Summary
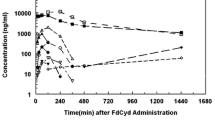
2′-deoxycoformycin (2′-dCF; Pentostatin), a stoichiometric inhibitor of mammalian adenosine deaminase (ado deaminase), exhibits immunosuppressive and antilymphocytic activity in animal test systems. A clinical pharmacology/phase I study of 2′-dCF administered as a single agent has been completed (18 patients). Dose levels ranged from 0.1 mg/kgx1 to 0.25 mg/kg/dayx5; ado deaminase and 2′-dCF were measured spectrophotometrically. Plasma decay curves were bi-exponential (α and βt1/2 values about 1 and 10 h respectively). Recovery of unchanged 2′-dCF from urine (48 h) was 32%–48% of the administered drug. Major toxic manifestations were lymphocytopenia (all patients) and urate nephropathy (1 patient, with subsequent patients in the series receiving allopurinol, 300 mg/day). Three partial responses were seen in seven patients with acute lymphocytic leukaemia receiving 0.25 mg 2′-dCF/kg/dayx5.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adamson RH, Chassin MM, Chirigos MA, Johns DG (1978) Some aspects of the pharmacology of the adenosine deaminase inhibitors, 2′-deoxycoformycin and erythro-9-(2-hydroxy-3-nonyl) adenine. In: Current chemotherapy: Proceedings of the Tenth International Congress of Chemotherapy, vol II. American Society for Microbiology, Washington, p 1116
Agarwal RP, Spector T, Parks RE Jr (1977) Inhibition of adenosine deaminase by various inhibitors. Biochem Pharmacol 26:359
Benjamin RS, Plunkett W, Keating MJ, Fenn LG, Hug V, Nelson JA, Bodey GP, Freireich EJ (1980) Phase I and biochemical pharmacological studies of deoxycoformycin. Proc Am Soc Clin Oncol 16: C-75, 337
Böyun A (1968) Isolation of mononuclear cells and granulocytes from human blood. Scand J Clin Lab Invest [Suppl 97] 21:77
Brady TG (1942) Adenosine deaminase. Biochem J 36:478
Brady TG, O'Donovan CI (1965) A study of tissue distribution of adenosine deaminase in six mammal species. Comp Biochem Physiol 14:101
Chassin MM, Louie AC, Chirigos MA, Adamson RH, Johns DG (1977) Adenosine deaminase inhibition for immunosuppression. N Engl J Med 296:1232
Chassin MM, Louie AC, Chirigos MA, Adamson RH, Johns DG (1978) Immune dysfunction produced by the adenosine deaminase inhibitor 2′-deoxycoformycin. Clin Res 26:513A
Chassin MM, Adamson RH, Zaharevitz DW, Johns DG (1979) Enzyme inhibition titration assay for 2′deoxycoformycin and its application to the study of the relationship between drug concentration and tissue adenosine deaminase in dogs and rats. Biochem Pharmacol 28:1849
Conway EJ, Cook R (1939) The deaminases of adenosine and adenylic acid in blood and tissues. Biochem J 33:479
Dissing J, Knudsen B (1972) Adenosine deaminase deficiency and combined immunodeficiency syndrome. Lancet 2:1316
Frieden C, Gilbert HR, Miller WH, Miller RL (1979) Adenylate deaminase: potent inhibition by 2′-deoxycoformycin 5′-phosphate. Biochem Biophys Res Commun 91:278
Giblett ER, Anderson JE, Cohen F, Pollara B, Meuwissen HJ (1972) Adenosine deaminase deficiency in two patients with severely impaired cellular immunity. Lancet 2:1067
Hall JG (1963) Adenosine deaminase activity in lymphoid cells during antibody production. Aust J Exp Biol Med Sci 41:93
Hirschhorn R (1979) Clinical delineation of adenosine deaminase deficiency. In: Enzyme defects and immune dysfunction (Ciba Foundation Symposium 68). Excerpta Medica Amsterdam, p 35
Hovi T, Smyth JF, Allison AC, Williams SC (1976) Role of adenosine deaminase in lymphocyte proliferation. Clin Exp Immunol 23:395
Kufe D, Major P, Agarwal R, Reinherz E, Frei E III (1980) Phase I–II trial of deoxycoformycin in T-cell malignancies. Proc Am Soc Clin Oncol 16:C-39, 328
LePage GA, Worth LS, Kimball AP (1976) Enhancement of the antitumor activity of arabinofuranosyladenine by 2′-deoxycoformycin. Cancer Res 36:1481
Lutz RJ, Dedrick RL, Straw JA, Hart MM, Klubes P, Zaharko DS (1975) The kinetics of methotrexate distribution in spontaneous canine lymphosarcoma. J Pharmacokinet Biopharm 3:77
McConnell WR, Suling WJ, Rice LS, Shannon WM, Hill DL (1978) Use of microbiologic and enzymatic assays in studies on the disposition of 2′-deoxycoformycin in the mouse. Cancer Treat Rep 62:1153
Nelson DJ, LaFon S, Lambe CU (1979) Alternations of nucleotide pools in vivo by adenosine deaminase inhibitors, 2′-deoxycoformycin and erythro-9-(2-hydroxyl-3-nonyl)adenine. In: Inborn errors of specific immunity. Academic Press, New York, p 327
Paine RM, Smyth JF, Harrap KR (1980) Biochemical consequences of treatment with the adenosine deaminase inhibitor 2′-deoxycoformycin. In: Proceedings of the International Symposium on Purine Metabolism in Man. Plenum Press, New York, p 365
Smyth JF (1976) The significance of adenosine deaminase activity in leukemia. Proc Am Assoc Cancer Res 12:235
Smyth JF, Harrap KR (1975) Adenosine deaminase activity in leukemia Br J Cancer 31:544
Smyth JF, Young RC, Young DM (1978) In vivo toxicity to lymphoid tissue by 2′-deoxycoformycin. Cancer Chemother Pharmacol 1:49
Venner PM, Glazer RI (1979) The metabolism of 2′-deoxycoformycin by L1210 cells in vitro. Biochem Pharmacol 28:3239
Woo PWK, Dion HW, Lange SM, Dahl LF, Durham LJA (1974) Novel adenosine and ara-A deaminase inhibitor, (R)-3-(2-deoxy-β-d-erythro-pentofuranosyl)-3,6,7,8-tetrahydro-imidazo (4,5-d)(1,3)diazepin-8-ol. Journal of Heterocyclic Chemistry 11:641
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smyth, J.F., Paine, R.M., Jackman, A.L. et al. The clinical pharmacology of the adenosine deaminase inhibitor 2′-deoxycoformycin. Cancer Chemother. Pharmacol. 5, 93–101 (1980). https://doi.org/10.1007/BF00435411
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00435411