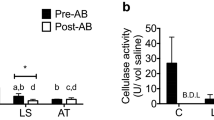
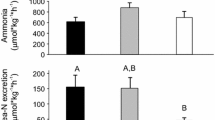
Summary
Intestinal nutrient transport in yearling coho salmon was characterized and adaptive changes in nutrient transport were described in relation to development, starvation, and environmental salinity. Salmon intestine exhibits a small transepithelial potential difference (TEP: −1.4 to 2.0 mV, mucosa ground) and low resistance (41 to 181 ohms·cm2) that varied with the region along the intestine, with starvation, and with environmental salinity. Addition of glucose or proline to the mucosal side of intestine caused a rapid increase in short-circuit current. Isotopic mucosalto-serosal net fluxes of glucose and proline were achieved across salmon intestine in the absence of transepithelial chemical or electrical gradients. A sleeve technique for measuring proline influx (Karasov and Diamond 1983a) was validated for use in salmon intestine. Comparison of total proline influx in different intestinal regions showed the following order (from highest to lowest rates): pyloric caeca ≫ anterior intestine > posterior intestine. Total proline influx was highest in April during the parr-smolt transformation.
The kinetics of Naα-dependent proline influx were altered by starvation and seawater adaptation. Starved fish exhibited a lowerK t but similarJ max in anterior intestine compared with values in fed fish. The effect of seawater adaptation on the kinetics of proline influx varied with the timing of entry into seawater, with length of seawater residence, and with season. Growth-inhibited SW stunts showed a reducedJ max of proline influx compared with that of normal SW smolts.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- FW :
-
freshwater (-adapted)
- SW :
-
seawater (-adapted)
- TEP :
-
transepithelial potential difference
- R :
-
transepithelial resistance
- I sc :
-
short-circuit current
- P a :
-
apparent passive permeability coefficient
- J max :
-
maximal influx
- K t :
-
half-saturation constant
References
Albus H, Siegenbeek van Heukelom J (1976) The electrophysiological characteristics of glucose absorption of the goldfish intestine as compared to mammalian intestines. Comp Biochem Physiol 54A:113–119
Ando M, Utida S, Nagahama H (1975) Active transport of chloride in eel intestine with special reference to seawater adaptation. Comp Biochem Physiol 51A:27–32
Boge G, Rigal A, Peres G (1979) A study of intestinal absorption in vivo and in vitro of different concentrations of glycine by the rainbow trout (Salmo gairdneri Richardson). Comp Biochem Physiol 62A:831–836
Boge G, Rigas A, Peres G (1981) A study of in vivo glycine absorption by fed and fasted rainbow trout (Salmo gairdneri). J Exp Biol 91:285–292
Buclon M (1974) Bioelectric potentials and the transfer of amino acids across the digestive epithelium of the tench (Tinca tinca L.). J Physiol (Paris) 68:157–180
Buclon M (1978) Comparative study of intestinal transport of amino acids in the tench and in the trout. Gastroenterol Clin Biol 2:339–340 (abstract)
Christensen HN (1975) Biological transport. WA Benjamin, Inc, London
Clarke WC, Nagahama Y (1977) Effect of premature transfer to seawater on growth and morphology of the pituitary, thyroid, pancreas, and interrenal in juvenile coho salmon (Oncorhynchus kisutch). Can J Zool 55:1620–1630
Cleland WW (1967) The statistical analysis of enzyme kinetic data. Adv Enzymol 29:1–32
Collie NL, Bern HA (1982) Changes in intestinal fluid transport associated with smoltification and seawater adaptation in coho salmon,Oncorhynchus kisutch (Walbaum). J Fish Biol 21:337–348
Cordier D, Maurice A (1956) Etude sur l'absorption intestinale des sucres chez l'Anguille (Anguilla vulgaris L.) vivant dans l'eau de mer ou dans l'eau douce. CR Soc Biol Paris 150:1957–1959
Duggleby RG (1981) A nonlinear regression program for small computers. Anal Biochem 110:9–18
Ferraris RP, Ahearn GA (1983) Intestinal glucose transport in carnivorous and herbivorous marine fishes. J Comp Physiol 152:79–90
Ferraris RP, Ahearn GA (1984) Sugar and amino transport in fish intestine. Comp Biochem Physiol 77A:397–413
Field M, Karnaky KJ, Smith PL, Bolton JE, Kinter WB (1978) Ion transport across the isolated intestinal mucosa of the winter flounderPseudopleuronectes americanus. I. Functional and structural properties of cellular and paracellular pathways for Na and Cl. J Membr Biol 41:203–208
Folmar LC, Dickhoff WW, Mahnken CVW, Waknitz FW (1982) Stunting and parr-reversion during smoltification of coho salmon (Oncorhynchus kisutch). Aquaculture 28:91–104
Frizzell RA, Schultz SG (1972) Ionic conductances of extracellular shunt pathway in rabbit ileum: influences of shunt on transmural sodium transport and electrical potential differences. J Gen Physiol 59:318–346
Gorbman A, Dickhoff WW, Mighell JL, Prentice EF, Waknitz FW (1982) Morphological indices of developmental progress in the parr-smolt coho salmonOncorhynchus kisutch. Aquaculture 28:1–19
Grau EG, Specker JL, Nishioka RS, Bern HA (1982) Factors determining the occurrence of the surge in thyroid activity in salmon during smoltification. Aquaculture 28:49–57
Habibi H, Ince BW, Matty AJ (1983) Effects of 17α-methyltestosterone and 17-β-oestradiol on intestinal transport and absorption of L-(14C)-leucine in vitro in rainbow trout (Salmo gairdneri). J Comp Physiol 151:247–252
Heinz E, Geck P (1978) The electrical potential difference as a driving force in Naα-linked cotransport of organic solutes, vol 1. In: Hoffman JF (ed) Membrane transport processes. Raven Press, New York, pp 13–30
Higgs DA, Fagerlund UHN, Eales JG, McBride JR (1982) Application of thyroid and steroid hormones as anabolic agents in fish culture. Comp Biochem Physiol 73B:143–176
Hokazono S, Tanaka Y, Katayama T (1979) Intestinal transport of L-lysine in rainbow trout,Salmo gairdneri. Bull Jpn Soc Sci Fish 45:845–848
Huang KC, Chen TST (1975) Comparative biological aspects of intestinal absorption. In: Csaky TZ (ed) Intestinal absorption and malabsorption. Raven Press, New York, pp 187–196
Ingham L, Arme C (1977) Intestinal absorption of amino acids by rainbow troutSalmo gairdneri (Richardson). J Comp Physiol 117:323–334
Karasov WH, Diamond JM (1983a) A simple method for measuring intestinal solute uptake in vitro. J Comp Physiol 152:105–116
Karasov WH, Diamond JM (1983b) Adaptive regulation of sugar and amino acid transport by vertebrate intestine. Am J Physiol 245:G443-G462
Karasov WH, Pond RSIII, Solberg DH, Diamond JM (1983) Regulation of proline and glucose transport in mouse intestine by dietary substrate levels. Proc Natl Acad Sci USA 80:7674–7677
Kitchin SE, Morris D (1971) The effect of acclimation temperature on amino acid transport in goldfish intestine. Comp Biochem Physiol 40A:431–443
Lind J, Munck BG, Olsen O (1980) Effects of dietary intake of sodium chloride on sugar and amino acid transport across isolated hen colon. J Physiol (Lond) 305:327–336
Loretz CA (1983) Ion transport by the intestine of the goby,Gillichthys mirabilis. Comp Biochem Physiol 75A:205–210
Miller DS, Kinter WB (1979) Pathways of cycloleucine transport in killifish small intestine. Am J Physiol 237:E567-E572
Munck BG (1981) Intestinal absorption of amino acids. In: Johnson LR (ed) Physiology of the gastrointestinal tract. Raven Press, New York, pp 1097–1122
Musch MW, Orellana SA, Kimberg LS, Field M, Halm DR, Kursny EJ, Frizzell RA (1982) Na−K−Cl co-transport in the intestine of a marine teleost. Nature 300:351–353
Pilch PF, Thompson PA, Czech MP (1980) Coordinate modulation ofd-glucose transport activity and bilayer fluidity in plasma membranes derived from control and insulin-treated adipocytes. Proc Natl Acad Sci (USA) 77:915–918
Powell DW (1979) Transport in large intestine. In: Giebisch G, Tosteson DC, Ussing HH (eds) Membrane transport in biology, vol IVB. Springer, New York Berlin, pp 781–809
Riggs TR (1964) Hormones and the transport of nutrients across cell membranes. In: Litwack G, Kritchevsky D (eds) Actions of hormones on molecular processes. John Wiley and Sons, New York, pp 1–57
Schultz SG (1979) Transport across small intestine. In: Giebisch G, Tosteson DC, Ussing HH (eds) Membrane transport in biology, vol IVB. Springer, New York Berlin, pp 749–780
Schultz SG (1981) Salt and water absorption in mammalian small intestine. In: Johnson LR (ed) Physiology of the gastrointestinal tract. Raven Press, New York, pp 983–989
Schultz SG, Curran PF (1970) Coupled transport of sodium and organic solutes. Physiol Rev 50:637–718
Schultz SG, Zalusky R (1964) Ion transport in isolated rabbit ileum II. The interaction between active sodium and active sugar transport. J Gen Physiol 47:1043–1059
Smith MW (1966) Sodium-glucose interactions in the goldfish intestine. J Physiol 182:559–573
Smith MW (1983) Membrane transport in fish intestine. Comp Biochem Physiol 75A:325–335
Stevens BR, Ross HJ, Wright EM (1982) Multiple transport pathways for neutral amino acids in rabbit jejunal brush border vesicles. J Membr Biol 66:213–225
Stokes RM, Fromm PO (1964) Glucose absorption and metabolism by the gut of rainbow trout. Comp Biochem Physiol 13:53–69
Westergaard H, Dietschy JM (1974) Delineation of the dimensions and permeability characteristics of the two major diffusion barriers to passive mucosal uptake in the rabbit intestine. J Clin Invest 54:718–732
Winne D (1973) Unstirred layer, source of biased Michaelis constant in membrane transport. Biochim Biophys Acta 298:27–31
Woo NYS, Bern HA, Nishioka RS (1978) Changes in body composition associated with smoltification and premature transfer to seawater in coho salmon (Oncorhynchus kisutch) and king salmon (O. tschawytscha). J Fish Biol 13:421–428
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Collie, N.L. Intestinal nutrient transport in coho salmon (Oncorhynchus kisutch) and the effects of development, starvation, and seawater adaptation. J Comp Physiol B 156, 163–174 (1985). https://doi.org/10.1007/BF00695770
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00695770