Abstract
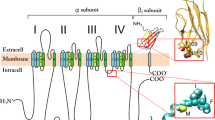
Na+ currents recorded from Xenopus oocytes expressing the Na+ channel α subunit alone inactivate with two exponential components. The slow component predominates in monomeric channels, while coexpression with the β1 subunit favors the fast component. Macropatch recordings show that the relative rates of these components are much greater than previously estimated from two-electrode measurements (≈30-fold vs ≈5-fold). A re-assessment of steady-state inactivation, h ∞(V), shows that there is no depolarized shift of the slow component, provided a sufficiently long prepulse duration and repetition interval are used to achieve steady-state entry and recovery from inactivation, respectively. Deletion mutagenesis of the β1 subunit was used to define which regions of the subunit are required to modulate inactivation kinetics. The carboxy tail, comprising the entire predicted intracellular domain, can be deleted without a loss of activity; whereas small deletions in the extracellular amino domain or the signal peptide totally disrupt function.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bennett PB, Makita N, George AL (1993) A molecular basis for gating mode transitions in human skeletal muscle sodium channels. FEBS Lett 326:21–24
Cannon SC, Brown RH, Corey DP (1991) A sodium channel defect in hyperkalemic periodic paralysis: potassium-induced failure of inactivation. Neuron 6:619–626
Cannon SC, Brown RH, Corey DP (1993) Theoretical reconstruction of myotonia and paralysis caused by incomplete inactivation of sodium channels. Biophys J 65:270–288
Cannon SC, McClatchey AI, Gusella JF (1993) Modification of the Na current conducted by the rat skeletal muscle α subunit by co-expression with a human brain β subunit. Pflügers Arch 423:155–157
Catterall WA (1992) Cellular and molecular biology of voltage-gated sodium channels. Physiol Rev 72:S15-S48
Fernandez JM, Fox AP, Kranse S (1984) Membrane patches and whole-cell membranes: a comparison of electrical properties in rat clonal pituitary cells. J Physiol (Lond) 356:565–585
Fleig APCR, Rayner MD (1994) Kinetic mode switch of rat brain IIA Na channels in Xenopus oocytes excised macropatches. Pflügers Arch 427:399–405
Ho SN, Hunt HD, Horton RM, Pullen JK, Pease LR (1989) Site-directed mutagenesis by overlap extension using the polymerase chain reaction. Gene 77:55–59
Isom LL, DeJongh KS, Patton DE, Reber BFX, Offord J et al. (1992) Primary structure and functional expression of the β1 subunit of the rat brain sodium channels. Science 256:839–842
Isom LL, DeJongh KS, Catterall WA (1994) Auxiliary subunits of voltage-gated ion channels. Neuron 12:1183–1194
Ji S, Sun W, George AL, Horn R, Barchi RL (1994) Voltage-dependent regulation of modal gating in the rat SkM1 sodium channel expressed in Xenopus oocytes. J Gen Physiol 104:625–643
Krafte DS, Snutch TP, Leonard JP, Davidson N, Lester HA (1988) Evidence for the involvement of more than one mRNA species in controlling the inactivation process of rat and rabbit brain Na channels expressed in Xenopus oocytes. J Neurosci 8:2859–2868
Krafte DS, Goldin AL, Auld VJ, Dunn RJ, Davidson N, Lester H (1990) Inactivation of cloned Na channels expressed in Xenopus oocytes. J Gen Physiol 96:689–706
Kupper J, Bowlby MR, Marom S, Levitan IB (1995) Intracellular and extracellular amino acids that influence C-type inactivation and its modulation in a voltage-dependent potassium channel. Pflügers Arch 430:1–11
Makita N, Bennett PB, George AL (1994) Voltage-gated Na+ channel β1 subunit mRNA expressed in adult human skeletal muscle, heart, and brain is encoded by a single gene. J Biol Chem 269:7571–7578
McClatchey AI, Cannon SC, Slaugenhaupt SA, Gusella JF (1993) The cloning and expression of a sodium channel β1-subunit cDNA from human brain. Hum Mol Genet 2:745–749
Moorman JR, Kirsch GE, Van Dongen AMJ, Joho RH, Brown AM (1990) Fast and slow gating of sodium channels encoded by a single mRNA. Neuron 4:243–252
Patton DE, Isom LL, Catterall WA, Goldin AL (1994) The adult rat brain β1 subunit modifies activation and inactivation gating of multiple sodium channel α subunits. J Bio Chem 269:17649–17655
Scheuer T, Auld V, Boyd S, Offord J, Dunn R, Catterall WA (1990) Functional properties of rat brain sodium channels expressed in a somatic cell line. Science 247:854–858
Simoncini L, Stühmer W (1987) Slow sodium channel inactivation in rat fast-twitch muscle. J Physiol (Lond) 383:327–337
Stühmer W, Methfessel C, Sakmann B, Noda M, Numa S (1987) Patch clamp characterization of sodium channels expressed from rat brain cDNA. Eur Biophys J 14:131–138
Stühmer W, Conti F, Suzuki H, Wang X, Noda M et al. (1989) Structural parts involved in activation and inactivation of the sodium channel. Nature 339:597–603
Tejedor FJ, Catterall WA (1988) Site of covalent attachment of α-scorpion toxin derivatives in domain I of the sodium channel α subunit. Proc Natl Acad Sci USA 85:8742–8746
Trimmer JS, Cooperman SS, Tomiko SA, Zhou J, Crean SM et al. (1989) Primary structure and functional expression of a mammalian skeletal muscle sodium channel. Neuron 3:33–49
Ukomadu C, Zhou J, Sigworth FJ, Agnew WS (1992) μ1 Na+ channels expressed transiently in human embryonic kidney cells: biochemical and biophysical properties. Neuron 8:663–676
Zhou J, Potts JF, Trimmer JS, Agnew WS, Sigworth FJ (1991) Multiple gating modes and the effect of modulating factors on the μ1 sodium channel. Neuron 7:775–785
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chen, C., Cannon, S.C. Modulation of Na+channel inactivation by the β1 subunit: A deletion analysis. Pflugers Arch. 431, 186–195 (1995). https://doi.org/10.1007/BF00410190
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00410190