Abstract
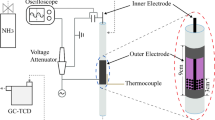
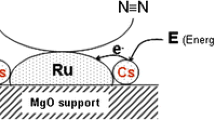
Ammonia synthesis from H2-N2 mixed gas was studied at room temperature in a glow-discharge plasma in the presence of metals or metal oxides. Magnesia (MgO) and calcia (CaO), which are oxides with solid basicity, revealed catalytic activity in the plasma synthesis of ammonia, although they are catalytically inactive in industrial ammonia synthesis. The acidic oxides (Al2O3, WO3, and SiO2-Al2O3) lead to the consumption of the reactant, i.e., the H2-N2 mixed gas. No ammonia was isolated. Metal catalysts showed higher activity than the above basic oxides. They have, however, different activities. The reaction was faster over the active materials than over sodium chloride (NaCl) or glass wool or in a blank reactor without any catalyst.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
A. K. Brewer and J. W. Westhaver,J. Phys. Chem. 33, 883 (1929).
A. K. Brewer and J. W. Westhaver,J. Phys. Chem. 34, 153 (1930).
A. K. Brewer and R. R. Miller,J. Am. Chem. Soc. 53, 2968 (1931).
E. Tiede and E. Hey,Chem. Ber B 66, 85 (1933).
M. Suzuki and H. Miyama,Nippon Kagaku Zasshi 75, 813, 900, 1253 (1954).
E. A. Rubtsova and E. N. Eremin,Zh. Fiz. Khim. 42, 2084 (1968).
A. H. Mal'tsev, L. A. Churina, and E. N. Eremin,Zh. Fiz. Khim. 42, 2334 (1968).
V. P. Lebedev, Yu. B. Fillippov, and N. I. Kobozev,Zh. Fiz. Khim. 24, 845 (1950).
E. N. Eremin, A. N. Mal'tsev, and V. M. Belova,Zh. Fiz. Khim. 43, 795 (1968).
Yu. B. Fillippov, V. V. Zapaman, and N. I. Kobozev,Zh. Fiz. Khim. 24, 1009 (1950).
T. E. Wolff, J. M. Berg, C. Warrick, K. O. Hodgson, R. H. Holm, and R. B. Frankel,J. Am. Chem. Soc. 100, 463 (1978).
M. Venugopalan and S. Veprek, “Plasma Chemistry” IV (Topics in Current chemistry No. 107) Springer, Berlin (1983), p. 1.
G. Y. Botchway and M. Venugopalan,Z. Phys. Chem. Neue Folge 120, 103 (1980).
K. S. Yin and M. Venugopalan,Plasma Chem. Plasma Process. 3, 343 (1983).
M. Kamo, Y. Sato, S. Matsumoto, and N. Setaka,J. Cryst. Growth 62, 642 (1983).
J. C. Angus, H. A. Will, and W. S. Stanko,J. Appl. Phys. 39, 2915 (1968).
B. V. Spitsin, L. L. Bouilov, and B. V. Derjaguin,J. Cryst. Growth 52, 219 (1981).
S. Matsumoto, Y. Sato, M. Kamo, and N. Setaka,J. Appl. Phys. 21, L183 (1982).
E. G. Spencer, P. H. Schmidt, D. C. Joy, and F. J. Sansalone,Appl. Phys. Lett. 29, 118 (1976).
M. Sokolowski, A. Sokolowskova, B. Gokiei, A. Michalski, A. Russek, and Z. Romanowski,J. Cryst. Growth 47, 421 (1979).
H. Vora and T. J. Moravec,J. Appl. Phys. 52, 6151 (1981).
C. Weissmantel, K. Bewilogua, D. Dietrich, H.-J. Erler, H.-J. Hinneberg, S. Klose, W. Nowick, and G. Reisse,Thin Solid Films 72, 19 (1980).
R. Mania, L. Stobierski, and R. Pampuch,Cryst. Res. Technol. 16, 785 (1981).
L. Holland and S. M. Ojha,Thin Solid Films 58, 107 (1979).
O. Nanura, H. Oyama, and Y. Sakamoto,Sci. Papers IPCR 75, 124 (1981).
K. Hiraoka, private communication; reported at the annual meeting of the chemical Society of Japan in April 1984, Tokyo, 1C29.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sugiyama, K., Akazawa, K., Oshima, M. et al. Ammonia synthesis by means of plasma over MgO catalyst. Plasma Chemistry and Plasma Processing 6, 179–193 (1986). https://doi.org/10.1007/BF00571275
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00571275