Summary
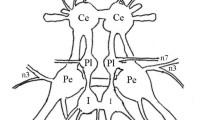
The response characteristics to light stimuli of first order ocellar interneurons in the bee (Apis mellifera carnica) were analysed with intracellular recordings. The neurons were identified using intracellular dye injection (Lucifer yellow).
The responses of L-(large) neurons can vary. They range from transient graded hyperpolarizations in response to light-On and -Off, (sometimes accompanied by tonic hyperpolarization during the light stimulus), to spontaneous tonic action potentials which are inhibited by light. Some neurons show both hyperpolarizations and action potentials.
The varying responses to light stimuli are dependent only on the state of the L-neuron and are not correlated systematically with particular types of L-neurons.
The experiments demonstrate that the action potentials are generated within the L-neurons and are not conducted antidromically from other neurons. The amplitude and frequency of the action potentials depend on the membrane potential of the L-neuron.
With light stimulation, or application of hyperpolarizing current, spikes can be generated within some of the L-neurons. Presumably non-spiking L-neurons are refractory due to maintained depolarization. They have membrane potentials of about −40 mV.
Action potentials in L-neurons were recorded only with high resistance microelectrodes. The vitality of the animal can not be correlated with the occurrence of spikes.
Functional significance of spontaneous tonic spike discharges in L-neurons is discussed in the context of behavioural experiments in the bee.
It is possible, that a given L-neuron uses either spiking or non-spiking signal transmission depending on the behavioural situation.
Although very fine microcapillary electrodes were used, no successful recordings or stainings of S-(small) neurons in the ocellar nerve were achieved. Therefore, results of non-identified ocellar interneurons which have been attributed to S-neurons should be critically reviewed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Autrum H, Metschl N (1963) Die Arbeitsweise der Ocellen der Insekten. Z Vergl Physiol 47:256–273
Autrum H, Zettler F, Järvilehto M (1970) Postsynaptic potentials from a single monopolar neuron of the ganglion opticum I of the blowflyCalliphora. Z Vergl Physiol 70:414–424
Burrows M, Siegler MVS (1976) Transmission without spikes between locust interneurons and motoneurones. Nature 262:222–224
Cajal SR (1918) Observaciones sobre la estructura de los ocelos y vias nerviosas ocelares de algunos insectos. Trab Lab Invest Biol Univ (Madrid) 16:109–131
Chappell RL, Dowling JE (1972) Neural organization of the median ocellus of the dragonfly. I. Intracellular electrical activity. J Gen Physiol 60:121–147
Chappell RL, Goodman LJ, Kirkham JB (1978) Lateral ocellar nerve projections in the dragonfly brain. Cell Tissue Res 190:99–114
Erber J (1980) Neural correlates of non-associative and associative learning in the honeybee. Verh Dtsch Zool Ges 1980:250–261
Erber J, Menzel R (1977) Visual interneurons in the median protocerebrum of the bee. J Comp Physiol 121:65–77
Erber J, Sandeman DC (1976) The detection of real and apparent motion by the crabLeptograpsus variegatus. II. Electrophysiology. J Comp Physiol 112:189–197
Goodman CS (1976) Anatomy of the ocellar interneurons of acridid grasshoppers. I. The large interneurons. Cell Tissue Res 175:184–203
Goodman CS, Williams JLD (1976) Anatomy of locust ocellar interneurons. II. The small interneurons. Cell Tissue Res 175:203–225
Goodman LJ (1970) The structure and function of the insect dorsal ocellus. Adv Insect Physiol 7:97–195
Goodman LJ (1975) The neural organization and physiology of the insect dorsal ocellus. InHorridge GA (ed) The compound eye and vision of insects. Oxford University Press, Oxford
Goodman LJ, Patterson JA, Mobbs PG (1975) The projection of ocellar neurons within the brain of the locust,Schistocerca gregaria. Cell Tissue Res 157:467–492
Gould JL (1975) Honey bee recruitment: The dance-language controversy. Science 189:685–693
Guy RG, Goodman LJ, Mobbs PG (1977) Ocellar connections with the ventral nerve cord in the locust,Schistocerca gregaria— Electrical and anatomical characteristics. J Comp Physiol 115:337–350
Guy RG, Goodman LJ, Mobbs PG (1979) Visual interneurons in the bee brain: Synaptic organization and transmission by graded potentials. J Comp Physiol 134:253–264
Heinzeller T (1976) Second-order ocellar neurons in the brain of the honeybee (Apis mellifera). Cell Tissue Res 171:91–99
Hengstenberg R (1977) Spike responses of “non-spiking” visual interneurone. Nature 270:338–340
Hertel H (1980) Chromatic properties of identified interneurons in the optic lobes of the bee. J Comp Physiol 137:215–231
Hoyle G (1955) Functioning of the insect ocellar nerve. J Exp Biol 32:397–407
Kondo H (1978) Efferent system of the lateral ocellus in the dragonfly: Its relationship with the ocellar afferent units, the compound eyes, and the wing sensory system. J Comp Physiol 125:341–349
Labhart T (1978) Electrophysiological investigations on the ocelli of the honeybee. Experientia 34:900
Laughlin SB (1973) Neural integration in the first optic neuropile of dragonflies. I. Signal amplification in dark adapted second order neurons. J Comp Physiol 84:335–355
Laughlin SB (1981) Neural principles in the peripheral visual system of invertebrates. In: Autrum H (ed) Handbook of sensory physiology, vol VII/6B. Springer, Berlin Heidelberg New York, pp 133–280
Menzel R (1974) Spectral sensitivity of monopolar cells in the bee lamina. J Comp Physiol 93:337–346
Metschl N (1963) Elektrophysiologische Untersuchungen an den Ocellen vonCalliphora. Z Vergl Physiol 47:230–255
Oertel D, Stuart AE (1981) Transformation of signals by interneurons in the barnackl's visual pathways. J Physiol 311:127–146
Pan KC, Goodman LJ (1977) Ocellar projections within the central nervous system of the worker honeybee (Apis mellifera). Cell Tissue Res 176:505–527
Patterson JA, Chappell RL (1980) Intracellular responses of procion filled cells and whole nerve cobalt impregnation in the dragonfly median ocellus. J Comp Physiol 139:25–39
Patterson JA, Goodman LJ (1974) Intracellular responses of receptor cells and second order cells in the ocelli of the desert locustSchistocerca gregaria. J Comp Physiol 95:237–250
Pearson KG (1976) Nerve cells without action potentials. In: Fentress JC (ed) Simpler network and behavior. Sinauer, Sunderland, MA, pp 99–110
Pearson KG, Fourtner CR (1975) Non-spiking interneurons in walking system of the cockroach. J Neurophysiol 38:33–52
Rosser BL (1974) A study of the afferent pathways of the dragonfly lateral ocellus from extracellularly recorded spike discharges. J Exp Biol 60:135–160
Ruck P (1961a) Electrophysiology of the insect dorsal ocellus. I. Origin of the components of the electroretinogram. J Gen Physiol 44:605–627
Ruck P (1961b) Electrophysiology of the insect dorsal ocellus. II. Mechanisms of generation and inhibition of impulses in the ocellar nerve of dragonflies. J Gen Physiol 44:629–639
Schricker B (1965) Die Orientierung der Honigbiene in der Dämmerung. Zugleich ein Beitrag zur Frage der Ocellenfunktion bei Bienen. Z Vergl Physiol 49:420–458
Shaw SR (1968) Organization of the locust retina. Symp Zool Soc(Lond)23:135–163
Stewart WW (1978) Functional connections between cells as revealed by dye-coupling with a highly fluorescent naphthalimide tracer. Cell 14:741–759
Stuart AE, Oertel D (1978) Neuronal properties underlying processing of visual information in the barnacle. Nature 275:287–290
Taylor CP (submitted) Contribution of compound eyes and ocelli to steering of locust in flight. I. Behavioral analysis. J Exp Biol
Wilson M (1978a) The functional organization of locust ocelli. J Comp Physiol 124:297–316
Wilson M (1978b) Generation of graded potential signals in the second order cells of locust ocellus. J Comp Physiol 124:317–331
Wilson M (1978c) The origin and properties of discrete hyperpolarizing potentials in the second order cells of locust ocellus. J Comp Physiol 128:347–358
Zettler F, Järvilehto M (1973) Active and passive axonal propagation of non-spike signals in the retina ofCalliphora. J Comp Physiol 85:89–104
Author information
Authors and Affiliations
Additional information
I am gtreatful to Benjamina Cwienczek, Alison Mercer, Joachim Erber and Randolf Menzel for their help with the manuscript. This work was supported by DFG grant Me 365/7
Rights and permissions
About this article
Cite this article
Milde, J. Graded potentials and action potentials in the large ocellar interneurons of the bee. J. Comp. Physiol. 143, 427–434 (1981). https://doi.org/10.1007/BF00609909
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00609909