Abstract
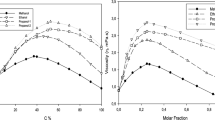
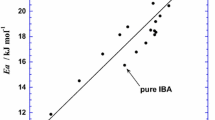
Viscosities of several mono-, di-, and trialkylamines have been measured in the temperature range 298 to 333 K. It is observed that viscosities are highly dependent on shape, size, and association through H-bond or through dipole. Following the transition state theory, energy, Gibbs free energy, and entropy of activation of viscous flow have been calculated. The values of expansion energy for these liquids have also been calculated using free volume theory, and subsequently amines have been classified as volume-restrained or energy-restrained liquids. The group contribution method of Van Velzen, Cardozo, and Langenkamp for estimating viscosity has been examined with the present and literature data, and the new group contribution increments ΔN i and ΔB i for amines have been evaluated.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. L. Oswal and A. V. Rao, Indian J. Chem. 24A:1024 (1985).
J. Shah, M. N. Vakharia, M. V. Pandya, G. D. Talele, K. G. Pathak, P. P. Palsanawala, and S. L. Oswal, Indian J. Tech. 26:383 (1988).
J. Shah, M. N. Vakharia, M. V. Pandya, A. T. Patel, J. H. Partiwala, P. P. Palsanawala, and S. L. Oswal, Indian J. Tech. 27:306 (1989).
S. L. Oswal and A. T. Patel, Int. J. Thermophys. 12:821 (1991).
D. Van Velzen, R. L. Cardozo, and H. Langenkamp, Ind. Eng. Chem. Fundam. 11:20 (1972).
S. L. Oswal and M. V. Rathnam, Can. J. Chem. 62:2581 (1984).
J. A. Riddick, W. B. Hunger, and T. K. Sakano, Organic Solvents Physical Properties and Methods of Purification, 4th ed. (Wiley Interscience, New York, 1986).
R. H. Stokes, J. Chem. Thermodyn. 5:379 (1973).
C. Klofutar, S. Paljk, and R. Malnersic, J. Chem. Soc. Faraday Trans. I 78:3693 (1982).
T. M. Letcher, J. Chem. Thermodyn. 4:159, 551 (1972).
A. Krishnaiah and P. R. Naidu, Acta Chem. Acad. Sci. Hung. 104:295 (1980).
J. M. Costello and S. T. Bowden, Rec. Trav. Chim. 78:391 (1970).
R. C. Weast, Hand Book of Chemistry and Physics, 59th ed. (Chemical Rubber Co., Ohio, 1979).
R. Philippe, G. Delmas, and M. Couchen, Can. J. Chem. 56:370 (1978).
C. Klofutar, S. Paljk, and D. Kremser, J. Inorg. Nucl. Chem. 37:1729 (1975).
A. S. Kertes and F. Grauber, J. Phys. Chem. 77:3107 (1973).
J. K. Shah, K. J. Dewitt et al., J. Chem. Eng. Data 14:333 (1969).
F. Kohler, H. Atrops, H. Kalali, E. Liebermann, E. Wilhelm, F. Ratkovics, and T. Salamon, J. Phys. Chem. 85:2520 (1981).
U. Langguth and H. J. Bittrich, Z. Phys. Chem. (Leipzig) 244:327 (1970).
J. Timmermans and Mme. Hennaut-Roland, J. Chim. Phys. 32:501, 589 (1935).
R. C. Reid, J. M. Prausnitz, and B. E. Poling, The Properties of Gases and Liquids, 4th ed. (McGraw-Hill, New York, 1986).
J. N. Friend and W. D. Hargreaves, Phil. Mag. 35:57, 619 (1944).
M. Sreenivasulu and P. R. Naidu, Indian J. Chem. Sect. A 19:470 (1980).
Virginia Chemicals, Inc., Virginia Amines (Virginia Chemicals, West Norfolk, 1967).
T. M. Letcher and J. W. Bayles, J. Chem. Eng. Data 16:266 (1971).
S. Glasstone, K. J. Laidler, and H. Eyring, The Theory of Rate Processes (McGraw-Hill, New York, 1941).
R. J. Moore, P. Gibbs, and H. Eyring, J. Phys. Chem. 57:172 (1953).
A. J. Batschinski, Z. Phys. Chim. 84:643 (1913).
J. H. Hildebrand, Viscosity and Diffusivity: A Predictive Treatment (Wiley Interscience, New York, 1977).
F. B. Macedo and T. A. Litovitz, J. Chem. Phys. 42:245 (1965).
R. S. Stairs, Can. J. Chem. 59:2170 (1981).
D. van Velzen, R. L. Cardozo, and H. Langenkamp, Liquid Viscosities and Chemical Constitution of Organic Compounds: A New Correlation and a Compilation of Literature Data, Euratom 4735e (Joint Nuclear Research Center, Ispra Establishment, Italy, 1972).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Oswal, S.L., Sindhe, R.G., Patel, A.T. et al. Study of viscosity of mono-, di-, and trialkylamines. Int J Thermophys 13, 617–628 (1992). https://doi.org/10.1007/BF00501944
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00501944