Schnedl W (1978) The nature of Ag-staining of nucleolus organizer regions: Electron-and light-microscopic studies on human cells in interphase, mitosis and meiosis. Cytogenet Cell Genet 20:24–39
Segretain D, Rambourg A, Clermont Y (1981) Three dimensional arrangement of mitochondria and endoplasmic reticulum in the heart muscle fiber of the rat. Anat Rec 200:139–151
Sheldon CA, Friedman WF, Sybers HD (1976) Scanning electron microscopy of fetal and neonatal lamb cardiac cells. J Molec Cell Cardiol 8:853–862
Simpson FO, Rayns DG (1968) The relationship between the transverse tubular system and other tubules at the Z disc levels of myocardial cells in the ferret. Am J Anat 122:193–208
Sommer JR, Johnson EA (1979) Ultrastructure of the cardiac muscle. In: Berne RM, Sperelakis N, Geiger SR (eds) Handbook of Physiology. Section 2: The Cardiovascular System, vol. I: The Heart. The American Physiological Society, Bethesda, p 113
Sommer JR, Waugh RA (1976) The ultrastructure of the mammalian cardiac muscle cell -With special emphasis on the tubular membrane systems. A review. Am J Pathol 82:192–232
Sperelakis N, Forbes MS, Rubio R (1974) The tubular systems of myocardial cells: Ultrastructure and possible function. In: Dhalla NS (ed) Recent Advances in Studies on Cardiac Structure and Metabolism, vol. 4, Myocardial Biology. University Park Press, Baltimore, p 163
Sætersdal TS, Myklebust R, Skagseth E, Engedal H (1976) Ultrastructural studies on the growth of filaments and sarcomeres in mechanically overloaded human hearts. Virch Arch [Cell Pathol] 21:91–112
Sybers HD, Ashraf M (1974) Scanning electron microscopy of cardiac muscle. Lab Invest 30:441–450
Sybers HD, Ashraf M (1975) Scanning electron microscopy of the heart. In: Fleckenstein A, Rona G (eds) Recent Advances in Studies on Cardiac Structure and Metabolism, vol. 6: Pathophysiology and Morphology of Myocardial Cell Alternation. University Park Press, Baltimore, p 305
Sybers HD, Sheldon CA (1975) SEM techniques for cardiac cells in fetal, adult and pathologic heart. (1975). Scan Elect Microsc I:275–280
Thiéry G, Bergeron M (1976) Morphologie spatiale des mitochondries des tubes proximaux et distaux du néphron. Rev Can Biol 35:211–216
Thiéry G, Rambourg A (1976) A new staining technique for studying thick sections in the electron microscope. J Microsc Biol Cell 26:103–106
Trump BF, Smuckler EA, Benditt EP (1961) A method for staining epoxy sections for light microscopy. J Ultrastruct Res 5:343–348
Van Winkle WB (1977) The fenestrated collar of mammalian cardiac sarcoplasmic reticulum: A freeze-fracture study. Am J Anat 149:277–282
Watson ML (1958) Staining of tissue sections for electron microscopy with heavy metals. J Biophys Biochem Cytol 4:475–479
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dalen, H., Ødegården, S. & Sætersdal, T. The application of various electron microscopic techniques for ultrastructural characterization of the human papillary heart muscle cell in biopsy material. Vichows Archiv A Pathol Anat 410, 265–279 (1987). https://doi.org/10.1007/BF00711284
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00711284