Abstract
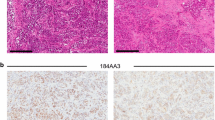
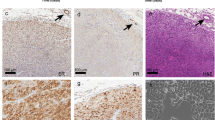
The non-steroidal antiestrogen tamoxifen (TAM) is successfully used to treat all stages of breast cancer in both pre- and postmenopausal women. Unfortunately, most women treated with TAM eventually develop resistant tumor recurrences which require intervention with a second-line endocrine therapy, or cytotoxic chemotherapy if the recurrence is completely endocrine insensitive. There is evidence that some recurrences may in fact be TAM stimulated. MCF-7 human breast cancer cells grown as solid tumors in athymic mice chronically treated with TAM reproducibly develop a TAM stimulated phenotype (Osborneet al., Eur J Cancer Clin Oncol 23: 1189-1196, 1987; Gottardis and Jordan, Cancer Res 48: 5183-5187, 1988; Osborneet al., J Natl Cancer Inst 83: 1477-1482, 1991; Wolfet al., J Natl Cancer Inst 85: 806-812, 1993). Tumors of this type may provide a useful model for a subset of therapeutic failures in the clinic. Therefore, we have extensively studied this model in an attempt to define the mechanism or mechanisms leading to TAM stimulated growth. In this paper we describe the characteristics of 4 TAM stimulated MCF-7 tumor variants. All of these tumors are growth stimulated by TAM, but vary in their response to estradiol (E2) treatment, and grow poorly in placebo treated hosts. All tumor variants express estrogen receptor (ER) RNA and protein, which at the RNA level appear to be down regulated by TAM, and to a greater extent by E2. All tumors also express epidermal growth factor receptor (EGFR) RNA, which is down regulated by TAM, and further down regulated by E2. However, among the tumor variants analyzed, ER and EGFR levels appear to be inversely related. Further, despite the expression of ER by all 4 TAM stimulated tumor variants, E2 induction of progesterone receptor expression is very weak or entirely absent.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Beatson GT: On the treatment of inoperable cases of carcinogen of the mamma: suggestions for a new method of treatment with illustrative cases. Lancet 2: 104–107, 162-167, 1896
Boyd S: On oophorectomy in cancer of the breast. Br Med J 2: 1161–1167, 1900
Dodds EC, Goldberg L, Lawson W, Robinson R: Oestrogenic activity of certain synthetic compounds. Nature 141: 247–248, 1938
Dodds EC, Lawson W, Noble RL: Biological effects of the synthetic oestrogenic substance 4:4′-dihydroxy-α:β-diethylstilbene. Lancet i: 1389–1391, 1938
Haddow A, Watkinson JM, Paterson E: Influence of synthetic oestrogens upon advanced disease. Br Med J 2: 393–398, 1944
Walpole A, Paterson E: Synthetic oestrogens in mammary cancer. Lancet ii: 783–789, 1949
Jensen EV, Jacobson HI: Basic guides to the mechanism of estrogen action. Recent Prog Horm Res 18: 387–414, 1962
Horwitz KB, McGuire WL, Pearson OH, Segaloff A: Predicting response to endocrine therapy in human breast cancer: A hypothesis. Science 189: 726–727, 1975
Clark GM, McGuire WL: Progesterone receptors and human breast cancer. Breast Cancer Res Treat 3: 157–163, 1983
Jordan VC, Wolf MF, Mirecki DM, Whitford DA, Welshons WV: Hormone receptor assays: clinical usefulness in the management of carcinoma of the breast. CRC Crit Rev Clin Lab Sci 26: 97–152, 1988
Muss HB: Endocrine therapy for advanced breast cancer: a review. Breast Cancer Res Treat 21: 15–26, 1992
Cole MP, Jones CJA, Todd IDH: A new antioestrogenic agent in late breast cancer. Br J Cancer 25: 270–275, 1971
Early Breast Cancer Trialists Collaborative Group: Systemic treatment of early breast cancer by hormonal, cytotoxic, or immune therapy: 133 randomized trials involving 31000 recurrences and 24000 deaths among 75000 women. Lancet 339: 1–15, 71-85, 1992
Lerner LJ, Jordan VC: Development of antiestrogens and their use in breast cancer. Eighth Cain Memorial Award Lecture. Cancer Res 50: 4177–4189, 1990
Legault-Poisson S, Jolivet J, Poisson R, Beretta-Piccoli M, Band PR: Tamoxifen-induced tumor stimulation and withdrawal response. Cancer Treat Rep 63: 1839–1841, 1979
Canney PA, Griffiths T, Latief TN, Priestman TJ: Clinical significance of tamoxifen withdrawal response. Lancet i: 36, 1987
Belani CP, Pearl P, Whitley NO, Aisner J: Tamoxifen withdrawal response. Report of a case. Archives of Internal Medicine 149: 449–450, 1989
Howell A, Dodwell DJ, Anderson H, Redford J: Response after withdrawal of tamoxifen and progestogens in advanced breast cancer. Ann Oncol 3: 611–617, 1992
Wolf DM, Langan-Fahey SM, Parker CP, McCague R, Jordan VC: Investigation of the mechanism of tamoxifen stimulated breast tumor growth with non-isomerizable analogs of tamoxifen and its metabolites. J Natl Cancer Inst 85: 806–812, 1993
Gottardis MM, Jordan VC: Development of tamoxifenstimulated growth of MCF-7 tumors in athymic mice after long-term antiestrogen administration. Cancer Res 48: 5183–5187, 1988
Osborne CK, Coronado EB, Robinson JP: Human breast cancer in the athymic nude mouse: cytostatic effects of longterm antiestrogen therapy. Eur J Cancer Clin Oncol 23: 1189–1196, 1987
Osborne CK, Coronado E, Allred DC, Wiebe V, DeGregorio M: Acquired tamoxifen (TAM) resistance: correlation with reduced breast tumor levels of tamoxifen and isomerization of trans-4-hydroxytamoxifen. J Natl Cancer Inst 83: 1477–1482, 1991
Osborne CK, Wiebe VJ, McGuire WL, Ciocca DR, DeGregorio M: Tamoxifen and the isomers of 4-hydroxytamoxifen in tamoxifen-resistant tumors from breast cancer patients. J Clin Oncol 10: 304–310, 1992
Wiebe VJ, Osborne CK, McGuire WL, DeGregorio MW: Identification of estrogenic tamoxifen metabolite(s) in tamoxifen-resistant human breast tumors. J Clin Oncol 10: 990–994, 1992
Robinson SP, Jordan VC: Antiestrogenic action of toremifene on hormone-dependent, independent, and heterogeneous breast tumor growth in the athymic mouse. Cancer Res 49: 1758–1762, 1989
Robinson SP, Langan FS, Jordan VC: Implications of tamoxifen metabolism in the athymic mouse for the study of antitumor effects upon human breast cancer xenografts. Eur J Cancer Clin Oncol 25: 1769–1776, 1989
Iino Y, Wolf DM, Langan-Fahey SM, Johnson DA, Ricchio M, Thompson ME, Jordan VC: Reversible control of oestradiol-stimulated growth of MCF-7 tumors by tamoxifen in the athymic mouse. Br J Cancer 64: 1019–1024, 1991
Chomczynski P, Sacchi N: Single-step method of RNA isolocation by acid-guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162: 156–159, 1987
Badley JE, Bishop GA, John TS, Frelinger JA: A simple rapid method for the purification of poly A+ RNA. BioTechniques 6: 114–116, 1988
Wolf DM, Jordan VC: A laboratory model to explain the sustained survival advantage observed in patients taking adjuvant tamoxifen therapy. In: Senn HJ, Goldhirsch A, Gelber RD, Turlmann B (eds) Adjuvant Therapy of Primary Breast Cancer IV. Recent Results in Cancer Research. Springer-Verlag, Berlin, 1993, Vol. 127, pp 22–33
Wolf DM, Arakawa RL, Friedl A, Jordan VC: Estradiol induced regression of the tamoxifen stimulated tumor variant MCF-7 TAM after prolonged exposure to tamoxifenin vivo (in preparation)
Wolf DM, Jordan VC: The estrogen receptor from a tamoxifen stimulated MCF-7 tumor variant contains a point mutation in the ligand binding domain. Breast Cancer Res Treat (this issue)
Martin PM, Berthois Y, Jensen EV: Binding of antiestrogens exposes an occult antigenic determinant in the human estrogen receptor. Proceedings of the National Academy of Sciences of the United States of America 85: 2533–2537, 1988
Gottardis MM, Wagner RJ, Borden EC, Jordan VC: Differential ability of antiestrogens to stimulate breast cancer cell (MCF-7) growthin vivo andin vitro. Cancer Res 49: 4756–4769, 1989
Dauvois S, Danielian PS, White R, Parker MG: Antiestrogen ICI 164,384 reduces cellular estrogen receptor content by increasing its turnover. Proc Natl Acad Sci USA 89: 4037–4041, 1992
Fritsch M, Wolf DM: Symptomatic side effects of tamoxifen therapy. In: Jordan VC (ed) Long-Term Tamoxifen Treatment for Breast Cancer. University of Wisconsin Press, Madison (in press)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wolf, D.M., Jordan, V.C. Characterization of tamoxifen stimulated MCF-7 tumor variants grown in athymic mice. Breast Cancer Res Tr 31, 117–127 (1994). https://doi.org/10.1007/BF00689682
Issue Date:
DOI: https://doi.org/10.1007/BF00689682