Summary
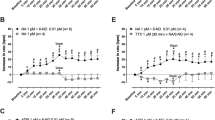
The role of adenosine 3′,5′-cyclic monophosphate (cAMP) in the release of noradrenaline from central neurones has been investigated by examining the effects of forskolin, 3-isobutyl-1-methylxanthine (IBMX), cis-6-(p-acetamidophenyl)-1,2,3,4,4a,10b-hexahydro-8,9-dimethoxy-2-methylbenzo[c] [1,6]-naphthyridine bis (hydrogenmaleinate) (AH21-132; a new phosphodiesterase inhibitor) and N6, O2′-dibutyryl-adenosine 3′,5′-cyclic monophosphate (dibutyryl-cAMP) on the outflow of tritiated compounds from rat and rabbit cerebral cortex slices preincubated with [3H]-noradrenaline. Forskolin, IBMX, AH21-132 and dibutyryl-cAMP produced a concentration-dependent increase in both basal and electrically-evoked efflux of tritium from rat and rabbit cortex slices. The increase in basal tritium efflux from rabbit cortex slices elicited by forskolin and IBMX could be attributed mainly to an increase in [3H]-DOPEG although a small increase in [3H]-noradrenaline was also observed. Forskolin and (when combined with noradrenaline) IBMX and AH21-132 increased the cAMP content of rat cortex slices at similar or somewhat higher concentrations that they increased tritium efflux. Neither forskolin nor IBMX or AH21-132 had any effect on the cocaine-sensitive uptake of [3H]-noradrenaline into synaptosomes prepared from rat or rabbit cortex. The effects of forskolin, IBMX and dibutyryl-cAMP on electrically-evoked overflow of tritium from rat and rabbit cortex slices were reduced when cocaine (10 μM) was present in the superfusion medium, although forskolin produced a similar increase in cAMP in the absence or presence of cocaine. It is suggested that cAMP may facilitate the normal process of noradrenaline release by nerve stimulation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Beisenherz G, Boltze HJ, Bücher T, Czok R, Garbade KH, Meyer-Arendt E, Pfleiderer G (1953) Reindarstellung von Enzymen. Z Naturforsch [C] 86:555–577
Burns TW, Langley PE, Terry BE, Bylund DB, Forte LR (1982) Alpha-2 adrenergic activation inhibits forskolin-stimulated adenylate cyclase activity and lipolysis in human adipocytes. Life Sci 31:815–821
Cubeddu L, Weiner N (1975) Release of norepinephrine and dopamine-β-hydroxylase by nerve stimulation. V. Enhanced release associated with a granular effect of a benzoquinolizine derivative with reserpine-like properties. J Pharmacol Exp Ther 193:757–774
Cubeddu L, Barnes E, Weiner N (1975) Release of norepinephrine and dopamine-β-hydroxylase by nerve stimulation. IV. An evaluation of a role for cyclic adenosine monophosphate. J Pharmacol Exp Ther 193:105–127
Cubeddu L, Hoffmann IS, Paris VB (1979) Effects of papaverine on the release and metabolism of dopamine in rat striatum. J Pharmacol Exp Ther 209:73–78
Dismukes RK, Mulder AH (1976) Cyclic AMP and α-receptor-mediated modulation of noradrenaline release from rat brain slices. Eur J Pharmacol 39:383–388
Ebstein RP, Seamon K, Creveling CR, Daly JW (1982) Release of norepinephrine from brain vesicular preparations: effects of an adenylate cyclase activator, forskolin, and a phosphodiesterase inhibitor. Cell Mol Neurobiol 2:179–192
Eitan A, Hershkowitz M (1977) The effects of dibutyryl cyclic AMP, theophylline and papaverine on the release of 3H-catecholamines from rat brain striatal and cortical synaptosomes. Eur J Pharmacol 46:323–327
Frandsen EK, Krishna G (1976) A simple ultrasensitive method for the assay of cyclic AMP and cyclic GMP in tissues. Life Sci 18:529–542
Graefe KH, Stefano FJE, Langer SZ (1973) Preferential metabolism of (-)-3H-norepinephrine through the deaminated glycol in the rat vas deferens. Biochem Pharmacol 22:1147–1160
Hedler L, Stamm G, Weitzell R, Starke K (1981) Functional characterization of central α-adrenoceptors by yohimbine diastereomers. Eur J Pharmacol 70:43–52
Katz IR, Smith D, Makman MH (1983) Forskolin stimulates the conversion of tyrosine to dopamine in catecholaminergic neural tissue. Brain Res 264:173–177
Metzger H, Lindner E (1981) The positive inotropic-acting forskolin, a potent adenylatecyclase activator. Arzneimittelforsch 31:1248–1250
Muller MJ, Baer HP (1983) Relaxant effects of forskolin in smooth muscle. Role of cyclic AMP. Naunyn-Schmiedeberg's Arch Pharmacol 322:78–82
Patrick RL, Barchas JD (1976) Dopamine synthesis in rat brain striatal synaptosomes. II. Dibutyryl cyclic adenosine 3′,5′-monophosphoric acid and 6-methyltetrahydropterine-induced synthesis increases without an increase in endogenous dopamine release. J Pharmacol Exp Ther 197:97–104
Pelayo F, Dubocovich ML, Langer SZ (1978) Possible role of cyclic nucleotides in regulation of noradrenaline release from rat pineal through presynaptic adrenoceptors. Nature 274:76–78
Rabe CS, Schneider J, McGee R (1982) Enhancement of depolarization-dependent neurosecretion from PC12 cells by forskolin-induced elevation of cyclic AMP. J Cyclic Nucleotide Res 8:371–384
Racké K, Rothländer M, Muscholl E (1982) Isoprenaline and forskolin increase evoked vasopressin release from rat pituitary. Eur J Pharmacol 82:97–100
Reichenbacher D, Reimann W, Starke K (1982) α-Adrenoceptor-mediated inhibition of noradrenaline release in rabbit brain cortex slices. Receptor properties and role of the biophase concentration of noradrenaline. Naunyn-Schmiedeberg's Arch Pharmacol 319:71–77
Reisine T, Chesselet MF, Glowinski J (1982) Striatal dopamine release in vitro: A β-adrenoceptor-regulated response not mediated through cyclic AMP. J Neurochem 39:976–981
Schoffelmeer ANM, Mulder AH (1983) 3H-noradrenaline release from rat neocortical slices in the absence of extracellular Ca2+ and its presynaptic alpha2-adrenergic modulation. Naunyn-Schmiedeberg's Arch Pharmacol 323:188–192
Schwertschlag U, Hackenthal E (1982) Forskolin stimulates renin release from the isolated perfused rat kidney.Eur J Pharmacol 84:111–113
Seamon KB, Daly JW (1983) Forskolin, cyclic AMP and cellular physiology. Trends in Pharmacol Sci 4:120–123
Siegl AM, Daly JW, Smith JB (1982) Inhibition of aggregation and stimulation of cyclic AMP generation in intact human platelets by the diterpene forskolin. Mol Pharmacol 21:680–687
Starke K (1977) Regulation of noradrenaline release by presynaptic receptor systems. Rev Physiol Biochem Pharmacol 77:1–124
Steiner AL, Kipnis DM, Utiger R, Parker C (1969) Radioimmunoassay for the measurement of adenosine 3′,5′-cyclic phosphate. Proc Nat Acad Sci 64:367–373
Stjärne L (1979) Role of prostaglandins and cyclic adenosine monophosphate in release. In: Paton DM (ed) The release of catecholamines from adrenergic neurons. Pergamon Press, Oxford, pp 111–142
Stjärne L, Bartfai T, Alberts P (1979) The influence of 8-Br 3′,5′-cyclic nucleotide analogs and of inhibitors of 3′,5′-cyclic nucleotide phosphodiesterase, on noradrenaline secretion and neuromuscular transmission in guinea-pig vas deferens. Naunyn-Schmiedeberg's Arch Pharmacol 308:99–105
Taube HD, Starke K, Borowski E (1977) Presynaptic receptor systems on the noradrenergic neurones of rat brain. Naunyn-Schmiedeberg's Arch Pharmacol 299:123–141
Wemer J, Schoffelmeer ANM, Mulder AH (1982) Effects of cyclic AMP analogues and phosphodiesterase inhibitors on K+-induced [3H]-noradrenaline release from rat brain slices and on its presynaptic α-adrenergic modulation. J Neurochem 39:349–356
Westfall TC, Kitay D, Wahl G (1976) The effect of cyclic nucleotides on the release of 3H-dopamine from rat striatal slices. J Pharmacol Exp Ther 199:149–157
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Markstein, R., Digges, K., Marshall, N.R. et al. Forskolin and the release of noradrenaline in cerebrocortical slices. Naunyn-Schmiedeberg's Arch. Pharmacol. 325, 17–24 (1984). https://doi.org/10.1007/BF00507049
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00507049