Abstract
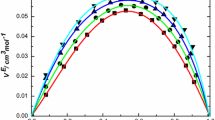
The sound velocities of aqueous solutions of methanol, ethanol, 2-propanol, tertbutanol and 2-butoxyethanol (BE) were measured over the whole mole fraction range at 25°C. The isentropic apparent and partial molar compressibilities, φK,S and φS,A were derived from these data. In the case ofBE, the isothermal partial molar compressibilities were also calculated. φK,S and φS,A for all alcohols except BE initially decrease slightly with the mole fraction and then increase sharply, especially with the higher members, to the value of the pure liquid. In the case of BE, φK,S and φS,A do not go through an initial minimum and the latter goes through a sharp maximum. The compressibilities of water in these mixtures are significantly lower than those of pure water itself. These data can be correlated with other properties and are consistent with the existence of microphase transitions in these aqueous organic mixtures.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
F. Franks and D. J. G. Ives,Quart. Rev. Chem. Soc. 20, 1 (1966).
F. Franks and D. J. Reid, inWater. A Comprehensive Treatise, Vol. 2, F. Franks, ed. (Plenum Press, New York, 1973), Chap. 5.
K. Tamura, M. Mackawa, and T. Yasunaga,J. Phys. Chem. 81, 2122 (1977).
N. Muller,J. Magn. Reson. 28, 203 (1977).
K. Iwasaki and T. Fujiyama,J. Phys. Chem. 81, 1908 (1977);83, 463 (1979).
C. de Visser, G. Perron, and J. E. Desnoyers,Can. J. Chem. 55, 856 (1977).
G. Roux, D. Roberts, G. Perron, and J. E. Desnoyers,J. Solution Chem. 9, 629 (1980).
G. Roux, G. Perron, and J. E. Desnoyers,J. Solution Chem. 7, 639 (1978).
C. D. Smith, C. E. Dorulan, and R. E. Barden,J. Coll. Interface Sci. 60, 488 (1977).
J. Lara, G. Perron, and J. E. Desnoyers,J. Phys. Chem. 85, 1600 (1981).
J. Lara, L. Avedikian, G. Perron, and J. E. Desnoyers,J. Solution Chem. 10, 301 (1981).
G. Perron, R. DeLisi, I. Davidson, S. Genereux, and J. E. Desnoyers,J. Coll. Interface Sci. 79, 432 (1981).
O. Kiyohara and G. C. Benson,J. Solution Chem. 10, 281 (1981).
R. Garnsey, R. Mahoney, and T. A. Litovitz,J. Chem. Phys. 64, 2073 (1964).
I. Davidson, G. Perron, and J. E. Desnoyers,Can. J. Chem. 59, 2212 (1981).
V. A. Del Grosso and C. W. Mader,J. Acoust. Soc. 52, 1442 (1972).
S. G. Bruun and A. Hvidt,Ber. Gunsengen Phys. Chem. 84, 930 (1977).
S. Nishikawa, M. Tanaka, and M. Mashiba,J. Phys. Chem. 85, 686 (1981).
S. Harada, T. Nakayima, T. Komatsu, and T. Nakagawa,J. Solution Chem. 7, 463 (1978).
M. V. Kaulgud and K. S. M. Rao,Indian J. Chem. 2237 (1978);J. Chem. Soc. Faraday I 75, 2237 (1979).
T. Nakayima, T. Komatsu, and T. Nakagawa,Bull. Chem. Soc. Japan. 48, 788 (1975).
S. Cabani, G. Conti, and E. Matteoli,J. Solution Chem. 8, 11 (1979).
H. Høiland and E. Vikingstad,Acta Chem. Scand. A30 692 (1976); see also H. Høiland,J. Solution Chem. 9, 857 (1980).
K. J. Patil and D. N. Raut,Indian J. Pure Appl. Phys. 18, 499 (1980).
J. H. Andreae, P. D. Edmonds, and J. F. McKellar,Acoustica 15, 74 (1965).
B. E. Conway and R. E. Verrall,J. Phys. Chem. 70, 3952 (1966).
L. H. Laliberté and B. E. Conway,J. Phys. Chem. 74, 4116 (1970).
J. E. Denoyers, M. Arel, G. Perron, and C. Jolicoeur,J. Phys. Chem. 73, 3346 (1969).
C. Jolicoeur, L. L. Lemelin, and R. Lapalme,J. Phys. Chem. 83, 2806 (1979).
J. E. Desnoyers, R. DeLisi, and G. Perron,Pure Appl. Chem. 52, 433 (1980).
A. Roux, in preparation.
J. E. Desnoyers and P. R. Philip,Can. J. Chem. 50, 1094 (1972).
J. L. Fortier, M. A. Simard, L. L. Lemelin, and C. Jolicoeur, unpublished data.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lara, J., Desnoyers, J.E. Isentropic compressibilities of alcohol-water mixtures at 25°C. J Solution Chem 10, 465–478 (1981). https://doi.org/10.1007/BF00652081
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00652081